Physicochemical Properties
| Molecular Formula | C34H49NO2 |
| Molecular Weight | 503.76 |
| Exact Mass | 503.376 |
| CAS # | 2486052-18-8 |
| PubChem CID | 155105032 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 9.3 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 11 |
| Heavy Atom Count | 37 |
| Complexity | 860 |
| Defined Atom Stereocenter Count | 6 |
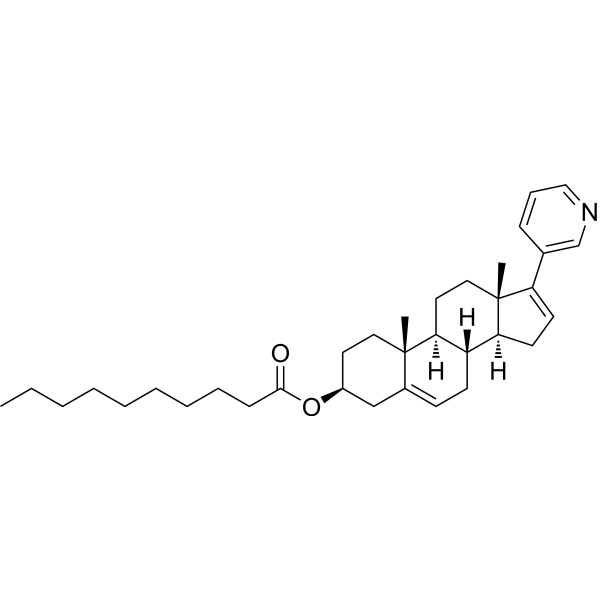
| SMILES | CCCCCCCCCC(=O)O[C@H]1CC[C@@]2([C@H]3CC[C@]4([C@H]([C@@H]3CC=C2C1)CC=C4C5=CN=CC=C5)C)C |
| InChi Key | XPCSGTPPHYORKJ-YHXMLEJGSA-N |
| InChi Code | InChI=1S/C34H49NO2/c1-4-5-6-7-8-9-10-13-32(36)37-27-18-20-33(2)26(23-27)14-15-28-30-17-16-29(25-12-11-22-35-24-25)34(30,3)21-19-31(28)33/h11-12,14,16,22,24,27-28,30-31H,4-10,13,15,17-21,23H2,1-3H3/t27-,28-,30-,31-,33-,34+/m0/s1 |
| Chemical Name | [(3S,8R,9S,10R,13S,14S)-10,13-dimethyl-17-pyridin-3-yl-2,3,4,7,8,9,11,12,14,15-decahydro-1H-cyclopenta[a]phenanthren-3-yl] decanoate |
| Synonyms | Abiraterone decanoate; PRL-02; 2486052-18-8; U6XMS339F5; UNII-U6XMS339F5; Abiraterone decanoate?; CHEMBL5314964; SCHEMBL22414033; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | CYP17 |
| ln Vitro | Abiraterone Decanoate is the decanoate form of abiraterone, a steroidal compound with antiandrogen activity. Abiraterone inhibits the enzymatic activity of steroid 17alpha-monooxygenase (17alpha-hydrolase/C17,20 lyase complex; CYP17A1), a member of the cytochrome p450 family that catalyzes the 17alpha-hydroxylation of steroid intermediates involved in testosterone synthesis. Administration of this agent may suppress testosterone production by both the testes and the adrenals to castrate-range levels. |
| ln Vivo | Chemical castration resulted in a plasma testosterone (T) decrease of 72% from intact. T was further reduced to sub-castrate levels by both AA and AD (98.6% and 99.7% maximum decrease from castrate baseline, respectively). All dose levels of AD were highly effective in reducing plasma T yet led to abiraterone plasma concentrations (Cmax, AUC, Cmin) that were much less than those associated with steady-state clinical levels from oral AA. Sustained T suppression was observed following all single AD IM doses for 14 weeks, the last timepoint tested.The addition of a glucocorticoid replacement (DEX or MPA) starting at week 9 further reduced T in all AD groups. Conclusions: Single-dose IM AD suppressed T to the same or greater extent than single-dose oral AA in castrate monkeys, while providing much lower abiraterone exposures than the oral acetate prodrug. As such, IM AD may offer an improved risk-benefit profile due to the consistently lower abiraterone levels. The durable, profound T reductions provided by all AD doses are consistent with a 3-month clinical regimen that can be given in conjunction with Lupron. IM AD may thus offer patients with CSPC and CRPC a more convenient, safe and effective alternative than daily oral AA.[1] |
| Animal Protocol |
Background: Oral abiraterone acetate (AA) is a standard of care for castration-resistant (CRPC) and castration-sensitive prostate cancer (CSPC). Due to poor oral bioavailability, the recommended AA dose is 1,000 mg (4 x 250mg) once-daily on an empty stomach. The daily oral regimen produces high peak plasma concentrations that may be associated with safety issues (e.g., hepatotoxicity) and low trough concentrations that may be associated with inadequate CYP17 inhibition. AD is one of a series of novel abiraterone prodrugs that were designed to provide a controlled release of abiraterone and long-acting CYP17 inhibition with IM delivery. Following successful preclinical pilot studies, AD was further developed into a clinically acceptable formulation (PRL-02) and its PK/PD characteristics were evaluated and compared to oral AA in a castrate monkey pharmacology model in support of future clinical development. [1] Methods: Sexually mature male cynomolgus monkeys underwent chemical castration using Lupron depot. Plasma samples were analyzed for prodrug, abiraterone and steroid concentrations following a single oral AA dose (5, 15 or 45 mg/kg) and a single IM AD injection (10, 30 or 100 mg/kg) (n=3/dose group). The combined activity of IM AD plus glucocorticoid replacement (single 0.5 mg/kg IM dexamethasone (DEX) dose or weekly doses of 1.29 mg/kg IM methylprednisolone acetate (MPA)) was also evaluated. AD, abiraterone and adrenal steroid levels were evaluated by LC-MS/MS. [1] |
| References |
[1]. Abiraterone decanoate (AD): Potent and long-acting activity of a novel intramuscular (IM) abiraterone prodrug depot in a castrate monkey model. Journal of Clinical Oncology,Volume 39, Issue 6_suppl. |
| Additional Infomation | Abiraterone Decanoate is the decanoate form of abiraterone, a steroidal compound with antiandrogen activity. Abiraterone inhibits the enzymatic activity of steroid 17alpha-monooxygenase (17alpha-hydrolase/C17,20 lyase complex; CYP17A1), a member of the cytochrome p450 family that catalyzes the 17alpha-hydroxylation of steroid intermediates involved in testosterone synthesis. Administration of this agent may suppress testosterone production by both the testes and the adrenals to castrate-range levels. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9851 mL | 9.9254 mL | 19.8507 mL | |
| 5 mM | 0.3970 mL | 1.9851 mL | 3.9701 mL | |
| 10 mM | 0.1985 mL | 0.9925 mL | 1.9851 mL |