Physicochemical Properties
| Molecular Formula | C69H111N23O16S |
| Molecular Weight | 1550.82914 |
| Exact Mass | 1549.829 |
| CAS # | 217082-58-1 |
| Related CAS # | Apelin-13 TFA |
| PubChem CID | 25078060 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.5±0.1 g/cm3 |
| Index of Refraction | 1.688 |
| LogP | -3.42 |
| Hydrogen Bond Donor Count | 19 |
| Hydrogen Bond Acceptor Count | 22 |
| Rotatable Bond Count | 47 |
| Heavy Atom Count | 109 |
| Complexity | 3140 |
| Defined Atom Stereocenter Count | 12 |
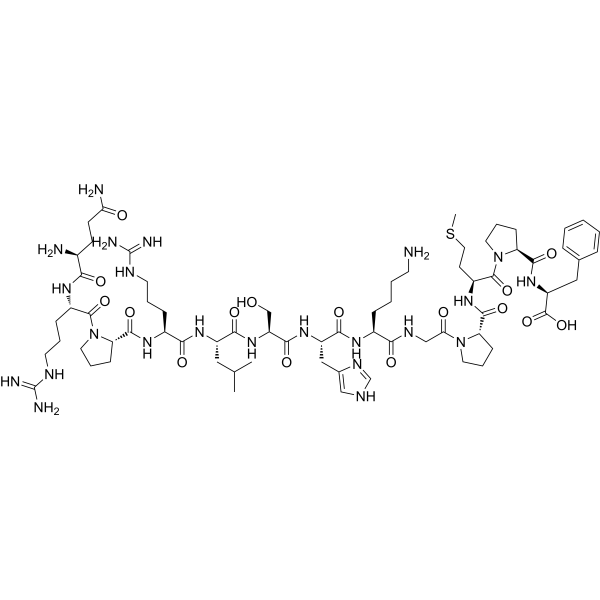
| SMILES | CC(C[C@H](NC([C@@H](NC([C@@H]1CCCN1C([C@@H](NC([C@@H](N)CCC(N)=O)=O)CCCNC(N)=N)=O)=O)CCCNC(N)=N)=O)C(N[C@H](C(N[C@H](C(N[C@H](C(NCC(N2CCC[C@H]2C(N[C@H](C(N3CCC[C@H]3C(N[C@H](C(O)=O)CC4=CC=CC=C4)=O)=O)CCSC)=O)=O)=O)CCCCN)=O)CC5=CN=CN5)=O)CO)=O)C |
| InChi Key | XXCCRHIAIBQDPX-PEWBXTNBSA-N |
| InChi Code | InChI=1S/C69H111N23O16S/c1-39(2)32-47(86-58(98)44(17-9-26-78-68(73)74)83-63(103)52-20-12-29-91(52)65(105)45(18-10-27-79-69(75)76)84-56(96)42(71)22-23-54(72)94)59(99)89-50(37-93)61(101)87-48(34-41-35-77-38-81-41)60(100)82-43(16-7-8-25-70)57(97)80-36-55(95)90-28-11-19-51(90)62(102)85-46(24-31-109-3)66(106)92-30-13-21-53(92)64(104)88-49(67(107)108)33-40-14-5-4-6-15-40/h4-6,14-15,35,38-39,42-53,93H,7-13,16-34,36-37,70-71H2,1-3H3,(H2,72,94)(H,77,81)(H,80,97)(H,82,100)(H,83,103)(H,84,96)(H,85,102)(H,86,98)(H,87,101)(H,88,104)(H,89,99)(H,107,108)(H4,73,74,78)(H4,75,76,79)/t42-,43-,44-,45-,46-,47-,48-,49-,50-,51-,52-,53-/m0/s1 |
| Chemical Name | (2S)-2-[[(2S)-1-[(2S)-2-[[(2S)-1-[2-[[(2S)-6-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-5-(diaminomethylideneamino)-2-[[(2S)-1-[(2S)-5-(diaminomethylideneamino)-2-[[(2S)-2,5-diamino-5-oxopentanoyl]amino]pentanoyl]pyrrolidine-2-carbonyl]amino]pentanoyl]amino]-4-methylpentanoyl]amino]-3-hydroxypropanoyl]amino]-3-(1H-imidazol-5-yl)propanoyl]amino]hexanoyl]amino]acetyl]pyrrolidine-2-carbonyl]amino]-4-methylsulfanylbutanoyl]pyrrolidine-2-carbonyl]amino]-3-phenylpropanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Apelin-13 targets apelin receptor (APJ) [1] |
| ln Vitro |
Apelin-13 (1 nM–100 nM) dose-dependently bound to human APJ receptors expressed in CHO cells and activated G protein-coupled signaling: 100 nM increased intracellular cAMP accumulation by 3.2-fold and induced calcium mobilization, confirming receptor agonism [1] In human hepatoma HepG2 cells, Apelin-13 (10 nM–50 nM) enhanced insulin-induced glucose uptake: 50 nM increased glucose uptake by 45% compared to insulin alone; it also reduced palmitate-induced lipid accumulation by 38% (50 nM) after 24 hours [2] |
| ln Vivo |
Apelin-13 (200 µg/kg; IP, once daily for 4 weeks) increases the levels of GLUT4 and p-AMPKα2, decreases serum TNF-α and leptin, enhances lipid metabolism, and improves cardiac function [2]. It also dramatically increases serum Apelin-12 expression. Most globus pallidus neurons exhibit increased spontaneous firing in response to Apelin-13 (10 and 100 μM; ICV, single dose) [4]. In high-fat diet-induced type 2 diabetic SD rats, intraperitoneal administration of Apelin-13 (20 μg/kg, q.d.) for 4 weeks improved metabolic and cardiovascular parameters: fasting blood glucose decreased from 18.6 mmol/L to 11.2 mmol/L, HOMA-IR (insulin resistance index) reduced by 52%, serum triglycerides decreased by 41%, and total cholesterol reduced by 35% [2] The same treatment in diabetic rats improved cardiac function: left ventricular end-diastolic diameter reduced by 18%, and systolic blood pressure decreased by 15 mmHg compared to the vehicle group [2] In adult SD rats, intracranial injection of Apelin-13 (0.5 μL, 1 μg/μL) into the globus pallidus regulated neuronal electrical activity: reduced the firing rate of pallidal neurons by 42% and increased hindlimb extension duration by 3.5-fold within 30 minutes, inducing significant postural changes [4] |
| Enzyme Assay |
APJ receptor binding assay: Membrane preparations from CHO cells transfected with human APJ were incubated with [¹²⁵I]-labeled Apelin-13 and unlabeled Apelin-13 (0.1 nM–1000 nM) in binding buffer at 25°C for 60 minutes. Bound ligands were separated from free ligands by filtration, and radioactivity of the bound fraction was measured. Competitive binding curves were generated to evaluate the binding affinity of Apelin-13 for APJ [1] cAMP accumulation assay: CHO-APJ cells were seeded in 96-well plates and treated with Apelin-13 (1 nM–100 nM) for 30 minutes. Intracellular cAMP was extracted using a lysis buffer, and cAMP concentration was quantified by a competitive binding assay with a cAMP-specific antibody [1] |
| Cell Assay |
Calcium mobilization assay: CHO-APJ cells were loaded with a calcium-sensitive fluorescent probe and incubated at 37°C for 30 minutes. Apelin-13 (1 nM–100 nM) was added to the cells, and real-time fluorescence intensity was monitored to detect intracellular calcium mobilization [1] Glucose uptake assay: HepG2 cells were seeded in 24-well plates and serum-starved for 12 hours. Cells were treated with Apelin-13 (10 nM–50 nM) and insulin (100 nM) for 1 hour, then incubated with [³H]-2-deoxyglucose for 30 minutes. Cells were washed to remove unincorporated radioactivity, and the radioactivity of cell lysates was measured to quantify glucose uptake [2] Lipid accumulation inhibition assay: HepG2 cells were treated with palmitate (200 μM) and Apelin-13 (10 nM–50 nM) for 24 hours. Intracellular lipids were stained with Oil Red O, and the stained lipids were eluted with isopropanol. Absorbance at 510 nm was measured to quantify lipid content [2] |
| Animal Protocol |
Animal/Disease Models: Goto-Kakizaki (GK) rat (12 weeks old; 240-280 g; fed high-fat diet: 66.5% standard chow, 10% lard, 20% sucrose, 2.5% cholesterol, and 1% porcine bile salts) )[2] Doses: 200 µg/kg Route of Administration: intraperitoneal (ip) injection, one time/day for 4 weeks Experimental Results: Significant reduction in heart rate; reduction in fasting plasma glucose (FPG), fasting insulin (FINS) and insulin resistance Homeostasis Model Assessment (HOMA-IR) levels; reduce serum total cholesterol (TC), triglyceride (TG) and low-density lipoprotein cholesterol (LDL-C) levels, and increase high-density lipoprotein cholesterol (HDL-C); in serum NO levels, cNOS activity, TNF-α and leptin were diminished; Apelin-12 expression was induced. Animal/Disease Models: Adult Wistar rat (SPF, 8-10 weeks, 240-280 g) [4] Doses: 10 and 100 μM Route of Administration: ICV, single dose Experimental Results: Increased spontaneous firing of most globus pallidus neurons. Type 2 diabetes rat model: Male SD rats were fed a high-fat diet for 12 weeks to induce type 2 diabetes. Rats were randomized into vehicle and Apelin-13 treatment groups (20 μg/kg, i.p., q.d., n=10/group). Apelin-13 was dissolved in normal saline. After 4 weeks of treatment, fasting blood glucose, insulin, and lipid profiles (triglycerides, total cholesterol) were measured; echocardiography was performed to assess cardiac structure and function [2] Globus pallidus electrophysiology rat model: Adult male SD rats were anesthetized and stereotaxically implanted with microelectrodes and injection cannulas targeting the globus pallidus. After a 7-day recovery period, Apelin-13 (1 μg/μL, 0.5 μL) was injected intracranially. Extracellular electrical activity of pallidal neurons was recorded for 60 minutes, and postural changes (hindlimb extension) were observed and quantified [4] |
| Toxicity/Toxicokinetics |
In diabetic rats treated with Apelin-13 (20 μg/kg, i.p., q.d.) for 4 weeks: no significant changes in body weight, hematological parameters (WBC, RBC, platelets), or biochemical parameters (ALT, AST, BUN, creatinine) were observed. Histopathological examination of liver, kidney, and heart showed no drug-related lesions [2] Intracranial injection of Apelin-13 (0.5 μL, 1 μg/μL) in rats did not induce toxic symptoms such as seizures, abnormal behavior, or tissue inflammation at the injection site [4] |
| References |
[1]. Isolation and characterizaton of a novel endogenous peptide ligand for the human APJ receptor. Biochemical and Biophysical Research Communications 251, 471-476 (1998). [2]. Apelin 13 ameliorates metabolic and cardiovascular disorders in a rat model of type 2 diabetes with a high fat diet. Mol Med Rep. 2018 Dec;18(6):5784-5790. [3]. Apelin-13 in blood pressure regulation and cardiovascular disease. Curr Opin Nephrol Hypertens. 2016 Sep;25(5):396-403. [4]. Apelin-13 regulates electrical activity in the globus pallidus and induces postural changes in rats. Neural Regen Res. 2021 Nov;16(11):2264-2268. |
| Additional Infomation |
Apelin-13 is a 13 amino acid oligopeptide which is the ligand for the apelin receptor (also known as the APJ receptor). It exhibits hypotensive and neuroprotective effects, and may be a potential prognostic biomarker for acute ischemic stroke and multiple sclerosis. It has a role as an antihypertensive agent, a biomarker, an autophagy inhibitor, a neuroprotective agent and a human metabolite. It is a conjugate base of an apelin-13(3+). Apelin-13 is an endogenous peptide ligand first isolated from bovine stomach tissue, identified as a specific ligand for the human APJ receptor [1] Its biological effects are mediated by binding to APJ, activating downstream G protein-coupled signaling pathways (cAMP accumulation, calcium mobilization) to regulate metabolic, cardiovascular, and neural functions [1,2,4] Apelin-13 exhibits potential therapeutic value for type 2 diabetes mellitus and metabolic syndrome by improving insulin sensitivity and regulating lipid metabolism [2] It protects cardiovascular function in diabetic rats, suggesting applicability in treating diabetes-related cardiovascular disorders [2] In the central nervous system, Apelin-13 modulates globus pallidus electrical activity and postural control, indicating potential roles in neurological disorders involving motor dysfunction [4] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.6448 mL | 3.2241 mL | 6.4482 mL | |
| 5 mM | 0.1290 mL | 0.6448 mL | 1.2896 mL | |
| 10 mM | 0.0645 mL | 0.3224 mL | 0.6448 mL |