Physicochemical Properties
| Molecular Formula | C23H25NO7 |
| Molecular Weight | 427.45 |
| Exact Mass | 427.163 |
| CAS # | 2730-82-7 |
| PubChem CID | 332237 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.29g/cm3 |
| Boiling Point | 786.9ºC at 760mmHg |
| Flash Point | 429.7ºC |
| Vapour Pressure | 9.93E-25mmHg at 25°C |
| Index of Refraction | 1.589 |
| LogP | 3.075 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 31 |
| Complexity | 815 |
| Defined Atom Stereocenter Count | 1 |
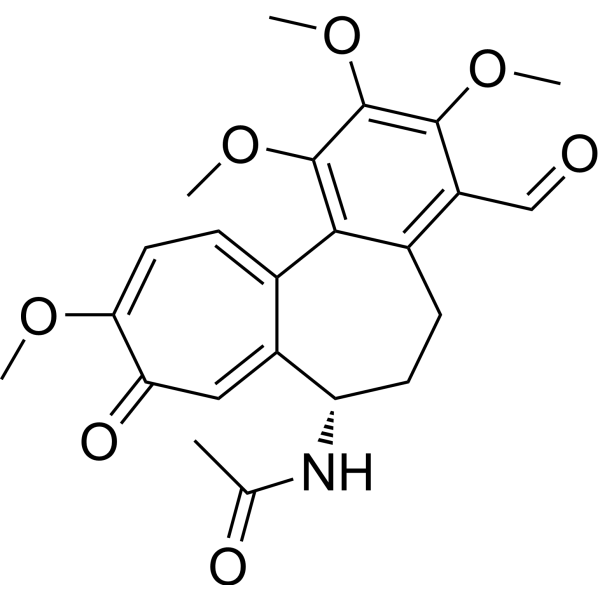
| SMILES | CC(=O)N[C@H]1CCC2=C(C(=C(C(=C2C3=CC=C(C(=O)C=C13)OC)OC)OC)OC)C=O |
| InChi Key | JUWAUXATKRGHCK-KRWDZBQOSA-N |
| InChi Code | InChI=1S/C23H25NO7/c1-12(26)24-17-8-6-14-16(11-25)21(29-3)23(31-5)22(30-4)20(14)13-7-9-19(28-2)18(27)10-15(13)17/h7,9-11,17H,6,8H2,1-5H3,(H,24,26)/t17-/m0/s1 |
| Chemical Name | N-[(7S)-4-formyl-1,2,3,10-tetramethoxy-9-oxo-6,7-dihydro-5H-benzo[a]heptalen-7-yl]acetamide |
| Synonyms | 4-Formylcolchicine; 2730-82-7; NSC328403; NSC-328403; CHEMBL155693; DTXSID30950024; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Microtubule/Tubulin |
| ln Vitro | 4-Formylcolchicine (compound 7) exhibits antiproliferative action against A549, HT-29, and HCT116 cells, with IC50s of 1.007, 0.128, and 0.054 µM, respectively[1]. |
| ln Vivo | Liver fibrosis was induced by chronically (7 weeks) administering CCl4, to rats. Animals were divided into four groups: (a) controls, (b) treated with CCI, alone, (c) treated with CCl4 and colchicine and (d) treated with CCl4 and 4-Formylcolchicine bound to lactosaminated serum albumin (FC-LASA). Liver dysfunction was monitored by biochemical tests (alkaline phosphatase [ALP], γ-glutamyltransferase [γGT], aspartate and alanine transaminases [AST and ALT], albumin and total bilirubin). Fibrosis was evaluated by determining hydroxyproline and by microscopic examination. The exposure to CCl4 produced major alterations of liver structure and collagen deposition. These effects were partially counteracted by colchicine and to a greater extent by FC-LASA. Morphological findings paralleled biochemical data. The information reported here indicates that colchicine has an antifibrotic activity on the liver of intoxicated rats and that FC-LASA is more active than colchicine itself as an antifibrotic agent [1]. |
| Cell Assay |
Cell Viability Assay[1] Cell Types: A549, HT-29, HCT116 cells Tested Concentrations: 0-5 µM Incubation Duration: 24 h Experimental Results: Inhibited cell growth with IC50s of 1.007, 0.128, 0.054 µM for A549, HT-29, HCT116 cells, respectively. |
| Animal Protocol |
Effect of colchicine and its derivatives on liver fibrosis [1] To study the effects of colchicine derivatives on liver fibrosis in vivo, we used adult Sprague-Dawley male rats (about 250 g body wt.). The animals were divided into four groups. (A) Controls received 2 ml.kg-1 body wt. mineral oil by intraperitoneal route and 250 ~tl of saline by injection into the tail vein three times a week for 7 weeks; (B) Animals were intoxicated with carbon tetrachloride. They were treated as those of groups A, except that they received a mixture of mineral oil and carbon tetrachloride (65:35 v/v, 2 ml'kg-1 body wt.); (C) The animals belonging to this group were treated as those of group B but they received colchicine (120 ~tg-kg-1 body wt., dissolved in saline) instead of saline; and (D) animals were treated as in (C), but colchicine was replaced by FC-LASA (120 ~tg.kg-~ body wt., as free colchicine). Doses were adjusted to body wt. and expressed as free colchicine. Owing to the high toxicity of CC14 (only about 40-50% of the animals survived 7 weeks' treatment), we began the experiment with 50 animals (various treatments) and 15 animals (controls), to have the final number of animals reported in Table 1. At the end of the experiment, animals were anaesthetised with diethyl ether and their blood was drained by heart puncture and collected in pre-heparinated vessels. Plasma was prepared by centrifugation at 1000 x g for 10 min and some biochemical parameters (alkaline phosphatase (ALP), aspartate and alanine transaminases (AST and ALT) y-glutamyl transferase (yGT), albumin and total bilirubin) were determined by commercial kits. Livers were used to verify collagen content (as hydroxyproline) and for histological examination. |
| References |
[1]. Formylcolchicine bound to lactosaminated serum albumin is a more active antifibrotic agent than free colchicine. Clin Chim Acta . 1996 Oct 29;254(2):149-57. [2]. Design, Synthesis, and Antitumor Activity of 4-Halocolchicines and Their Pro-drugs Activated by Cathepsin B. ACS Med Chem Lett. 2011 Mar 4;2(5):348-52. |
| Additional Infomation |
Histological findings agree with the biochemical tests and especially with the determination of liver hydroxyproline, which assesses liver fibrosis better than microscopic evaluation. All tests indicate that FC-LASA is more effective than colchicine to decrease liver fibrosis following CC14 administration in the rat. [1] Novel colchicine derivatives possessing various substituents at the C4 position were prepared. Among them, 4-halo derivatives 3-6 were found to exhibit higher activity against cancer cell lines (A549, HT29, HCT116) as well as on mice transplanted with the HCT116 human colorectal carcinoma cell line than colchicine (1). Further, utilizing the 4-substituted colchicines, we prepared pro-drugs having a dipeptide side chain and demonstrated that these pro-drugs were activated by cathepsin B, an enzyme overexpressed in tumor cells, and exhibited selective toxicity to the tumor cells.[2] |
Solubility Data
| Solubility (In Vitro) | DMSO: 160 mg/mL (374.31 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3395 mL | 11.6973 mL | 23.3945 mL | |
| 5 mM | 0.4679 mL | 2.3395 mL | 4.6789 mL | |
| 10 mM | 0.2339 mL | 1.1697 mL | 2.3395 mL |