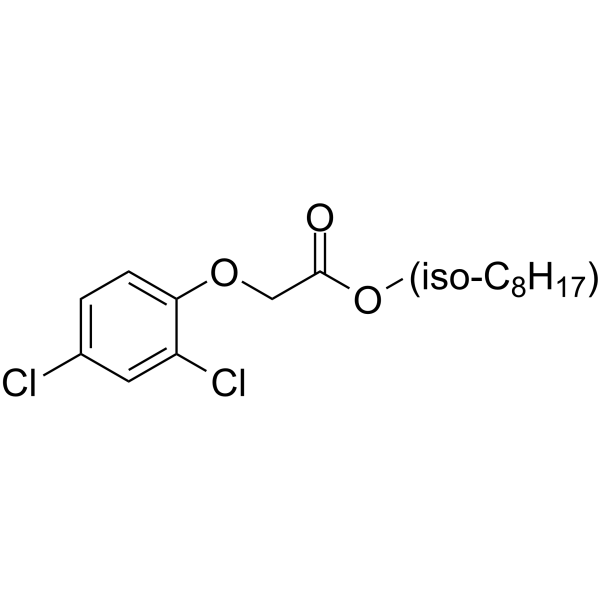
Physicochemical Properties
| Molecular Formula | C16H22CL2O3 |
| Molecular Weight | 333.25 |
| Exact Mass | 332.095 |
| CAS # | 25168-26-7 |
| PubChem CID | 24678 |
| Appearance | Colorless to light yellow liquid |
| Density | 1.15 g/cm3 |
| Boiling Point | 396.9ºC at 760 mmHg |
| Melting Point | 12ºC |
| Flash Point | 48ºC |
| Vapour Pressure | 1.65E-06mmHg at 25°C |
| Index of Refraction | 1.506 |
| LogP | 5.131 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 10 |
| Heavy Atom Count | 21 |
| Complexity | 297 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CCCCC(COC(COC1=CC=C(Cl)C=C1Cl)=O)CC |
| InChi Key | BBPLSOGERZQYQC-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C16H22Cl2O3/c1-12(2)6-4-3-5-9-20-16(19)11-21-15-8-7-13(17)10-14(15)18/h7-8,10,12H,3-6,9,11H2,1-2H3 |
| Chemical Name | 6-methylheptyl 2-(2,4-dichlorophenoxy)acetate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion ... ISOOCTYL ESTER OF 2,4-D DISAPPEARED FROM BODIES OF INJECTED MICE MORE RAPIDLY THAN 2,4-D ACID. Leaves absorb nonpolar (ester) forms most readily. ... The esters of 2,4-D tend to resist washing from plants and are rapidly converted to the acid by the plants. ... Following foliar absorption, 2,4-D translocates within the phloem, probably moving with food material. Following root absorption, it may move upward in the transpiration stream. Translocation is influenced by the growth status of the plant. Accumulation of the herbicide occurs principally at the meristematic regions of shoots and roots. /2,4-D or its esters/ Plasma 2,4-D concn did not exceed 0.2 mg/l in workers exposed to 2,4-D ester at an atmospheric concn of 0.1 to 0.2 mg/l; no accumulation was noted during the work week. ... Workers ... developed 2,4-D urine concentrations of 3 to 14 mg/l after a day of exposure. /2,4-Dichlorophenoxyacetic acid or its esters/ AFTER SC INJECTION OF 2,4-D & ITS BUTYL & ISOOCTYL ESTERS INTO MICE @ 100 MG/KG, ESTERS WERE ELIM RAPIDLY, & ONLY 5-10% OF THE 2,4-D REMAINED AFTER 1 DAY. ... 2,4-D WAS ELIM IN MILK OF COWS MAINTAINED IN PASTURES TREATED WITH 2,4-D OR ITS BUTYL OR ISOOCTYL ESTER. For more Absorption, Distribution and Excretion (Complete) data for 2,4-D ISOOCTYL ESTERS (7 total), please visit the HSDB record page. Metabolism / Metabolites Plants hydrolyze 2,4-D esters to 2,4-D, which is the active herbicide. ... Further metabolism ... occurs through three mechanisms, namely, side chain degradation, hydroxylation of the aromatic ring, and conjugation with plant constituents. /2,4-D esters/ HERBICIDAL ACTIVITY OF ESTERS, NITRILES, AMINES (&, OF COURSE, SALTS) APPEARS SIMILAR IF NOT IDENTICAL TO PARENT ACID. THIS IS APPARENTLY DUE TO PRESENCE OF HYDROLYTIC ENZYMES IN PLANTS & IN SOIL MICROORGANISMS THAT CONVERT THESE DERIVATIVES TO PARENT ACID. /SRP: 2,4-D OR ITS ESTERS/ 2,4-D ESTERS ARE HYDROLYZED IN ANIMALS. THE PHENOXY ACIDS ARE EXCRETED PREDOMINANTLY AS SUCH IN THE URINE OF RATS AFTER THEIR ORAL ADMIN, ALTHOUGH MINOR PORTION IS CONJUGATED WITH AMINO ACIDS GLYCINE & TAURINE & WITH GLUCURONIC ACID. /2,4-D AND ESTERS/ Metabolites of 2,4-D other than conjugates have not been detected in human urine. Biological Half-Life THESE HERBICIDES DO NOT ACCUM IN ANIMALS. THEY ARE NOT EXTENSIVELY METAB BUT ARE ACTIVELY EXCRETED INTO THE URINE ... THEIR PLASMA HALF-LIFE IN MAN IS ABOUT 1 DAY. /CHLOROPHENOXY COMPOUNDS/ The hydrolysis half-life of the n-butyl ester of 2,4-D was determined to be 100 hours in neutral water and much less in aqueous soil suspensions. Smith (1976) reported that the n-butyl ester of 2,4-D undergoes almost complete hydrolysis to 2,4-D in less than 24 hours in moist soil. Similar results were reported for the hydrolysis of the n-butyl and isooctyl esters of 2,4,5-T to 2,4,5-T. Esters of 2,4-D were completely hydrolyzed to 2,4-D within 9 days in lake water. Therefore, rates of biological hydrolysis appear to be greater than rates of chemical hydrolysis. |
| Toxicity/Toxicokinetics |
Interactions Cutthroat trout (Salmo clarki) were treated with six different paired mixtures of dicamba, picloram, 2,4-D butyl ester, 2,4-D isooctyl ester, and 2,4-D propylene glycol butyl ether ester. Except for 2,4-D isooctyl ester, the LC50's resulting from mixtures of 2,4-D esters and picloram were lower than LC50's of those herbicides tested individually. Dicamba and 2,4-D isooctyl ester were the least toxic individually and mixtures of dicamba or 2,4-D isooctyl ester with the other herbicides tested did not result in increased toxicity. Results reflect the importance of using combination exposures in determining the biological significance of the simultaneous occurrence of more than one herbicide in surface waters. Non-Human Toxicity Values LD50 Rabbit skin absorption > 4000 mg/kg LD50 Rat oral 982 mg/kg |
| References |
[1]. Preparation and characterization of controlled-release poly(melamine-formaldehyde) microcapsules filled with 2,4-D isooctyl ester, International Journal of Polymeric Materials and Polymeric Biomaterials, 66:18, 963-969. |
| Additional Infomation |
Ded-weed lv-69 67 percent 2,4-d isooctyl ester appears as white crystals in commerce, a brown or dark brown liquid. (NTP, 1992) Mechanism of Action ... /CHLOROPHENOXY CMPD INCL 2,4-D ESTERS/ EXERT THEIR HERBICIDAL ACTION BY ACTING AS GROWTH HORMONES IN PLANTS. /CHLOROPHENOXY COMPOUNDS/ |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0008 mL | 15.0038 mL | 30.0075 mL | |
| 5 mM | 0.6002 mL | 3.0008 mL | 6.0015 mL | |
| 10 mM | 0.3001 mL | 1.5004 mL | 3.0008 mL |