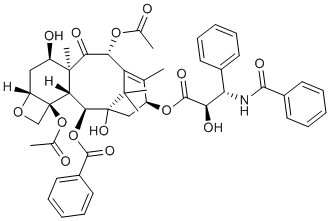
7-epi-Taxol, the epimer of taxol (paclitaxel), is a major and active metabolite of taxol isolated from Taxus brevifolia with comparable anticancr/antitubulin activity to that of paclitaxek/taxol. Similar to paclitaxel, 7-epi-Taxol promotes microtubule stabilization and inhibits microtubule depolymerization. It is also active against cell proliferation. Therefore, the overall biological activity of taxol remains unaffected by its presence.
Physicochemical Properties
| Molecular Formula | C47H51NO14 |
| Molecular Weight | 853.9062 |
| Exact Mass | 853.33 |
| CAS # | 105454-04-4 |
| Related CAS # | 105454-04-4 |
| PubChem CID | 184492 |
| Appearance | White to off-white solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 957.1±65.0 °C at 760 mmHg |
| Melting Point | 168-170?C |
| Flash Point | 532.6±34.3 °C |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.637 |
| LogP | 7.38 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 14 |
| Rotatable Bond Count | 14 |
| Heavy Atom Count | 62 |
| Complexity | 1790 |
| Defined Atom Stereocenter Count | 11 |
| SMILES | O1C([H])([H])[C@@]2([C@@]1([H])C([H])([H])[C@]([H])([C@@]1(C([H])([H])[H])C([C@@]([H])(C3=C(C([H])([H])[H])[C@]([H])(C([H])([H])[C@](C3(C([H])([H])[H])C([H])([H])[H])([C@]([H])([C@]21[H])OC(C1C([H])=C([H])C([H])=C([H])C=1[H])=O)O[H])OC([C@@]([H])([C@]([H])(C1C([H])=C([H])C([H])=C([H])C=1[H])N([H])C(C1C([H])=C([H])C([H])=C([H])C=1[H])=O)O[H])=O)OC(C([H])([H])[H])=O)=O)O[H])OC(C([H])([H])[H])=O |
| InChi Key | RCINICONZNJXQF-LYTKHFMESA-N |
| InChi Code | InChI=1S/C47H51NO14/c1-25-31(60-43(56)36(52)35(28-16-10-7-11-17-28)48-41(54)29-18-12-8-13-19-29)23-47(57)40(61-42(55)30-20-14-9-15-21-30)38-45(6,32(51)22-33-46(38,24-58-33)62-27(3)50)39(53)37(59-26(2)49)34(25)44(47,4)5/h7-21,31-33,35-38,40,51-52,57H,22-24H2,1-6H3,(H,48,54)/t31-,32+,33+,35-,36+,37+,38-,40-,45+,46-,47+/m0/s1 |
| Chemical Name | [(1S,2S,3R,4S,7R,9R,10S,12R,15S)-4,12-diacetyloxy-15-[(2R,3S)-3-benzamido-2-hydroxy-3-phenylpropanoyl]oxy-1,9-dihydroxy-10,14,17,17-tetramethyl-11-oxo-6-oxatetracyclo[11.3.1.03,10.04,7]heptadec-13-en-2-yl] benzoate |
| Synonyms | 7-epi-Taxol; 7-epi-Paclitaxel; 7-Epitaxol; 7-epi-Taxol; 105454-04-4; 7-Epipaclitaxel; 7-epi-Paclitaxel; Epitaxol; [(1S,2S,3R,4S,7R,9R,10S,12R,15S)-4,12-diacetyloxy-15-[(2R,3S)-3-benzamido-2-hydroxy-3-phenylpropanoyl]oxy-1,9-dihydroxy-10,14,17,17-tetramethyl-11-oxo-6-oxatetracyclo[11.3.1.03,10.04,7]heptadec-13-en-2-yl] benzoate; 7-Epipaclitaxel? |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Microtubule/Tubulin; impurity/metabolite of paclitaxel |
| ln Vitro | 7-epi-Taxol (7-Epitaxol) is a taxol metabolite[1]. In terms of cell replication, microtubule bundle formation, and in vitro microtubule polymerization, 7-epi-Taxol exhibits activity similar to that of taxol[2]. |
| ln Vivo | Reversed-phase high-performance liquid chromatography/mass spectrometry (LC/MS), with an atmospheric-pressure chemical ionization (APCI) interface, has been applied to the identification of metabolites and derivatives of paclitaxel (taxol) in plasma and urine of patients treated with this new anticancer drug. Protonated molecules with substantial fragmentation were obtained using this ionization technique. The three ion series observed are characteristic of the intact molecule, the taxane ring, and the side chain at C13. Their analysis gives information about chemical modifications of the taxane structure at different positions of the molecule. Urine and plasma extracts were evaluated using the capacity to perform MS analysis directly on the entire effluent from conventional LC columns. Excellent spectra were obtained with 50 pmol of separated compounds in full scan mode. This technique allowed highly sensitive identification of 6 alpha-hydroxytaxol, the major human biliary metabolite, and of 7-epitaxol in extracts of plasma and urine from patients. Taxol hydrolysis derivatives were observed for the first time in urine 24 hours after the end of the infusion period. Sensitivity could be increased further using single ion monitoring (SIM) mode, once a target derivative was identified. These results demonstrate that LC/MS with an APCI interface is useful for the characterization and pharmacokinetic analysis of taxoids in biological matrices[1]. |
| Cell Assay | The hydrolysis products of taxol have been isolated by high-performance liquid chromatography and identified by nuclear magnetic resonance and mass spectroscopy. In contrast to taxol, the major hydrolysis product, baccatin III, has little cytotoxic activity and does not promote in vitro microtubule assembly. In cell culture medium, the concentration of taxol decreases with time and 7-epitaxol, which exhibits properties comparable to those of taxol both on cells and on in vitro microtubuli polymerization, is formed. Baccatin III is found in small quantities in the cell medium, although it is barely detectable within the cells. It is concluded that 7-epitaxol is the major derivative of taxol found in cells and that its presence does not alter, in a major way, the overall biological activity of taxol[2]. |
| References |
[1]. Paclitaxel metabolites in human plasma and urine: identification of 6 alpha-hydroxytaxol, 7-epitaxol and taxol hydrolysis products using liquid chromatography/atmospheric-pressure chemical ionization mass spectrometry. Rapid Commun Mass Spectrom. 1995;9(6):495-502. [2]. Taxol is converted to 7-epitaxol, a biologically active isomer, in cell culture medium. J Pharmacol Exp Ther. 1987 Aug;242(2):692-8. |
| Additional Infomation |
7-epi-Taxol has been reported in Taxus cuspidata, Corylus avellana, and other organisms with data available. A cyclodecane isolated from the bark of the Pacific yew tree, TAXUS BREVIFOLIA. It stabilizes MICROTUBULES in their polymerized form leading to cell death. |
Solubility Data
| Solubility (In Vitro) | DMSO: ~100 mg/mL (~117.1 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (2.93 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (2.93 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.1711 mL | 5.8554 mL | 11.7108 mL | |
| 5 mM | 0.2342 mL | 1.1711 mL | 2.3422 mL | |
| 10 mM | 0.1171 mL | 0.5855 mL | 1.1711 mL |