Physicochemical Properties
| Molecular Formula | C11H14O2 |
| Molecular Weight | 178.22766 |
| Exact Mass | 178.099 |
| CAS # | 6379-72-2 |
| PubChem CID | 637776 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.0±0.1 g/cm3 |
| Boiling Point | 271.1±20.0 °C at 760 mmHg |
| Flash Point | 104.5±21.3 °C |
| Vapour Pressure | 0.0±0.5 mmHg at 25°C |
| Index of Refraction | 1.534 |
| LogP | 3.05 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 13 |
| Complexity | 166 |
| Defined Atom Stereocenter Count | 0 |
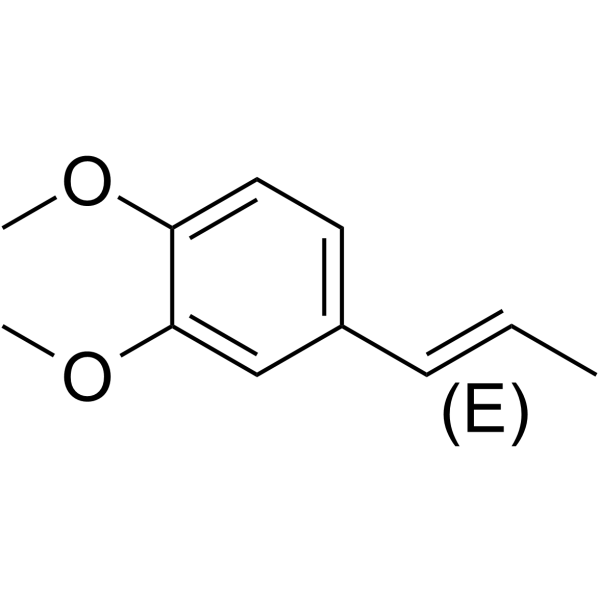
| SMILES | C/C=C/C1=CC=C(OC)C(OC)=C1 |
| InChi Key | NNWHUJCUHAELCL-SNAWJCMRSA-N |
| InChi Code | InChI=1S/C11H14O2/c1-4-5-9-6-7-10(12-2)11(8-9)13-3/h4-8H,1-3H3/b5-4+ |
| Chemical Name | 1,2-dimethoxy-4-[(E)-prop-1-enyl]benzene |
| Synonyms | Methylisoeugenol; 93-16-3; trans-Methylisoeugenol; Methyl isoeugenol; ...; 6379-72-2; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Natural product from Acorus calamus L; insect chemosterilant |
| ln Vitro | The molecular structure of the effective compound from oil of A. calamus is given in Fig. 2 I. The position of different groups in the aromatic ring is known to play an important part in various activities, as shown by some allelochemics 1 2 , where the position of the hydroxyl groups is responsible for activities such as antiassimilation and reduction of survival or pupation. Similarly in our study the position of the O-methyl groups on the ring, and of the double bond in the side-chain of fJ-asarone, plays an important part in the antigonadal activity (Table I, Fig. 2). After testing the various compounds it was found that the O-methyl group at position R in the ring (Fig. 2, V) is important for the resorption of the oocytes, which is further supported by the suppression in the ovary due to the O-methyl group at R2 (as in Eugenol, VI and methyl isoeugenol, III---Fig. 2). The suppression in the ovary due to the O-methyl group at R2 seems true of precocene 112 also because the removal of the O-methyl group from R2 position in that molecule also reduces activity (precocene I of Bowers2). Comparison of the activities of compounds I and II (Table 1) suggests that the cis configuration of the side-chain double bond in fJ-asarone is important for antigonadal activity. It is interesting, however, that III which has a trans configuration, but lacks the O-methyl group at position R also has significant suppression activity. [1] |
| References |
[1]. A new insect chemosterilant isolated from Acorus calamus L. |
| Additional Infomation |
Isomethyleugenol is a phenylpropanoid, an olefinic compound and a dimethoxybenzene. Methylisoeugenol has been reported in Perilla frutescens, Punica granatum, and other organisms with data available. PLANT extracts have been tested for insecticide and juvenile hormone activity and some have been found with anti-juvenile hormone activity. We have suggested that an essential oil of Acorus calamus L. inhibits interstitial cell activity; this would represent a new concept in insect chemosterilisation. We report here the identification of the component of the oil that is responsible for that inhibition; we have also established the importance of substitute groups and a side chain by testing various allyl benzene analogues. [1] |
Solubility Data
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.6107 mL | 28.0536 mL | 56.1073 mL | |
| 5 mM | 1.1221 mL | 5.6107 mL | 11.2215 mL | |
| 10 mM | 0.5611 mL | 2.8054 mL | 5.6107 mL |