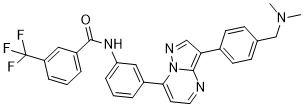
B-Raf IN 1 is a novel, potent and selective B-Raf inhibitor with IC50 of 24 nM; it is equally potent against c-Raf with IC50 of 25 nM. Moderate selectivity was observed for compound 10n versus p38a (IC50: 0.216 lM) and CAMKII (IC50: 0.822 lM), while high selectivity was observed versus CDK2, CDK4, PKCa, IKKb, JNK1, MK2, PKA, Src, MKK6, PLK1, p70S6K, PI3 Ka, and PDK1 (IC50s: >2 lM). It binds to B-Raf kinase without forming a hinge-binding hydrogen bond. With basic amine residues appended to C-3 aryl residues, cellular activity and solubility were enhanced over previously described compounds of this class.
Physicochemical Properties
| Molecular Formula | C29H24F3N5O | |
| Molecular Weight | 515.53 | |
| Exact Mass | 515.193 | |
| CAS # | 950736-05-7 | |
| Related CAS # |
|
|
| PubChem CID | 24884503 | |
| Appearance | Light yellow to yellow solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Index of Refraction | 1.621 | |
| LogP | 4.39 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 7 | |
| Rotatable Bond Count | 6 | |
| Heavy Atom Count | 38 | |
| Complexity | 787 | |
| Defined Atom Stereocenter Count | 0 | |
| InChi Key | AIWJVLQNYNCDSL-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C29H24F3N5O/c1-36(2)18-19-9-11-20(12-10-19)25-17-34-37-26(13-14-33-27(25)37)21-5-4-8-24(16-21)35-28(38)22-6-3-7-23(15-22)29(30,31)32/h3-17H,18H2,1-2H3,(H,35,38) | |
| Chemical Name | N-[3-[3-[4-[(dimethylamino)methyl]phenyl]pyrazolo[1,5-a]pyrimidin-7-yl]phenyl]-3-(trifluoromethyl)benzamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
B-Raf IN 1 (compound 10n) targets Proto-Oncogene Proteins B-raf (B-Raf kinase) [1] |
||
| ln Vitro |
WM 266-4 and HT29 cells are inhibited by B-Raf IN 1 (Compound 10n) at IC50s of 0.92 and 0.78 μM, respectively[1]. 1. B-Raf IN 1 (compound 10n), a C-3 substituted N-(3-(pyrazolo[1,5-a]pyrimidin-7-yl)phenyl)-3-(trifluoromethyl)benzamide, was identified as a potent B-Raf kinase inhibitor [1] 2. X-ray crystallography analysis demonstrated that B-Raf IN 1 bound to B-Raf kinase without forming a hinge-binding hydrogen bond, which is a distinct binding mode compared with conventional B-Raf kinase inhibitors [1] 3. The introduction of basic amine residues to the C-3 aryl residues of pyrazolo[1,5-a]pyrimidines (the structural class of B-Raf IN 1) enhanced cellular activity and solubility relative to previously reported compounds of the same class [1] |
||
| ln Vivo |
|
||
| Enzyme Assay |
1. X-ray crystallography assay for B-Raf kinase binding mode: The binding interaction between B-Raf IN 1 and B-Raf kinase was characterized using X-ray crystallography. The experiment was designed to determine the structural basis of the inhibitor's binding to the kinase, focusing on whether hydrogen bonds were formed at the hinge region of B-Raf kinase. The results confirmed that B-Raf IN 1 bound to B-Raf kinase without forming a hinge-binding hydrogen bond [1] 2. Structure-activity relationship (SAR) assay for pyrazolo[1,5-a]pyrimidines: A series of C-3 substituted N-(3-(pyrazolo[1,5-a]pyrimidin-7-yl)phenyl)-3-(trifluoromethyl)benzamides (including B-Raf IN 1) were synthesized and evaluated for their B-Raf kinase inhibitory activity. The impact of structural modifications (e.g., appending basic amine residues to C-3 aryl residues) on the potency, cellular activity and solubility of the compounds was analyzed [1] |
||
| Animal Protocol |
|
||
| References |
[1]. Non-hinge-binding pyrazolo[1,5-a]pyrimidines as potent B-Raf kinase inhibitors. Bioorg Med Chem Lett. 2009 Dec 1;19(23):6519-23. |
||
| Additional Infomation |
1. B-Raf IN 1 belongs to a series of C-3 substituted N-(3-(pyrazolo[1,5-a]pyrimidin-7-yl)phenyl)-3-(trifluoromethyl)benzamides, which are designed as B-Raf kinase inhibitors [1] 2. The non-hinge-binding mode of B-Raf IN 1 to B-Raf kinase is a key structural feature that distinguishes it from traditional B-Raf kinase inhibitors [1] 3. Structural optimization of pyrazolo[1,5-a]pyrimidines (including B-Raf IN 1) by adding basic amine residues to C-3 aryl residues addresses the limitations (e.g., low cellular activity, poor solubility) of earlier compounds in this class [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 3.25 mg/mL (6.30 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 32.5 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 + to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9398 mL | 9.6988 mL | 19.3975 mL | |
| 5 mM | 0.3880 mL | 1.9398 mL | 3.8795 mL | |
| 10 mM | 0.1940 mL | 0.9699 mL | 1.9398 mL |