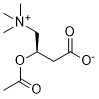
Acetyl-L-carnitine is a novel and potent stimulator of α-secretase activity and metabolism of amyloid precursor protein (APP). It has been shown to induce NF-κB-mediated upregulation of mGluR2 receptors, thereby exhibiting antidepressant, neuroprotective, analgesic, and antinociceptive activities.
Physicochemical Properties
| Molecular Weight | 203.24 |
| Exact Mass | 203.115 |
| CAS # | 3040-38-8 |
| PubChem CID | 7045767 |
| Appearance | White to off-white solid powder |
| Melting Point | 145°C |
| LogP | -4.15 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 14 |
| Complexity | 214 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | CC(O[C@@H](C[N+](C)(C)C)CC([O-])=O)=O |
| InChi Key | RDHQFKQIGNGIED-MRVPVSSYSA-N |
| InChi Code | InChI=1S/C9H17NO4/c1-7(11)14-8(5-9(12)13)6-10(2,3)4/h8H,5-6H2,1-4H3/t8-/m1/s1 |
| Chemical Name | (3R)-3-Acetyloxy-4-(trimethylazaniumyl)butanoate |
| Synonyms | Acetyl-L-carnitine Acetyl L carnitine ALCAR L-Acetylcarnitine L-O-Acetylcarnitine LAC |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Acetylcarnitine supplements are absorbed in a similar manner to L-carnitine. Acetylcarnitine is eliminated in a similar manner as L-carnitine. Both of which are eliminated by the kidneys and involve tubular secretion. In mammals, the carnitine pool consists of nonesterified L-carnitine and many acylcarnitine esters. Of these esters, acetyl-L-carnitine is quantitatively and functionally the most significant. Carnitine homeostasis is maintained by absorption from diet, a modest rate of synthesis, and efficient renal reabsorption. Dietary L-carnitine is absorbed by active and passive transfer across enterocyte membranes. Bioavailability of dietary L-carnitine is 54-87% and is dependent on the amount of L-carnitine in the meal. Absorption of L-carnitine dietary supplements (0.5-6 g) is primarily passive; bioavailability is 14-18% of dose. Unabsorbed L-carnitine is mostly degraded by microorganisms in the large intestine. Circulating L-carnitine is distributed to two kinetically defined compartments: one large and slow-turnover (presumably muscle), and another relatively small and rapid-turnover (presumably liver, kidney, and other tissues). At normal dietary L-carnitine intake, whole-body turnover time in humans is 38-119 h. In vitro experiments suggest that acetyl-L-carnitine is partially hydrolyzed in enterocytes during absorption. In vivo, circulating acetyl-L-carnitine concentration was increased 43% after oral acetyl-L-carnitine supplements of 2 g/day, indicating that acetyl-L-carnitine is absorbed at least partially without hydrolysis. After single-dose intravenous administration (0.5 g), acetyl-L-carnitine is rapidly, but not completely hydrolyzed, and acetyl-L-carnitine and L-carnitine concentrations return to baseline within 12 h. At normal circulating l-carnitine concentrations, renal l-carnitine reabsorption is highly efficient (90-99% of filtered load; clearance, 1-3 mL/min), but displays saturation kinetics. Thus, as circulating L-carnitine concentration increases (as after high-dose intravenous or oral administration of L-carnitine), efficiency of reabsorption decreases and clearance increases, resulting in rapid decline of circulating L-carnitine concentration to baseline. Elimination kinetics for acetyl-L-carnitine are similar to those for L-carnitine. There is evidence for renal tubular secretion of both L-carnitine and acetyl-L-carnitine. Future research should address the correlation of supplement dosage, changes and maintenance of tissue L-carnitine and acetyl-L-carnitine concentrations, and metabolic and functional changes and outcomes. Acetyl-L-carnitine (ALC), a physiological component of the L-carnitine family, has been proposed for treating Alzheimer's disease in pharmacological doses. As this condition requires prolonged therapy, its kinetics has been examined after a multiple dose regimen, involving different routes of administration, in 11 patients suffering from Senile Dementia of Alzheimer Type. The study design comprised a 3-day basal observation period, sham treatment with repeated blood sampling; treatment with 30 mg/kg iv [corrected] for 10 days (plasma kinetics was studied on the 7th day), and 50 days of 1.5 g/day [corrected] po given in three daily doses. Total acid soluble L-carnitine, L-carnitine and acetyl-L-carnitine in plasma and CSF were evaluated using an enantioselective radioenzyme assay. Short chain L-carnitine esters were calculated as the difference between total and free-L-carnitine. The plasma concentrations of individual components of the L-carnitine family did not change during the three days of the basal period, nor were they affected during the sham therapy period. Following the iv.bolus injections, the plasma concentrations showed a biphasic curve, with average t1/2 of 0.073 hr and 1.73 hr, respectively. At the end of oral treatment, plasma acetyl-L-carnitine and L-carnitine short chain esters were significantly higher than during the run-in phase. The CSF concentrations paralleled those in plasma, suggesting that ALC easily crosses the blood-brain barrier. ... The pharmacokinetics of acetyl-L-carnitine hydrochloride were investigated in 6 healthy volunteers of both sexes after iv injection of 500 mg of the drug, expressed as inner salt. Plasma concentrations and urinary excretion of acetyl-L-carnitine (A), L-carnitine (B) and total acid soluble L-carnitine fraction were evaluated over a period lasting from 24 h before to 48 h after the administration. Plasma concentrations of A increased quickly after administration and then declined reaching base values within 12 h. Conversely, plasma concentrations of B rose more slowly, reaching a peak in 30-60 min, and then declined to base values within 24 h. Most of the injected dose of acetyl-L-carnitine was recovered in the urine during the first 24 h after administration as B and A. Mean renal clearance of both A and B during the first 12 h after injection was higher than the base values, suggesting the presence of a saturable tubular reabsorption process which may counterbalance major changes occurring in plasma concentrations of L-carnitine pattern. /Acetyl-L-carnitine hydrochloride/ L-carnitine and its short-, medium- and long-chain acyl esters constitute the L-carnitine family. These compounds in the body are equilibrated according to a homeostatic equilibrium preserved and, when impaired, restored by a dynamic inter-exchange between L-carnitine and its esters, catalysed by carnitine acyl transferases, and a tubular reabsorption process with differentiated thresholds for each component. The interaction of these compounds with albumin and plasma proteins of rats, dogs and humans was carefully investigated by means of ultrafiltration and gel filtration techniques. Results obtained demonstrate that L-carnitine and its short-chain esters, namely acetyl-L-carnitine and propionyl-L-carnitine, do not interact with either albumin or plasma proteins; octanoyl-L-carnitine interacts in a measurable even if poor extent (12-30%), whereas palmitoyl-L-carnitine, a molecule with a detergent activity, is completely bound to albumin and plasma proteins. For more Absorption, Distribution and Excretion (Complete) data for ACETYL-L-CARNITINE (8 total), please visit the HSDB record page. Metabolism / Metabolites L-carnitine participates in a reversible transesterification reaction, in which an acyl group is transferred from coenzyme A to the hydroxyl group of L-carnitine ... Acetyl-L-carnitine ... is biosynthesized in this manner. Acetyl-L-carnitine (ALC) is an ester of the trimethylated amino acid, L-carnitine, and is synthesized in the human brain, liver, and kidney by the enzyme ALC-transferase. ... Experimental studies have shown that in rats and humans, kidneys are able to synthesize acetyl-L-carnitine from L-carnitine and either acetoacetate or beta-hydroxybutyrate ... |
| Toxicity/Toxicokinetics |
Interactions ... A ... study on 56 infertile males ... found the combination of L-carnitine ... and acetyl-L-carnitine ... led to significant improvement in sperm motility. ... combined administration of N-acetyl cysteine (an antioxidant and glutathione precursor that protects against A beta neurotoxicity), acetyl-L-carnitine (which raises ATP levels, protects mitochondria, and buffers A beta neurotoxicity), and S-adenosylmethionine (which facilitates glutathione usage and maintains acetylcholine levels) enhanced or maintain cognitive function, and attenuated or prevented aggression, in mouse models of aging and neurodegeneration. Enhancement of cognitive function was rapidly reversed upon withdrawal of the formulation and restored following additional rounds of supplementation. Behavioral abnormalities correlated with a decline in acetylcholine, which was also prevented by this nutriceutical combination, suggesting that neurotransmitter imbalance may contribute to their manifestation. Treatment with this nutriceutical combination was able to compensate for lack of dietary folate and vitamin E, coupled with administration of dietary iron as a pro-oxidant (which collectively increase homocysteine and oxidative damage to brain tissue), indicating that it provided antioxidant neuroprotection. ... Both HIV infection per se and antiretroviral drugs might contribute to oxidative stress and mitochondrial dysfunctions. ... /This/ study ... assessed zidovudine, stavudine and didanosine on U937 and CEM cell lines. All these drugs induced apoptosis and increased intracellular hydrogen peroxide but not superoxide anions. The addition of acetyl-l-carnitine (ALC) was able to prevent the pro-oxidant effect of the drugs tested. Supplementation with ALC, deficient in certain cohorts of HIV-infected individuals, especially on high active antiretroviral therapy regimen, has been associated with favorable effects. These data suggest that one of these effects could be a direct anti-oxidant action. Beagle dogs between 7.6 and 8.8 years of age administered a twice daily supplement of alpha-lipoic acid (LA) and acetyl-L-carnitine (ALC) over approximately 2 months made significantly fewer errors in reaching the learning criterion on two landmark discrimination tasks compared to controls administered a methylcellulose placebo. Testing started after a 5 day wash-in. The dogs were also tested on a variable delay version of a previously acquired spatial memory task; results were not significant. The improved performance on the landmark task of dogs supplemented with LA + ALC provides evidence of the effectiveness of this supplement in improving discrimination and allocentric spatial learning. /It was suggested/ that long-term maintenance on LA and ALC may be effective in attenuating age-associated cognitive decline by slowing the rate of mitochondrial decay and cellular aging. For more Interactions (Complete) data for ACETYL-L-CARNITINE (9 total), please visit the HSDB record page. |
| References | Serhiyenko VA, Serhiyenko AA. Cardiac autonomic neuropathy: Risk factors, diagnosis and treatment. World J Diabetes. 2018 Jan 15;9(1):1-24. doi: 10.4239/wjd.v9.i1.1. Review. |
| Additional Infomation |
Therapeutic Uses L-Carnitine, acetyl-L-carnitine, and/or propionyl-L-carnitine may be used for replacement therapy to restore normal carnitine concn and/or a normal nonesterified-to-esterified carnitine ratio ... /EXPL THER/ ... A recent meta-analysis of 21 double-blind clinical trials of ALC in the treatment of mild cognitive impairment and mild Alzheimer's disease showed significant efficacy vs. placebo. A meta-analysis of 4 clinical trials of LA for treatment of neuropathic deficits in diabetes showed significant efficacy vs. placebo. /EXPL THER/ An open, cross-over study was performed on a population of 24 geriatric patients hospitalized because of depressive syndrome. They were subdivided, according to Hamilton's Scale as modified for the aged, into low- and high-score subgroups. The study period covered 2 months. Half the patients received acetylcarnitine for 1 month and placebo thereafter (Group A); the other half received placebo and acetyl-carnitine thereafter (Group B). Statistical evaluation was twofold: parametrical analysis of variance was carried out on 4 subgroups (A1, A2, B1 and B2) and analysis of the score percentage modifications before and after treatment was performed on Groups A and B. The experimental results showed that acetylcarnitine treatment was highly effective and statistically significant in subgroups A1/B1, A2/B2, A1, B1 and B2. In particular, it should be noted that depressive tendencies were significantly modified in most groups, whereas general somatic symptoms as well as anxiety, asthenia and sleep disturbances proved to be little affected. Clinical evaluation, carried out by calculation of modifications in pre- and post-treatment score percentages, provided clear evidence that acetylcarnitine was particularly effective in patients showing more serious clinical symptoms. The drug caused no side-effects at the doses and regimens used. /EXPL THER/ ... Two multicenter clinical trials of subjects with either type 1 or type 2 diabetes found that treatment with acetyl-L-carnitine ... provided significant relief of nerve pain and improved vibration perception in those with diabetic neuropathy. The treatment was most effective in subjects with type 2 diabetes of short duration. For more Therapeutic Uses (Complete) data for ACETYL-L-CARNITINE (22 total), please visit the HSDB record page. Pharmacodynamics The complete physiological effects of acetycarnitine are still being studied. What has been discovered so far is that acetylcarinitine has positive effects on mental fatigue, neurodegenerative disorders, cognitive functions, peripheral neuropathy, and sperm motility. Specifically, in one study involving patients with HIV, patients on acetylcarnitine supplementation had increased CD4 cells, decreased lymphocyte apoptosis, improved polyneuropathy and cardiovascular damage, and decreased triglyceride and TNF alpha levels in the blood. Another study showed that acetylcarnitine increased glucose disposal in type 2 diabetic patients through possibly increasing the activity of glycogen synthase. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.9203 mL | 24.6015 mL | 49.2029 mL | |
| 5 mM | 0.9841 mL | 4.9203 mL | 9.8406 mL | |
| 10 mM | 0.4920 mL | 2.4601 mL | 4.9203 mL |