|
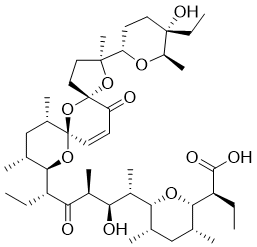
A28086B is an ionophore antibiotic structurally related to narasin, it was derived from fermentation and the structure was elucidated using electron impact and field desorption mass spectrometry. The ionic fragmentation of A28086B and its derivatives is discussed and compared with that of salinomycin, whose structure has been determined by X-ray diffraction.
|
Physicochemical Properties
| Molecular Formula |
C43H70O11
|
| Molecular Weight |
763.0093
|
| Exact Mass |
762.492
|
| Elemental Analysis |
C, 67.69; H, 9.25; O, 23.06
|
| CAS # |
58439-94-4
|
| Related CAS # |
|
| PubChem CID |
191478
|
| Appearance |
Typically exists as solid at room temperature
|
| LogP |
6.642
|
| Hydrogen Bond Donor Count |
3
|
| Hydrogen Bond Acceptor Count |
11
|
| Rotatable Bond Count |
12
|
| Heavy Atom Count |
54
|
| Complexity |
1400
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
CCC(C1OC(C(C)C(O)C(C)C(=O)C(CC)C2OC3(OC4(CCC(C)(O4)C4CCC(O)(CC)C(C)O4)C(=O)C=C3)C(C)CC2C)C(C)CC1C)C(O)=O |c:41|
|
| InChi Key |
FBLJTCGAXDPRJH-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C43H70O11/c1-12-30(35(46)27(8)34(45)28(9)36-23(4)21-24(5)37(51-36)31(13-2)39(47)48)38-25(6)22-26(7)42(52-38)18-15-32(44)43(54-42)20-19-40(11,53-43)33-16-17-41(49,14-3)29(10)50-33/h15,18,23-31,33-34,36-38,45,49H,12-14,16-17,19-22H2,1-11H3,(H,47,48)
|
| Chemical Name |
2-[6-[6-[3-(5-ethyl-5-hydroxy-6-methyloxan-2-yl)-3,10,12-trimethyl-15-oxo-4,6,8-trioxadispiro[4.1.57.35]pentadec-13-en-9-yl]-3-hydroxy-4-methyl-5-oxooctan-2-yl]-3,5-dimethyloxan-2-yl]butanoic acid
|
| Synonyms |
| A28086B; A-28086B; Narasin B; A28086B; Antibiotic A 28086B; (4S)-20-Deoxy-20-oxo-4beta-methylsalinomycin; A 28086B; A-28086 B; 2-[6-[6-[3-(5-ethyl-5-hydroxy-6-methyloxan-2-yl)-3,10,12-trimethyl-15-oxo-4,6,8-trioxadispiro[4.1.57.35]pentadec-13-en-9-yl]-3-hydroxy-4-methyl-5-oxooctan-2-yl]-3,5-dimethyloxan-2-yl]butanoic acid; A 28086B |
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder-20°C 3 years
4°C 2 years
In solvent -80°C 6 months
-20°C 1 month
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
|
Biological Activity
| Targets |
Antibiotic
|
| ln Vitro |
Using electron impact and field desorption mass spectrometry, structures are proposed for narasin and a related ionophore, A28086B, derived from fermentation. The ionic fragmentation of these compounds and their derivatives is discussed and compared with that of salinomycin, whose structure has been determined by X-ray diffraction[1].
|
| References |
Biomed Mass Spectrom.1976 Dec;3(6):272-7.
|
|
Solubility Data
| Solubility (In Vitro) |
|
| Solubility (In Vivo) |
(Please use freshly prepared in vivo formulations for optimal results.)
|
| Preparing Stock Solutions |
|
1 mg |
5 mg |
10 mg |
| 1 mM |
1.3106 mL |
6.5530 mL |
13.1060 mL |
| 5 mM |
0.2621 mL |
1.3106 mL |
2.6212 mL |
| 10 mM |
0.1311 mL |
0.6553 mL |
1.3106 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles. |