A-83-01 is an ALK inhibibitor. It was found that A-83-01 inhibited the transcriptional activity induced by TGF-beta type I receptor ALK-5 and that by activin type IB receptor ALK-4 and nodal type I receptor ALK-7, the kinase domains of which are structurally highly related to those of ALK-5. A-83-01 was found to be more potent in the inhibition of ALK5 than a previously described ALK-5 inhibitor, SB-431542, and also to prevent phosphorylation of Smad2/3 and the growth inhibition induced by TGF-beta. A-83-01 inhibited the epithelial-to-mesenchymal transition induced by TGF-beta, suggesting that A-83-01 and related molecules may be useful for preventing the progression of advanced cancers.
Physicochemical Properties
| Molecular Formula | C25H19N5S |
| Molecular Weight | 421.52 |
| Exact Mass | 421.136 |
| Elemental Analysis | C, 71.23; H, 4.54; N, 16.61; S, 7.61 |
| CAS # | 909910-43-6 |
| Related CAS # | A 83-01 sodium;2828431-89-4;A 83-01;909910-43-6 |
| PubChem CID | 16218924 |
| Appearance | Off-white to light yellow solid powder |
| Density | 1.27g/cm3 |
| Boiling Point | 590ºC at 760 mmHg |
| Flash Point | 310.6ºC |
| Index of Refraction | 1.706 |
| LogP | 5.786 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 31 |
| Complexity | 609 |
| Defined Atom Stereocenter Count | 0 |
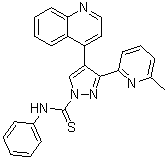
| SMILES | S=C(N1C=C(C2C3C(=CC=CC=3)N=CC=2)C(C2C=CC=C(C)N=2)=N1)NC1C=CC=CC=1 |
| InChi Key | HIJMSZGHKQPPJS-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C25H19N5S/c1-17-8-7-13-23(27-17)24-21(19-14-15-26-22-12-6-5-11-20(19)22)16-30(29-24)25(31)28-18-9-3-2-4-10-18/h2-16H,1H3,(H,28,31) |
| Chemical Name | 3-(6-methylpyridin-2-yl)-N-phenyl-4-(quinolin-4-yl)-1H-pyrazole-1-carbothioamide |
| Synonyms | A8301; A 8301; 909910-43-6; A 83-01; 3-(6-methylpyridin-2-yl)-N-phenyl-4-(quinolin-4-yl)-1H-pyrazole-1-carbothioamide; A-83-01; A83-01; Stemolecule A83-01; 3-(6-methylpyridin-2-yl)-N-phenyl-4-quinolin-4-ylpyrazole-1-carbothioamide; X3ZNM7QJ2Q; A-8301 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product is not stable in solution, please use freshly prepared working solution for optimal results. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | ALK5: 12 nM (IC50); ALK4: 45 nM (IC50); ALK7: 7.5 nM (IC50) |
| ln Vitro | A 83-01 is a strong inhibitor of TGF-β type I receptor ALK5 kinase, ALK4 and ALK7. It also weakly suppresses transcription induced by constitutively active ALK-6, ALK-2, ALK-3, and ALK-1. In Mv1Lu cells, it reduces the level of ALK-5-induced transcription with an IC50 of 12 nM. It also blocks transcription induced by ALK4-TD and ALK7-TD, with IC50s of 45 nM and 7.5 nM in R4-2 cells. At concentrations of 0.03–10 μM, A 83-01 effectively counteracts the growth-inhibitory effects of TGF-β, and at 3 μM, it totally eliminates them. HaCaT cells' Smad activation caused by TGF-β is inhibited by A 83-01 (1–10 μM)[1]. A 83-01 (1 μM) does not alter cell proliferation, but it does reduce TGF-β1-induced cell motility, adhesion, and invasion in HM-1 cells[2]. |
| ln Vivo | Mice without body weight or neurobehavioral performance showed a substantial increase in the quarter rate when given an intraperitoneal dose of 83-01 (50, 150, and 500 μg/mouse) [2]. In mice, 83-01 M109 cells administered intraperitoneally at a dose of 0.5 mg/kg had strong antitumor effects [3]. |
| Enzyme Assay | The original constructions of constitutively active forms of ALK-1 through -7 in mammalian expression vectors were described previously. The 9xCAGA-luciferase plasmid contains nine repeats of the CAGA Smad binding element driving luciferase expression. The (BRE)2-luciferase plasmid contains two repeats of the BMP responsive elements of the Id1 promoter cloned upstream of a minimal promoter driving luciferase expression. The 3GC2-luciferase plasmid contains three repeats of a GC-rich sequence derived from the proximal BMP responsive element of the Smad6 promoter. [1] |
| Cell Assay | Mv1Lu cells were seeded in duplicate at a density of 2.5 × 104 cells/well in 24-well plates. The following day, cells were pretreated for 1 h with 1 µM small molecule inhibitors and then cultured with TGF-β 1 ng/mL) for 24 h, 48 h, or 72 h. Cells were trypsinized and counted with a Coulter counter. To explore whether small molecule inhibitors reduced the growth-inhibitory effects of TGF-β in concentration-dependent fashion, Mv1Lu cells were seeded as above and pretreated for 1 h with various concentrations of small molecule inhibitors. After pretreatment, cells were cultured with TGF-β 1 ng/mL) for 48 h and counted.[1] |
| Animal Protocol | Female B6C3F1 mice used for the in vivo studies are maintained under specific pathogen-free conditions. To evaluate the effect of A 83-01 on the survival of mice bearing peritoneal dissemination, HM-1 cells (1×106) are injected into the abdominal cavity via the left flank of the mouse. Starting the next day, A 83-01 (150 μg/body) or vehicles (PBS with 0.5% DMSO) are injected into the abdominal cavity three times per week. Mice are euthanized before reaching the moribund state. |
| References |
[1]. Tojo M, et al. The ALK-5 inhibitor A-83-01 inhibits Smad signaling and epithelial-to-mesenchymal transition by transforming growth factor-beta. Cancer Sci. 2005 Nov;96(11):791-800. [2]. Yamamura S, et al. The activated transforming growth factor-beta signaling pathway in peritoneal metastases is a potential therapeutic target in ovarian cancer. Int J Cancer. 2012 Jan 1;130(1):20-8. [3]. Taniguchi Y, et al. Enhanced antitumor efficacy of folate-linked liposomal Adriamycin with TGF-β type I receptor inhibitor. Cancer Sci. 2010 Oct;101(10):2207-13 |
| Additional Infomation | Transforming growth factor (TGF)-beta signaling facilitates tumor growth and metastasis in advanced cancer. Use of inhibitors of TGF-beta signaling may thus be a novel strategy for the treatment of patients with such cancer. In this study, we synthesized and characterized a small molecule inhibitor, A-83-01, which is structurally similar to previously reported ALK-5 inhibitors developed by Sawyer et al. (2003) and blocks signaling of type I serine/threonine kinase receptors for cytokines of the TGF-beta superfamily (known as activin receptor-like kinases; ALKs). Using a TGF-beta-responsive reporter construct in mammalian cells, we found that A-83-01 inhibited the transcriptional activity induced by TGF-beta type I receptor ALK-5 and that by activin type IB receptor ALK-4 and nodal type I receptor ALK-7, the kinase domains of which are structurally highly related to those of ALK-5. A-83-01 was found to be more potent in the inhibition of ALK5 than a previously described ALK-5 inhibitor, SB-431542, and also to prevent phosphorylation of Smad2/3 and the growth inhibition induced by TGF-beta. In contrast, A-83-01 had little or no effect on bone morphogenetic protein type I receptors, p38 mitogen-activated protein kinase, or extracellular regulated kinase. Consistent with these findings, A-83-01 inhibited the epithelial-to-mesenchymal transition induced by TGF-beta, suggesting that A-83-01 and related molecules may be useful for preventing the progression of advanced cancers.[1] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~25 mg/mL (~59.31 mM) H2O : < 0.1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.08 mg/mL (4.93 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: 1.25 mg/mL (2.97 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 3: ≥ 1.25 mg/mL (2.97 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3724 mL | 11.8618 mL | 23.7237 mL | |
| 5 mM | 0.4745 mL | 2.3724 mL | 4.7447 mL | |
| 10 mM | 0.2372 mL | 1.1862 mL | 2.3724 mL |