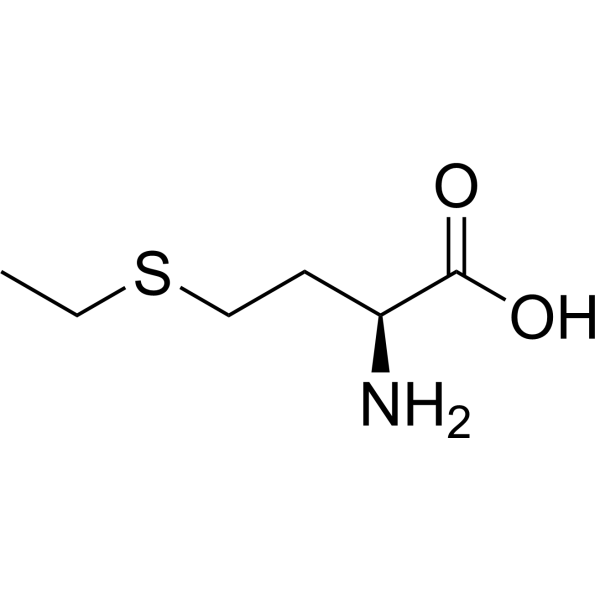
Physicochemical Properties
| Molecular Formula | C6H13NO2S |
| Exact Mass | 163.066 |
| CAS # | 13073-35-3 |
| PubChem CID | 25674 |
| Appearance | Crystals |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 310.4±37.0 °C at 760 mmHg |
| Melting Point | 280ºC (dec.)(lit.) |
| Flash Point | 141.5±26.5 °C |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C |
| Index of Refraction | 1.524 |
| LogP | 0.9 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 10 |
| Complexity | 108 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | CCSCC[C@@H](C(=O)O)N |
| InChi Key | GGLZPLKKBSSKCX-YFKPBYRVSA-N |
| InChi Code | InChI=1S/C6H13NO2S/c1-2-10-4-3-5(7)6(8)9/h5H,2-4,7H2,1H3,(H,8,9)/t5-/m0/s1 |
| Chemical Name | (2S)-2-amino-4-ethylsulfanylbutanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion IN RAT KIDNEY RADIOACTIVITY WAS MAINLY LOCALIZED IN MEDULLA FOLLOWING INJECTION WITH (14)C-ETHIONINE, WHEReAS (14)C-METHIONINE WAS HOMOGENEOUSLY DISTRIBUTED THROUGHOUT WHOLE PARENCHYMA IN RATS. Metabolism / Metabolites S-ADENOSYLETHIONINE IS A SUGGESTED ALKYLATING METABOLITE OF CARCINOGEN ETHIONINE. /FROM TABLE/ ETHIONINE IS BIOACTIVATED IN LIVER TO S-ADENOSYL-L-ETHIONINE, WHICH CAN ETHYLATE MACROMOLECULES CAUSING CELL ABNORMALITIES. ADENOSYLETHIONINE IS CHEM STABLE & CAN PENETRATE TO EMBRYONIC TISSUES & CAUSE TERATOGENIC EFFECTS. IN URINE OF TREATED RATS, 4 PRINCIPAL ETHIONINE EXCRETORY PRODUCTS WERE IDENTIFIED...N-ACETYLETHIONINE SULFOXIDE, S-ADENOSYLETHIONINE, ETHIONINE SULFOXIDE, & FREE ETHIONINE. |
| Toxicity/Toxicokinetics |
Interactions DL-METHIONINE @ 0.6-0.8% IN DIET FED SIMULTANEOUSLY WITH ETHIONINE PREVENTS DEVELOPMENT OF LIVER TUMORS /IN RATS/. /ETHIONINE/ IN RATS...LABELLING IN VIVO OF LIVER PROTEINS BY TRITIATED DIMETHYLNITROSAMINE WAS...LARGELY INHIBITED BY...ETHIONINE... /ETHIONINE/ ...TUMOR OF THE IRIS HAS BEEN OBSERVED IN RATS' EYES AFTER REPEATED SYSTEMIC ADMIN IN COMBINATION WITH N-2-FLUORENYLACETAMIDE. The effects of SA3443, a novel disulfide cmpd on acute liver injuries induced by carbon tetrachloride, D-galactosamine and DL-ethionine were studied in rats or mice. SA3443 (100-300 mg/kg, po) significantly suppressed the incr of serum transaminase activity and liver triglyceride content in the carbon tetrachloride or DL-ethionine induced model. ... |
| Additional Infomation |
Dl-ethionine is a white crystalline flakes. (NTP, 1992) L-ethionine is an S-ethylhomocysteine that has S-configuration at the chiral centre. It has a role as an antimetabolite and a carcinogenic agent. It is an enantiomer of a D-ethionine. 2-Amino-4-(ethylthio)butyric acid. An antimetabolite and methionine antagonist that interferes with amino acid incorporation into proteins and with cellular ATP utilization. It also produces liver neoplasms. Mechanism of Action /ETHIONINE/ INHIBITS THE INCORPORATION OF METHIONINE AND GLYCINE INTO RAT PROTEIN IN VIVO AND THE CONVERSION OF METHIONINE INTO CYSTEINE. ETHIONINE IS DEETHYLATED, AND THE ETHYL GROUP APPEARS IN ANALOGUES OF METABOLITES KNOWN TO PARTICIPATE IN TRANSMETHYLATION REACTIONS, EG ETHYL CHOLINE. IN THE RAT IT IS ITSELF INCORPORATED INTO ABNORMAL PROTEINS COMPETITIVELY, BUT ITS AFFINITY IS ONLY 1/600 THAT OF METHIONINE. /ETHIONINE/ ... The mechanism by which ethionine induces fatty liver is thought to be due to the inhibition of synthesis of very low density lipoprotein. ... The synthesis of lipoprotein is correlated with liver estradiol receptor concn, /an examination/ of the effect of ethionine on estradiol receptor concn in rat liver /was examined/. Estradiol receptor concn in both cytosolic and nuclear fractions were greatly decr by a single injection of ethionine at a dose of 0.5 mg/kg bw. In particular, the decr in nuclear receptor concn was observed a few hr after the injection and correlated with the decr in triglyceride content in the very low density lipoprotein fraction. /Data suggests/ that liver estradiol receptor is involved in the pathogenesis of ethionine induced fatty liver of rats. |
Solubility Data
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |