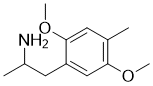
2,5-Dimethoxy-4-Methylamphetamine is a novel and potent psychedelic drug
Physicochemical Properties
| Molecular Formula | C12H19NO2 |
| Molecular Weight | 209.28476 |
| Exact Mass | 209.141 |
| CAS # | 15588-95-1 |
| Related CAS # | 15589-00-1 (hydrochloride) |
| PubChem CID | 85875 |
| Appearance | Typically exists as solid at room temperature |
| Melting Point | 60.5 to 60.1 °C |
| LogP | 2.2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 15 |
| Complexity | 187 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | NTJQREUGJKIARY-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C12H19NO2/c1-8-5-12(15-4)10(6-9(2)13)7-11(8)14-3/h5,7,9H,6,13H2,1-4H3 |
| Chemical Name | 1-(2,5-dimethoxy-4-methylphenyl)propan-2-amine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion DOM produces effects within 1 to 2 hours of ingestion of doses ranging from 2 to 14 mg. Twenty percent of an ingested dose appears unchanged in the urine within 24 hours. Metabolism / Metabolites In an effort to more fully characterize the metabolic fate of the psychotomimetic amine (1-(2, 5-dimethoxy-4-methylpheny)-2-aminopropane (DOM, STP), the formation of the two possible monophenols and the p-hydroquinone metabolites of the parent drug by 10,000 X g supernatent fractions of rabbit liver homogenates has been investigated. All three metabolites have been fully characterized and quantitatively estimated by chemical ionization mass spectrometry with the aid of deuterium enriched compounds. The stereochemical course of the mono-O-demethylation reactions has been shown to proceed with enantiomeric enrichment of the S-aminophenols. The mechanistic implications concerning the mode of action of the parent drug and metabolic O-demethylation are discussed. The possible relationship between metabolism and psychotomimetic activity among the methoxylated 1-phenyl-2-aminopropanes led to our investigation of the in vitro O-demethylation of 1-(2,5-dimethoxy-4-methylphenyl)-2-aminopropane (1, DOM, STP). Employing a sensitive and highly selective stable isotope dilution assay, we observed that rabbit liver homogenates biotransform the amine 1 to its 2-O-demethyl, 5-O-demethyl, and bis (O-demethyl) metabolite metabolites. Both monophenolic metabolites are enriched in their S enantiomers. The bis(O-demethyl) metabolite has structural, chemical, and electrochemical similarities to the sympatholytic agent "6-hydroxydopamine". The possible significance of metabolic O-demethylation in terms of the psychotomimetic properties of amine 1 is discussed. Metabolites of STP have been synthesized and identified in rat urine and feces. A 4-hydroxymethyl compound which resulted from hydroxylation of the 4-methyl group of STP accounted for half of the total amount of the metabolites isolated in the 24 hour urine. The further oxidation product of the 4-hydroxymethyl compound, the 4-carboxy metabolite and unchanged STP were present in a ratio of 2 to 7. A trace amount of the ketone, derived from oxidation of the side-chain of STP, was also detected. The feces at the same time interval contained almost exclusively the 4-carboxy metabolite, with the 4-hydroxymethyl metabolite and a relatively small quantity of unchanged STP amounting to less than 7%. In the present study /investigators/ attempted to isolate metabolites of STP in monkey and rat brains, as well as in regional areas of the monkey brain for any possible correlation between metabolism and and the behavioral effects. The designer drug 2,5-dimethoxy-4-methyl-amphetamine (DOM, STP) is known to be extensively metabolized in various species. The current study showed that cytochrome P450 2D6 was the only isoenzyme involved in formation of the main metabolite hydroxy DOM. In addition, the authors' systematic toxicological analysis (STA) procedure using full-scan GC-MS was suitable to prove an intake of a common drug users' dose of DOM by detection of hydroxy DOM in rat urine. Assuming similar metabolism, the described STA procedure should be suitable for proof of an intake of DOM in human urine. However, DOM and/or other metabolites such as deamino-oxo-hydroxy DOM might be the target analyte in urine of CYP2D6 poor metabolizers. |
| Toxicity/Toxicokinetics |
Non-Human Toxicity Values LD50 Mouse ip 89 + or -4.2 mg/kg /hydrochloride/ LD50 Mouse ip 94 mg/kg LD50 Rabbit oral 200 mg/kg |
| Additional Infomation |
A psychedelic phenyl isopropylamine derivative, commonly called DOM, whose mood-altering effects and mechanism of action may be similar to those of LSD. A psychedelic phenyl isopropylamine derivative, commonly called DOM, whose mood-altering effects and mechanism of action may be similar to those of LSD. Mechanism of Action Alterations in brain serotonergic function have been implicated in the mechanism of action of LSD, mescaline, and other similarly acting hallucinogenic drugs of abuse such as STP (2,5-dimethoxyphenylisopropylamine; DOM). In order to test the hypothesis that the mechanism of action of LSD and phenylisopropylamine hallucinogens is through stimulation of a specific brain serotonin receptor sub-type, the affinities of these compounds for radiolabelled 5-HT2, 5-HT1A, 5-HT1B, and 5-HT1C receptors have been determined using recently developed in vitro radioligand binding methodologies. The 5-HT2 receptor was labelled with the agonist/hallucinogen radioligand 3H-DOB (4-bromo-2,5-dimethoxyphenylisopropylamine). The 5-HT1A, 5-HT1B, and 5-HT1C receptors were labelled with 3H-OH-DPAT, 3H-5-HT, and 3H-mesulergine, respectively. In general, the phenylisopropylamines displayed 10-100 fold higher affinities for the 5-HT2 receptor than for the 5-HT1C receptor and 100-1000 fold higher affinities for the 5-HT2 receptor than for the 5-HT1A or 5-HT1B receptor. There was a strong correlation between hallucinogenic potencies and 5-HT2 receptor affinities of the phenylisopropylamines (r = 0.90); the correlation coefficients for the 5-HT1A, 5-HT1B, and 5-HT1C were 0.73, 0.85, and 0.78, respectively. Because there is no evidence that 5-HT1A-selective or 5-HT1B-selective agonists are hallucinogenic and because the phenylisopropylamines are potent hallucinogens, a 5-HT2 receptor interaction is implicated and supports previous suggestions to this effect. A secondary role for 5-HT1C receptors cannot be discounted at this time. 2,5-Dimethoxy-4-methylamphetamine (DOM, "STP") is a potent hallucinogen, proposed to be a serotonin receptor agonist. Its effects have not previously been tested upon central neurons where serotonin is excitatory and serotonin antagonists are effective. Extracellular single unit recordings were obtained from facial motoneurons in anesthetized rats, and drugs were applied from five-barrelled micropipettes by iontophoresis. Facial motoneurons were commonly silent. During subthreshold application of glutamate, firing could be induced by dopamine and DOM. As reported by others, serotonin and noradrenaline also excited facial motoneurons under these conditions. Methysergide antagonized responses to serotonin and DOM but not those to noradrenaline; methysergide could not usually discriminate between responses to serotonin and dopamine. Ketanserin reversibly antagonized (but could not discriminate between) responses to serotonin, dopamine, and noradrenaline. Chlorpromazine antagonized responses to dopamine at doses that did not alter serotonin-induced excitation, and responses to DOM were not reduced by doses of chlorpromazine, that had no local anaesthetic effect on action potentials elicited by DOM and serotonin. These results suggest that DOM is an agonist on at least one type of central serotonin receptor. This receptor may also be a ketanserin (5-HT2) binding site. ... In perfused dog forelimb DOM produced a vasodilation followed by a vasoconstriction. The vasoconstriction produced by DOM was not blocked by phentolamine but was antagonized by methysergide. In isolated superfused vascular strips of dog dorsal metatarsal vein, DOM elicited a muscle contraction which was not blocked by phentolamine or cocaine but was antagonized by cinanserin. It was concluded that DOM had a direct stimulatory effect on serotonin receptors. The vasodilation in the perfused forelimb of the dog induced by DOM was not antagonized by propranolol, atropine, pyrilamine or bulbocapnine. ... |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.7783 mL | 23.8914 mL | 47.7829 mL | |
| 5 mM | 0.9557 mL | 4.7783 mL | 9.5566 mL | |
| 10 mM | 0.4778 mL | 2.3891 mL | 4.7783 mL |