Physicochemical Properties
| Molecular Formula | C20H30O4 |
| Molecular Weight | 334.4498 |
| Exact Mass | 334.214 |
| CAS # | 4176-97-0 |
| PubChem CID | 11624161 |
| Appearance | White to off-white solid powder |
| Density | 1.15g/cm3 |
| Boiling Point | 509.5ºC at 760 mmHg |
| Melting Point | 176-178ºC |
| Flash Point | 177.3ºC |
| Index of Refraction | 1.551 |
| LogP | 2.991 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 24 |
| Complexity | 566 |
| Defined Atom Stereocenter Count | 5 |
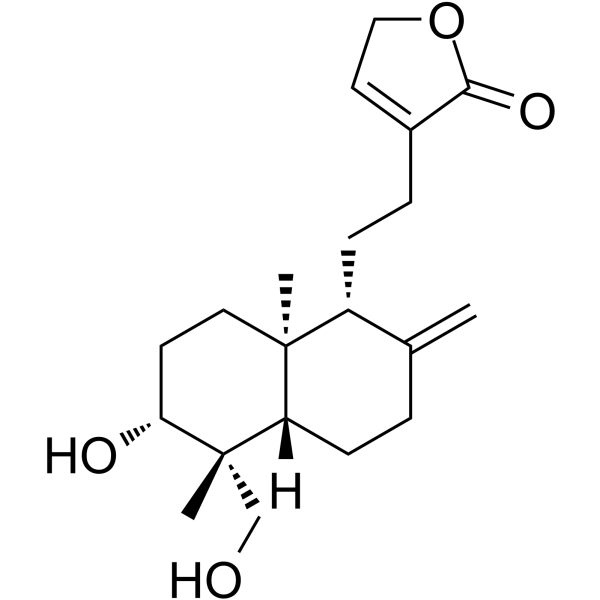
| SMILES | C[C@@]12CC[C@H]([C@@]([C@H]1CCC(=C)[C@H]2CCC3=CCOC3=O)(C)CO)O |
| InChi Key | GVRNTWSGBWPJGS-YSDSKTICSA-N |
| InChi Code | InChI=1S/C20H30O4/c1-13-4-7-16-19(2,10-8-17(22)20(16,3)12-21)15(13)6-5-14-9-11-24-18(14)23/h9,15-17,21-22H,1,4-8,10-12H2,2-3H3/t15-,16+,17-,19+,20+/m1/s1 |
| Chemical Name | 4-[2-[(1R,4aS,5R,6R,8aS)-6-hydroxy-5-(hydroxymethyl)-5,8a-dimethyl-2-methylidene-3,4,4a,6,7,8-hexahydro-1H-naphthalen-1-yl]ethyl]-2H-furan-5-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- 14-Deoxyandrographolide targets calcium-mediated signaling pathways in rat uterine smooth muscle.[1] - 14-Deoxyandrographolide targets tumor necrosis factor receptor superfamily member 1A (TNFR1A) in hepatocytes, acting via the NO/cGMP pathway [2] |
| ln Vitro |
- Inhibition of rat uterine smooth muscle contractility: Isolated rat uterine smooth muscle strips were treated with 14-Deoxyandrographolide at 10, 30, and 100 μM. At 100 μM, it inhibited KCl (60 mM)-induced contractions by 62% and oxytocin (10 nM)-induced contractions by 58%, compared to the control group. The inhibition was concentration-dependent, with no significant effect at 10 μM. Mechanistically, it reduced calcium influx into smooth muscle cells, as evidenced by decreased intracellular calcium levels (measured via fluorescent calcium indicator)[1] - Desensitization of hepatocytes to TNF-α-induced apoptosis: Primary rat hepatocytes were pretreated with 14-Deoxyandrographolide (10, 30 μM) for 1 hour, then exposed to TNF-α (10 ng/mL) for 24 hours. The 30 μM dose reduced apoptosis rate by 45% (assessed via Hoechst 33258 staining) compared to the TNF-α-only group. It also increased the release of TNFR1A into the culture supernatant by 35% (30 μM) and elevated intracellular NO levels by 40% (30 μM) and cGMP levels by 38% (30 μM). The effect was reversed by a NO synthase inhibitor (L-NAME), confirming dependence on the NO/cGMP pathway[2] |
| Enzyme Assay |
- NO concentration assay in hepatocytes: Primary rat hepatocytes were incubated with 14-Deoxyandrographolide (10, 30 μM) for 1 hour. The culture supernatant was collected, and NO concentration was measured using the nitrate reductase method: supernatant was mixed with nitrate reductase and cofactors, incubated at 37°C for 30 minutes, then reacted with Griess reagent. Absorbance was read at 540 nm, and NO concentration was calculated from a standard curve[2] |
| Cell Assay |
- Hepatocyte apoptosis assay: Primary rat hepatocytes were seeded in 6-well plates at 5×10⁵ cells/well and cultured for 24 hours. Cells were pretreated with 14-Deoxyandrographolide (10, 30 μM) for 1 hour, then treated with TNF-α (10 ng/mL) for 24 hours. Apoptosis was detected by two methods: 1) Hoechst 33258 staining (counting apoptotic nuclei under fluorescence microscope, apoptosis rate = apoptotic nuclei/total nuclei × 100%); 2) caspase-3 activity assay (lysing cells, incubating with caspase-3 substrate, measuring fluorescence at 460 nm after excitation at 380 nm). The 30 μM dose reduced caspase-3 activity by 42% compared to the TNF-α-only group[2] |
| Animal Protocol |
- Rat uterine smooth muscle preparation: Female Sprague-Dawley rats (200–220 g) in estrus were sacrificed by cervical dislocation. The uterus was excised, cleaned of connective tissue, and cut into 2–3 mm longitudinal strips. Each strip was mounted in a 5 mL organ bath containing Krebs-Ringer bicarbonate solution (maintained at 37°C, gassed with 95% O₂ and 5% CO₂) and connected to a force transducer to record contractile tension. After a 30-minute equilibration period, KCl (60 mM) or oxytocin (10 nM) was added to induce contractions. Once contractions stabilized, 14-Deoxyandrographolide (dissolved in DMSO, diluted in Krebs solution to final concentrations of 10, 30, 100 μM) was added sequentially, and contractile changes were recorded for 20 minutes per concentration[1] |
| References |
[1]. Effect of 14-deoxyandrographolide on calcium-mediated rat uterine smooth muscle contractility. Phytother Res. 2003;17(9):1011-1015. [2]. 14-Deoxyandrographolide desensitizes hepatocytes to tumour necrosis factor-alpha-induced apoptosis through calcium-dependent tumour necrosis factor receptor superfamily member 1A release via the NO/cGMP pathway. Br J Pharmacol. 2010 Aug;160. |
| Additional Infomation |
14-Deoxyandrographolide is a diterpene lactone. 14-Deoxyandrographolide has been reported in Andrographis paniculata and Taiwanofungus camphoratus with data available. - The inhibitory effect of 14-Deoxyandrographolide on uterine smooth muscle contractility is mediated by reducing extracellular calcium influx, rather than blocking intracellular calcium release[1] - 14-Deoxyandrographolide desensitizes hepatocytes to TNF-α-induced apoptosis by promoting TNFR1A release (reducing cell surface TNFR1A available for TNF-α binding), and this process requires activation of the NO/cGMP pathway[2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~299.00 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.47 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.47 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9900 mL | 14.9499 mL | 29.8998 mL | |
| 5 mM | 0.5980 mL | 2.9900 mL | 5.9800 mL | |
| 10 mM | 0.2990 mL | 1.4950 mL | 2.9900 mL |