Description: ACY-1083 is a novel, potent, selective and brain-penetrant HDAC6 inhibitor with an IC50 of 3 nM. ACY-1083 shows 260-fold selectivity towards HDAC6 vs other HDACs. HDAC6 inhibition by ACY-1083 prevented cisplatin-induced mechanical allodynia, and also completely reversed already existing cisplatin-induced mechanical allodynia, spontaneous pain, and numbness. These findings were confirmed using the established HDAC6 inhibitor ACY-1215 (Ricolinostat), which is currently in clinical trials for cancer treatment. Mechanistically, treatment with the HDAC6 inhibitor increased α-tubulin acetylation in the peripheral nerve. In addition, HDAC6 inhibition restored the cisplatin-induced reduction in mitochondrial bioenergetics and mitochondrial content in the tibial nerve, indicating increased mitochondrial transport. At a later time point, dorsal root ganglion mitochondrial bioenergetics also improved. HDAC6 inhibition restored the loss of intraepidermal nerve fiber density in cisplatin-treated mice. These results demonstrate that pharmacological inhibition of HDAC6 completely reverses all the hallmarks of established cisplatin-induced peripheral neuropathy by normalization of mitochondrial function in dorsal root ganglia and nerve, and restoration of intraepidermal innervation. These results are especially promising because one of the HDAC6 inhibitors tested here is currently in clinical trials as an add-on cancer therapy, highlighting the potential for a fast clinical translation of these findings.
Physicochemical Properties
| Molecular Formula | C17H18F2N4O2 |
| Molecular Weight | 348.3472 |
| Exact Mass | 348.139 |
| CAS # | 1708113-43-2 |
| PubChem CID | 91844718 |
| Appearance | White to off-white solid powder |
| LogP | 2.5 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 25 |
| Complexity | 454 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | FC1(C([H])([H])C([H])([H])C(C2C([H])=C([H])C([H])=C([H])C=2[H])(C([H])([H])C1([H])[H])N([H])C1=NC([H])=C(C(N([H])O[H])=O)C([H])=N1)F |
| InChi Key | MRWCVKLVSWYIFO-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C17H18F2N4O2/c18-17(19)8-6-16(7-9-17,13-4-2-1-3-5-13)22-15-20-10-12(11-21-15)14(24)23-25/h1-5,10-11,25H,6-9H2,(H,23,24)(H,20,21,22) |
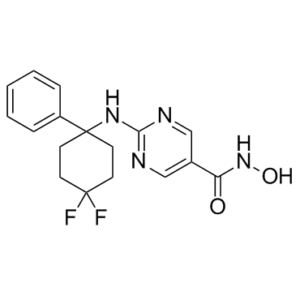
| Chemical Name | 2-[(4,4-difluoro-1-phenylcyclohexyl)amino]-N-hydroxypyrimidine-5-carboxamide |
| Synonyms | ACY-1083; ACY 1083; ACY1083 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Treatment with ACY-1083 (30 and 300 nM) greatly enhanced cell viability in a dose-dependent way [2]. |
| ln Vivo | ACY-1083 (ip; 10 mg/kg; for 7 days) efficiently alleviates cisplatin-induced mechanical allodynia in C57BL/6J mice ACY-1083 (oral dose 3 mg/kg ACY-1083 for 7 days ) can reverse paclitaxel-induced mechanical allodynia in adult male SD rats. In mice treated 5 mg/kg ACY-1083 via intraperitoneal (ip) injection, the maximum plasma concentration (Cmax) was 936 ng/mL, the half-life (T1/2) was 3.5 hours, and the bioactive plasma exposure was 8 postdose. Hour. |
| Cell Assay |
Cell Viability Assay [2] Cell Types: Mouse Hippocampus (HT22) Cell Tested Concentrations: 30 and 300 nM Incubation Duration: Experimental Results: Cell viability was Dramatically improved. |
| Animal Protocol |
Animal/Disease Models: 8-10 weeks old adult male C57BL/6J mice [1] Doses: 3 or 10 mg/kg Route of Administration: intraperitoneal (ip) injection; daily; 7 days Experimental Results: 10 mg/kg dose can alleviate cisplatin ( 2.3 mg/kg), but not the 3 mg/kg dose. Animal/Disease Models: Adult male Sprague Dawley rats [1] Doses: 3 mg/kg Route of Administration: Two oral doses per day for 7 days. Experimental Results: Paclitaxel (6 mg/kg or 12 mg/kg)-induced mechanical allodynia was reversed. |
| References |
[1]. HDAC6 inhibition effectively reverses chemotherapy-induced peripheral neuropathy. Pain. 2017 Jun;158(6):1126-1137. [2]. Isoform 6-selective histone deacetylase inhibition reduces lesion size and brain swelling following traumatic brain injury and hemorrhagic shock. J Trauma Acute Care Surg. 2019 Feb;86(2):232-239. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~200 mg/mL (~574.14 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 5 mg/mL (14.35 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 50.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 5 mg/mL (14.35 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 50.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 5 mg/mL (14.35 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 50.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8707 mL | 14.3534 mL | 28.7068 mL | |
| 5 mM | 0.5741 mL | 2.8707 mL | 5.7414 mL | |
| 10 mM | 0.2871 mL | 1.4353 mL | 2.8707 mL |