Physicochemical Properties
| Molecular Formula | C10H14CL3N5O2 |
| Molecular Weight | 342.609458446503 |
| Exact Mass | 341.02 |
| Elemental Analysis | C, 35.06; H, 4.12; Cl, 31.04; N, 20.44; O, 9.34 |
| CAS # | 1683617-62-0 |
| Related CAS # | 1683535-53-6 (HCL) |
| PubChem CID | 127243466 |
| Appearance | Off-white to yellow solid powder |
| LogP | 2.2 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 20 |
| Complexity | 297 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | JQTSJVDIFMKETH-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C10H14Cl3N5O2/c11-10(12,13)7(17-8-15-4-2-5-16-8)18-9(19)20-6-1-3-14/h2,4-5,7H,1,3,6,14H2,(H,18,19)(H,15,16,17) |
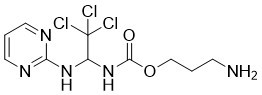
| Chemical Name | 3-aminopropyl N-[2,2,2-trichloro-1-(pyrimidin-2-ylamino)ethyl]carbamate |
| Synonyms | Apcin-A; Apcin A; 1683617-62-0; 3-Aminopropyl (2,2,2-trichloro-1-(pyrimidin-2-ylamino)ethyl)carbamate; starbld0000888; SCHEMBL22567019; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Anaphase-promoting complex (APC) |
| ln Vitro |
The structures of CP5V (apcin-A-PEG5-VHL1) and VHL-CP5V-Cdc20 complex were generated using as templates the Protein Data Bank (PDB) structures with ids 5T35 and 4N14 for the VHL1 and apcin-A fragments, respectively. [1] the Cdc20 small molecule inhibitors apcin (APC inhibitor) and apcin-A, which can competitively occupy the D-box binding pocket of Cdc20 to inhibit the ubiquitination of Cdc20 substrates, as well as small molecule inhibitors that antagonize the APC/C-Cdc20 interaction, TAME and proTAME. [1] |
| Enzyme Assay |
Apcin-A is utilized as Cdc20 targeting ligand, and VHL and CRBN binding moieties VHL1 and thalidomide are respectively used to recruit the VHL/VBC complex and Celebron E3 ligase in the Cdc20 PROTACs. A series of polyethylene glycol (PEG) molecules were used to link apcin-A and VHL1/thalidomide.[1] CP5V was designed based on apcin-A with a medium binding affinity. Lead optimization methods could be used to develop more potent Cdc20 inhibitors . Apcin and apcin-A have similar binding affinities14 and they do not interact directly with the polar residues around the D-box binding sites . The low binding affinity of apcin-P indicates that the aromatic group of the pyrimidine ring in apcin (and apcin-A), which interacts with Trp209, is important to enable stronger affinity14. [1] We decided to develop a novel chimera molecule, utilizing the PROTAC platform, to circumvent the challenges met by current Cdc20 small molecule inhibitors. Based on the feature that apcin-A can efficiently bind to Cdc20 and is easier to modify, we used apcin-A rather than apcin as the warhead to target Cdc20 . Regarding the E3 ligase recruited for Cdc20 ubiquitination followed by proteolysis, we considered VHL/VBC (VHL-Elongin BC) and CRBN (Celebron), both of which have reliable binding moieties (VHL ligand 1 or VHL1, and thalidomide, respectively) that have been applied in the design of several PROTAC molecules16,17,23. To search for an optimal chemical linker maximizing the formation of a stable Cdc20-PROTAC-VHL/VBC ternary complex, we have designed and tested a series of PEG-based linkers with different lengths, such as PEG2, PEG3, PEG4, PEG5, PEG6, PEG7, and PEG9. |
| References |
[1]. A novel strategy to block mitotic progression for targeted therapy. EBioMedicine. 2019 Oct 25. pii: S2352-3964(19)30677-2. |
| Additional Infomation | Background: Blockade of mitotic progression is an ideal approach to induce mitotic catastrophe that suppresses cancer cell expansion. Cdc20 is a critical mitotic factor governing anaphase initiation and the exit from mitosis through recruiting substrates to APC/C for degradation. Results from recent TCGA (The Cancer Genome Atlas) and pathological studies have demonstrated a pivotal oncogenic role for Cdc20-APC/C in tumor progression as well as drug resistance. Thus, deprivation of the mitotic role for Cdc20-APC/C by either inhibition of Cdc20-APC/C activity or elimination of Cdc20 protein via induced protein degradation emerges as an effective therapeutic strategy to control cancer. Methods: We designed a proteolysis targeting chimera, called CP5V, which comprises a Cdc20 ligand and VHL binding moiety bridged by a PEG5 linker that induces Cdc20 degradation. We characterized the effect of CP5V in destroying Cdc20, arresting mitosis, and inhibiting tumor progression by measuring protein degradation, 3D structure dynamics, cell cycle control, tumor cell killing and tumor inhibition using human breast cancer xenograft mouse model. Findings: Results from our study demonstrate that CP5V can specifically degrade Cdc20 by linking Cdc20 to the VHL/VBC complex for ubiquitination followed by proteasomal degradation. Induced degradation of Cdc20 by CP5V leads to significant inhibition of breast cancer cell proliferation and resensitization of Taxol-resistant cell lines. Results based on a human breast cancer xenograft mouse model show a significant role for CP5V in suppressing breast tumor progression. Interpretation: CP5V-mediated degradation of Cdc20 could be an effective therapeutic strategy for anti-mitotic therapy. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO: ~100 mg/mL (~291.9 mM |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.5 mg/mL (7.30 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (7.30 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (7.30 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9188 mL | 14.5939 mL | 29.1877 mL | |
| 5 mM | 0.5838 mL | 2.9188 mL | 5.8375 mL | |
| 10 mM | 0.2919 mL | 1.4594 mL | 2.9188 mL |