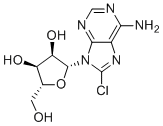
Physicochemical Properties
| Molecular Formula | C10H12CLN5O4 |
| Molecular Weight | 301.687 |
| Exact Mass | 301.057 |
| CAS # | 34408-14-5 |
| PubChem CID | 147569 |
| Appearance | White to off-white solid powder |
| Density | 2.2±0.1 g/cm3 |
| Boiling Point | 707.4±70.0 °C at 760 mmHg |
| Flash Point | 381.6±35.7 °C |
| Vapour Pressure | 0.0±2.4 mmHg at 25°C |
| Index of Refraction | 1.912 |
| LogP | -0.46 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 20 |
| Complexity | 367 |
| Defined Atom Stereocenter Count | 4 |
| SMILES | C1=NC(=C2C(=N1)N(C(=N2)Cl)[C@H]3[C@@H]([C@@H]([C@H](O3)CO)O)O)N |
| InChi Key | MHDPPLULTMGBSI-UUOKFMHZSA-N |
| InChi Code | InChI=1S/C10H12ClN5O4/c11-10-15-4-7(12)13-2-14-8(4)16(10)9-6(19)5(18)3(1-17)20-9/h2-3,5-6,9,17-19H,1H2,(H2,12,13,14)/t3-,5-,6-,9-/m1/s1 |
| Chemical Name | (2R,3R,4S,5R)-2-(6-amino-8-chloropurin-9-yl)-5-(hydroxymethyl)oxolane-3,4-diol |
| Synonyms | 8-Chloroadenosine 8Chloroadenosine 8 Chloroadenosine NSC 354258 NSC354258 NSC-354258 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
8-Chloroadenosine (8-Cl-Ado) is metabolized intracellularly to 8-Cl-ATP, which can inhibit transcription and poly(A) polymerase [1]. 8-Cl-ADP and 8-Cl-ATP are inhibitors of mitochondrial complex V (ATP synthase) [1]. The drug also activates AMP-activated protein kinase (AMPK), inhibits the mammalian target of rapamycin (mTOR) pathway, and induces autophagy via phosphorylation of Unc51-like kinase 1 (ULK1) [1]. |
| ln Vitro |
At the LC3B-II and p62 levels, 8-Chloroadenosine (8-Cl-Ado; 10 μM; 24-72 hours) stimulates autophagic flux [1]. In MCF-7 and BT-474 cells, 8-chloroadenosine (10 μM; 7–21 hours) stimulates AMPK (Thr172) phosphorylation in a time-dependent manner. The levels of total AMPK protein were constant. In MCF-7 cells, 8-chloroadenosine quickly causes raptor Ser792 to become phosphorylated. In MCF-7 and BT-474 cells, 8-chloroadenosine (10 μM; 4-72 hours) phosphorylates ULK1 at Ser555 [1]. 8-MTOR activity is reduced in breast cancer cells by chloroadenosine. P53 is not necessary for 8-chloroadenosine to function[1]. In the MCF-7 breast cancer cell line, 8-chloroadenosine (10 μM; 3 days) decreases clonal survival by over 90%. Merely approximately 30% of apoptosis is induced [1]. In T47D, SK-BR-3, and ZR-75-1 cells, treatment with 8-chloroadenosine (10 μM) results in fast ATP depletion within 12 hours [1]. A 3-day treatment with 10 µM 8-Cl-Ado caused a 90% loss of clonogenic survival in MCF-7 and BT-474 breast cancer cells [1]. Treatment with 10 µM 8-Cl-Ado for up to 3 days induced only ~30% apoptosis (annexin V/PI staining) in MCF-7 and BT-474 cells, suggesting a non-apoptotic cell death mechanism [1]. Treatment with 10 µM 8-Cl-Ado led to rapid depletion of endogenous ATP within 12 hours and accumulation of 8-Cl-ATP [1]. In MCF-7 cells, 8-Cl-ATP levels peaked at 24 hours, while in BT-474 cells, they peaked at 72 hours [1]. The ratio of 8-Cl-ATP to ATP was highest in MCF-7 cells at 12 hours and continually increased in BT-474 cells until 24 hours [1]. Treatment with 10 µM 8-Cl-Ado for 18 hours perturbed basal mitochondrial respiration (oxygen consumption rate, OCR) and glycolysis (extracellular acidification rate, ECAR) in MCF-7 and BT-474 cells [1]. Treatment with 10 µM 8-Cl-Ado induced phosphorylation of AMPK (Thr172) within 7-12 hours and phosphorylation of its downstream target acetyl-CoA carboxylase (ACC, Ser79) within 4 hours [1]. 8-Cl-Ado treatment (10 µM) induced phosphorylation of raptor (Ser792), attenuated mTOR autophosphorylation (Ser2481), and reduced phosphorylation of 4E-BP1 (Ser65) [1]. 8-Cl-Ado treatment (10 µM) induced phosphorylation of ULK1 (Ser555), lipidation of LC3B-I to LC3B-II, LC3B aggregation, autophagic flux (p62 degradation), and formation of acidic vesicular organelles (stained with monodansylcadaverine and acridine orange) in MCF-7 and BT-474 cells [1]. Knockdown of autophagy proteins ATG7 or beclin 1 by siRNA partially protected MCF-7 cells from 8-Cl-Ado-induced loss of clonogenic survival but did not alter apoptosis levels [1]. |
| ln Vivo |
8-Chloroadenosine (25–100 mg/kg; IP; thrice weekly for 3 weeks) at 100 mg/kg suppresses the growth of BT-474 and MCF-7 xenograft tumors. 8-Chloroadenosine did not inhibit the growth of BT-474 xenograft tumors when administered at a dose of 50 mg/kg [1]. Intraperitoneal injection of 8-Cl-Ado (100 mg/kg, 3 days/week for 3 weeks) significantly suppressed the growth of orthotopic MCF-7 and BT-474 breast cancer xenografts in nude mice [1]. In BT-474 xenografts, significant growth inhibition was observed by the third day of treatment, and 9 out of 22 tumors treated with 100 mg/kg/day showed no macroscopically detectable tumor after 3 weeks [1]. Final excised tumor volumes were significantly smaller in the 8-Cl-Ado-treated groups compared to PBS-treated controls [1]. |
| Cell Assay |
Clonogenic assay: Breast cancer cells (MCF-7, BT-474) were treated with 8-Cl-Ado (e.g., 10 µM) for 3 days, washed with PBS, and cultured in fresh medium for 10-14 days. Colonies of >50 cells were counted under a dissecting microscope [1]. Flow cytometry for apoptosis: Cells were treated with 8-Cl-Ado, then stained with annexin V and propidium iodide (PI) and analyzed by flow cytometry [1]. Flow cytometry for autophagy (acidic vesicular organelles): Cells were treated with 8-Cl-Ado, stained with acridine orange (1 µg/mL) for 15 minutes at 37°C, and analyzed by flow cytometry. Bafilomycin A1 (0.1 µg/mL) was added 30 minutes before staining as a negative control; rapamycin (50 nM) served as a positive control [1]. Western blot analysis: Cells treated with 8-Cl-Ado were lysed, and proteins were separated by SDS-PAGE, transferred to membranes, and probed with specific antibodies (e.g., p-AMPK, AMPK, p-ACC, LC3B, p62, beclin 1, ATG7, p-ULK1, p-raptor, p-4E-BP1). GAPDH was used as a loading control. Detection was performed using an infrared imaging system [1]. Microscopy for autophagy: MCF-7 cells stably expressing GFP-LC3 were treated with 8-Cl-Ado and observed by fluorescence microscopy for LC3B aggregation. For autolysosome visualization, cells were stained with monodansylcadaverine (MDC, 50 µM) and SYTO 61 (5 µM) and imaged by fluorescence microscopy [1]. Metabolic analysis (glycolysis and mitochondrial respiration): Cells were seeded in specialized microplates and treated with 8-Cl-Ado for 18 hours. The extracellular acidification rate (ECAR, glycolysis) and oxygen consumption rate (OCR, mitochondrial respiration) were measured using an extracellular flux analyzer. For mitochondrial stress tests, oligomycin (1.25 µM), FCCP (1 µM), antimycin (0.75 µM), and rotenone (1.25 µM) were sequentially injected [1]. Nucleotide extraction and HPLC analysis: Cells treated with 8-Cl-Ado were extracted with perchloric acid. Neutralized extracts were analyzed by HPLC to quantify ATP, 8-Cl-ATP, and other nucleotides [1]. siRNA transfection: MCF-7 cells were transfected with siRNA targeting ATG7 or beclin 1 (or control siRNA) using a transfection reagent. Knockdown was allowed for ~2 days before treatment with 8-Cl-Ado [1]. |
| Animal Protocol |
Breast cancer xenograft efficacy study: Orthotopic tumors were established in female nude mice by inoculating MCF-7 (5×10⁶ cells) or BT-474 (1×10⁷ cells) into mammary fat pads. Mice were supplemented with estrogen pellets. When tumors reached ~3 mm in diameter, mice were randomized into groups (8-10 mice/group). 8-Cl-Ado was dissolved in PBS and administered intraperitoneally at a dose of 100 mg/kg, three times per week for 3 weeks. Control mice received PBS. Tumor diameters were measured three times per week. Mice were sacrificed 1-3 days after the final treatment, and tumors were excised and measured for volume calculation [1]. |
| ADME/Pharmacokinetics |
In a separate study cited within this work (reference [20]), intravenous administration of 50 and 100 mg/kg 8-Cl-Ado to mice resulted in 1-hour accumulation of 8-Cl-ATP in peripheral blood mononuclear cells of ~350 µM and ~1150 µM, respectively [1]. The elimination kinetics of 8-Cl-ATP in vitro after drug removal showed a biphasic pattern in MCF-7 cells with half-lives of 3.8 and 25.5 hours, and in BT-474 cells with half-lives of 6.4 hours and >7 days. Monophasic elimination half-lives were 5.8 hours (MCF-7) and 11.4 hours (BT-474) [1]. |
| Toxicity/Toxicokinetics |
An extensive toxicology assessment in CD1 mice (cited from reference [36]) showed no toxicity at doses used in efficacy studies (up to 100 mg/kg) based on hematology, clinical chemistry, and microscopic pathology parameters [1]. |
| References |
[1]. ATP directed agent, 8-chloro-adenosine, induces AMP activated protein kinase activity, leading to autophagic cell death in breast cancer cells. J Hematol Oncol. 2014 Mar 14;7:23. |
| Additional Infomation |
8-Chloroadenosine is under investigation in clinical trial NCT02509546 (8-chloroadenosine in Treating Patients With Relapsed or Refractory Acute Myeloid Leukemia). 8-Chloroadenosine is an antimetabolite and a chlorine derivative of the purine nucleoside adenosine, with potential antineoplastic activity. Upon administration, 8-chloro-adenosine is phosphorylated to form 8-chloro-adenosine triphosphate (8-chloro-ATP), which functions as a ribonucleoside analogue and competes with ATP during transcription. Therefore, this agent causes RNA synthesis inhibition, inhibits cellular proliferation, and induces apoptosis. 8-Chloroadenosine (8-Cl-Ado) is a ribonucleoside analog currently in a Phase I clinical trial for hematological malignancies (e.g., chronic lymphocytic leukemia) at the time of this publication [1]. It acts as a prodrug; it is metabolized by adenosine kinase to 8-Cl-AMP and further to 8-Cl-ATP intracellularly [1]. Its cytotoxicity in breast cancer cells involves two mechanisms: 1) transcription inhibition and apoptosis induction via 8-Cl-ATP accumulation, and 2) ATP depletion leading to AMPK activation, mTOR inhibition, and autophagic cell death [1]. Preliminary analysis of cells from patients in the clinical trial indicated ATP depletion, AMPK activation, and autophagy induction during treatment [1]. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~125 mg/mL (~414.33 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.3147 mL | 16.5733 mL | 33.1466 mL | |
| 5 mM | 0.6629 mL | 3.3147 mL | 6.6293 mL | |
| 10 mM | 0.3315 mL | 1.6573 mL | 3.3147 mL |