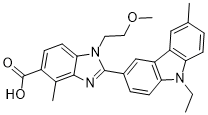
BAY-1316957 (BAY1316957) is a novel, highly potent, selective, and orally bioavailable antagonist of human prostaglandin E2 receptor subtype 4 (hEP4 receptor) with the potential to be used for the Treatment of Endometriosis. It inhibits hEP4-R with an IC50 of 15.3 nM. The presence and growth of endometrial tissue outside the uterine cavity in endometriosis patients are primarily driven by hormone-dependent and inflammatory processes-the latter being frequently associated with severe, acute, and chronic pelvic pain. The EP4 subtype of prostaglandin E2 (PGE2) receptors (EP4-R) is a particularly promising anti-inflammatory and antinociceptive target as both this receptor subtype and the pathways forming PGE2 are highly expressed in endometriotic lesions. High-throughput screening resulted in the identification of benzimidazole derivatives as novel hEP4-R antagonists. Careful structure-activity relationship investigation guided by rational design identified a methyl substitution adjacent to the carboxylic acid as an appropriate means to accomplish favorable pharmacokinetic properties by reduction of glucuronidation. Further optimization led to the identification of benzimidazolecarboxylic acid BAY 1316957, a highly potent, specific, and selective hEP4-R antagonist with excellent drug metabolism and pharmacokinetics properties. Notably, treatment with BAY 1316957 can be expected to lead to prominent and rapid pain relief and significant improvement of the patient's quality of life.
Physicochemical Properties
| Molecular Formula | C27H27N3O3 |
| Molecular Weight | 441.5216 |
| Exact Mass | 441.205 |
| CAS # | 1613264-40-6 |
| PubChem CID | 90202558 |
| Appearance | White to off-white solid powder |
| LogP | 4.9 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 33 |
| Complexity | 704 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O(C)CCN1C2C=CC(C(=O)O)=C(C)C=2N=C1C1C=CC2=C(C=1)C1C=C(C)C=CC=1N2CC |
| InChi Key | FHXIZAPGGULPIK-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C27H27N3O3/c1-5-29-22-9-6-16(2)14-20(22)21-15-18(7-10-23(21)29)26-28-25-17(3)19(27(31)32)8-11-24(25)30(26)12-13-33-4/h6-11,14-15H,5,12-13H2,1-4H3,(H,31,32) |
| Chemical Name | 2-(9-Ethyl-6-methyl-9H-carbazol-3-yl)-1-(2-methoxyethyl)-4-methyl-1H-benzimidazole-5-carboxylic acid |
| Synonyms | BAY1316957; BAY 1316957; BAY-1316957 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Caco-2 cells demonstrate the excellent solubility and permeability of BAY-1316957 (Compound 32) [1]. |
| ln Vitro |
Caco-2 cells demonstrate the excellent solubility and permeability of BAY-1316957 (Compound 32) [1]. BAY-1316957 is a highly potent and selective full antagonist of the human EP4 receptor, with an IC₅₀ of 15.3 ± 10.7 nM in a cell-based functional cAMP assay. It maintains excellent selectivity over human EP2 and DP1 receptors, with no significant antagonistic activity detected against these receptors. The compound also shows favorable potency across species (rat, mouse, cynomolgus monkey) based on earlier lead optimization data. [1] Metabolic stability studies in human, rat, mouse, dog, and monkey hepatocytes indicate that the primary biotransformation pathway is direct glucuronidation of the carboxylic acid moiety, mainly catalyzed by UGT1A1 and to a lesser extent UGT1A3. [1] |
| ln Vivo |
In the dmPGE2 pain model, treatment with BAY-1316957 (Compound 32; 0.2–5 mg/kg; oral; once) dramatically decreased mechanical allodynia [1]. In Wistar rats, the pharmacokinetic characteristics of BAY-1316957 (Compound 32) demonstrated a lengthy half-life, low clearance, and high bioavailability (F%=90%). Research on the metabolism of BAY-1316957 (compound 32) in hepatocytes from humans, rats, mice, dogs, and monkeys revealed that the production of acyl glucuronides is another frequent and important pathway for biotransformation, mostly catalyzed and transformed by UGT1A1 to a lesser extent by UGT1A3 [1]. Oral administration of BAY-1316957 (5 mg/kg) significantly reduced mechanical allodynia in a rat model of dmPGE2-induced inflammatory pain, as measured by increased paw withdrawal thresholds compared to vehicle-treated controls. This demonstrates its potent anti-nociceptive and anti-inflammatory activity in vivo. [1] |
| Enzyme Assay |
A cell-based functional assay measuring antagonism of human EP4 receptor activity was used. Frozen cells expressing the receptor were incubated with test compounds and stimulated with PGE2 agonist. Agonist-induced cAMP production was measured using a non-radioactive HTRF assay based on competition between endogenous cAMP and a fluorescently labeled cAMP-d2 donor for binding to a Europium cryptate-labeled anti-cAMP antibody. The assay was performed in 384-well or 1536-well formats, and IC₅₀ values were determined by 4-parameter fitting. [1] Similar assay formats were used for species-specific EP4 receptor activity (rat, mouse, cynomolgus monkey) and for selectivity screening against human EP2 and DP1 receptors. [1] |
| Cell Assay |
The same cell-based cAMP HTRF assay was used for all receptor activity and selectivity profiling. Cells were thawed, resuspended in appropriate medium, seeded into assay plates, and preincubated with test compounds before agonist addition. After incubation, cells were lysed, and cAMP levels were detected via HTRF readout. Data were analyzed using commercial or in-house software for IC₅₀ calculation. [1] Caco-2 permeability assays were conducted to evaluate absorption potential. Cells were grown on inserts for 15 days, and test compounds were added to either apical or basolateral compartments. Permeability (Papp) and efflux ratios were calculated after 2-hour incubation. [1] |
| Animal Protocol |
Animal/Disease Models: Male adult Sprague Dawley rats (220-265 g) were injected with 16,16-dimethyl prostaglandin E2 (dmPGE2) [1] Doses: 0.2 mg/kg, 1 mg/kg, 5 mg/kg Route of Administration: oral administration; injection administration; injection administration. Experimental Results: Paw withdrawal threshold was Dramatically diminished in the dmPGE2 pain model. For pharmacokinetic studies in rats, BAY-1316957 was administered intravenously (0.3–1 mg/kg) or orally (0.5–10 mg/kg) formulated in PEG400-containing solutions. Blood samples were collected at multiple time points, plasma was separated, and compound concentration was determined by LC-MS/MS. PK parameters were calculated using non-compartmental analysis. [1] For the dmPGE2-induced pain model, male Sprague-Dawley rats were orally administered BAY-1316957 (0.2, 1.0, or 5.0 mg/kg) suspended in 0.5% carboxymethylcellulose (CMC) solution, 60 minutes before intraplantar injection of dmPGE2 (10 µg) into the hind paw. Mechanical allodynia was assessed using an electronic von Frey apparatus, measuring paw withdrawal thresholds before and after treatment. [1] |
| ADME/Pharmacokinetics |
BAY-1316957 exhibits low blood clearance (CLblood = 0.43 L/h/kg in rats), long half-life (t₁/₂ = 24 h in rats after oral dosing), high oral bioavailability (F = 90% in rats), and low to moderate plasma protein binding (free fraction not specified but inferred from stability studies). [1] The main elimination pathway is direct glucuronidation via UGT1A1/1A3, forming an acyl glucuronide metabolite, which is excreted in bile and partly cleaved back to parent compound in feces. [1] Caco-2 permeability is high (Papp A-B = 205.9 nm/s) with low efflux ratio (0.6), indicating good absorption potential. [1] |
| References |
[1]. Identification of a Benzimidazolecarboxylic Acid Derivative (BAY 1316957) as a Potent and Selective Human Prostaglandin E2 Receptor Subtype 4 (hEP4-R) Antagonist for the Treatment of Endometriosis. J Med Chem. 2019 Mar 14;62(5):2541-2563. |
| Additional Infomation |
BAY-1316957 is a benzimidazolecarboxylic acid derivative developed as a potent and selective EP4 receptor antagonist for the treatment of endometriosis-associated inflammatory pain. [1] Its design incorporated a methyl group adjacent to the carboxylic acid to reduce glucuronidation and improve metabolic stability. [1] The compound is intended for once-daily oral administration in humans due to its favorable pharmacokinetic profile. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~226.49 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 5 mg/mL (11.32 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 50.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.71 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.71 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2649 mL | 11.3245 mL | 22.6490 mL | |
| 5 mM | 0.4530 mL | 2.2649 mL | 4.5298 mL | |
| 10 mM | 0.2265 mL | 1.1325 mL | 2.2649 mL |