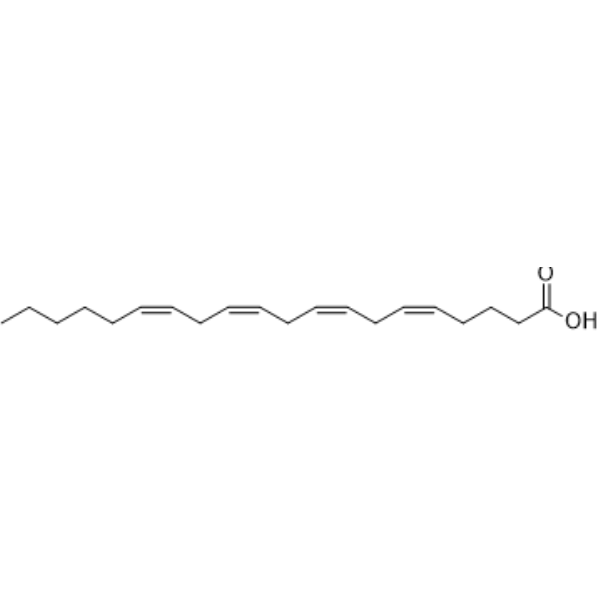
Arachidonic acid is a long-chain fatty acid that is a C20, polyunsaturated fatty acid having four (Z)-double bonds at positions 5, 8, 11 and 14. It has a role as a human metabolite, an EC 3.1.1.1 (carboxylesterase) inhibitor, a Daphnia galeata metabolite and a mouse metabolite. It is an icosa-5,8,11,14-tetraenoic acid, an omega-6 fatty acid and a long-chain fatty acid. It is a conjugate acid of an arachidonate. It derives from a hydride of a (5Z,8Z,11Z,14Z)-icosa-5,8,11,14-tetraene.
Arachidonic Acid is an unsaturated, essential fatty acid. It is found in animal and human fat as well as in the liver, brain, and glandular organs, and is a constituent of animal phosphatides. It is formed by the synthesis from dietary linoleic acid and is a precursor in the biosynthesis of prostaglandins, thromboxanes, and leukotrienes.(ChemID Plus) Arachidonic Acid is an unsaturated, essential fatty acid. It is found in animal and human fat as well as in the liver, brain, and glandular organs, and is a constituent of animal phosphatides. It is formed by the synthesis from dietary linoleic acid and is a precursor in the biosynthesis of prostaglandins, thromboxanes, and leukotrienes.
Physicochemical Properties
| Molecular Formula | C20H32O2 |
| Molecular Weight | 304.4669 |
| Exact Mass | 304.24 |
| Elemental Analysis | C, 78.90; H, 10.59; O, 10.51 |
| CAS # | 506-32-1 |
| Related CAS # | Arachidonic acid-d8;69254-37-1;Arachidonic acid-d5;123167-26-0;Arachidonic acid-d11;2692624-11-4 |
| PubChem CID | 444899 |
| Appearance | Colorless to light yellow liquid |
| Density | 0.9±0.1 g/cm3 |
| Boiling Point | 407.5±0.0 °C at 760 mmHg |
| Melting Point | −49 °C(lit.) |
| Flash Point | 336.3±18.0 °C |
| Vapour Pressure | 0.0±2.0 mmHg at 25°C |
| Index of Refraction | 1.501 |
| LogP | 6.91 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 14 |
| Heavy Atom Count | 22 |
| Complexity | 362 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O([H])C(C([H])([H])C([H])([H])C([H])([H])/C(/[H])=C(/[H])\C([H])([H])/C(/[H])=C(/[H])\C([H])([H])/C(/[H])=C(/[H])\C([H])([H])/C(/[H])=C(/[H])\C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H])=O |
| InChi Key | YZXBAPSDXZZRGB-DOFZRALJSA-N |
| InChi Code | InChI=1S/C20H32O2/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-20(21)22/h6-7,9-10,12-13,15-16H,2-5,8,11,14,17-19H2,1H3,(H,21,22)/b7-6-,10-9-,13-12-,16-15- |
| Chemical Name | (5Z,8Z,11Z,14Z)-icosa-5,8,11,14-tetraenoic acid |
| Synonyms | arachidonic acid; 506-32-1; (5Z,8Z,11Z,14Z)-icosa-5,8,11,14-tetraenoic acid; (all-Z)-5,8,11,14-Eicosatetraenoic acid; cis-5,8,11,14-Eicosatetraenoic acid; 5,8,11,14-Eicosatetraenoic acid, (all-Z)-; all-cis-5,8,11,14-eicosatetraenoic acid; Arachidonsaeure; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). This product requires protection from light (avoid light exposure) during transportation and storage.(2). Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Endogenous Metabolite; Cyclooxygenase (COX); Lipoxygenase (LOX)[1] |
| ln Vivo |
In a rat adjuvant-induced arthritis (AIA) model, dietary supplementation with arachidonic acid (1% w/w in feed for 7 weeks) significantly increased arachidonic acid content in paw tissues by 2.3-fold compared to control. However, it did not alter arthritis severity (measured by paw swelling and clinical scores) or paw prostaglandin E₂ (PGE₂) content, indicating no pro-inflammatory exacerbation. Serum analysis confirmed elevated arachidonic acid levels in supplemented rats but showed no correlation with disease progression or systemic PGE₂ changes.[1] In animal modeling, arachidonic acid can be utilized to create an obese foot model. |
| Animal Protocol | AIA Model: Female Lewis rats received 1% (w/w) arachidonic acid in standard chow for 7 weeks (starting 1 week pre-adjuvant injection). Arthritis was induced at day 0 by intradermal tail injection of heat-killed Mycobacterium tuberculosis in mineral oil. Paw swelling was measured weekly; tissues/serum were collected at endpoint for lipidomics and PGE₂ analysis.[1] |
| ADME/Pharmacokinetics |
Metabolism / Metabolites Arachidonic acid has known human metabolites that include 17-HETE, 18-HETE, 8,9-EET, 20-HETE, 9-HETE, 8-HETE, 5-HETE, 11-HETE, 11,12-EET, 5,6-EET, 13-HETE, 7-HETE, 10-HETE, 12-HETE, 14,15-EET, 15-HETE, 16-HETE, and 19-HETE. |
| Toxicity/Toxicokinetics |
Toxicity Summary
Cosmetic Ingredient Review Conclusion The safety of this ingredient has not been documented and substantiated for cosmetic product use. The CIR Expert Panel cannot conclude whether Arachidonic Acid is safe for use in cosmetic products until such time that the appropriate safety data have been obtained and evaluated. Ingredients for which the data are insufficient and their use in cosmetics is not supported |
| References |
[1]. Dietary supplementation with arachidonic acid increases arachidonic acid content in paw, but does not affect arthritis severity or prostaglandin E2 content in rat adjuvant-induced arthritis model. Lipids Health Dis. 2015 Jan 16;14:3. |
| Additional Infomation |
Arachidonic acid is an ω-6 polyunsaturated fatty acid serving as a precursor for inflammatory eicosanoids like PGE₂. This study demonstrates that increased tissue arachidonic acid availability does not necessarily translate to amplified inflammation in arthritis. The dissociation between tissue arachidonic acid content and PGE₂ levels suggests complex regulatory mechanisms in eicosanoid biosynthesis during chronic inflammation.[1] Arachidonic acid is a long-chain fatty acid that is a C20, polyunsaturated fatty acid having four (Z)-double bonds at positions 5, 8, 11 and 14. It has a role as a human metabolite, an EC 3.1.1.1 (carboxylesterase) inhibitor, a Daphnia galeata metabolite and a mouse metabolite. It is an icosa-5,8,11,14-tetraenoic acid, an omega-6 fatty acid and a long-chain fatty acid. It is a conjugate acid of an arachidonate. It derives from a hydride of a (5Z,8Z,11Z,14Z)-icosa-5,8,11,14-tetraene. Arachidonic acid has been reported in Agaricus blazei, Mortierella hygrophila, and other organisms with data available. Arachidonic Acid is an unsaturated, essential fatty acid. It is found in animal and human fat as well as in the liver, brain, and glandular organs, and is a constituent of animal phosphatides. It is formed by the synthesis from dietary linoleic acid and is a precursor in the biosynthesis of prostaglandins, thromboxanes, and leukotrienes.(ChemID Plus) An unsaturated, essential fatty acid. It is found in animal and human fat as well as in the liver, brain, and glandular organs, and is a constituent of animal phosphatides. It is formed by the synthesis from dietary linoleic acid and is a precursor in the biosynthesis of prostaglandins, thromboxanes, and leukotrienes. |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~100 mg/mL (~328.44 mM) Ethanol : ~100 mg/mL (~328.44 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.21 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (8.21 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 3: ≥ 2.08 mg/mL (6.83 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 4: 10 mg/mL (32.84 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication (<60°C). (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2844 mL | 16.4220 mL | 32.8440 mL | |
| 5 mM | 0.6569 mL | 3.2844 mL | 6.5688 mL | |
| 10 mM | 0.3284 mL | 1.6422 mL | 3.2844 mL |