Physicochemical Properties
| Molecular Formula | C6H6O6 |
| Molecular Weight | 174.1082 |
| Exact Mass | 174.016 |
| CAS # | 4023-65-8 |
| Related CAS # | (Z)-Aconitic acid-13C6;2456413-64-0 |
| PubChem CID | 444212 |
| Appearance | White to off-white solid powder |
| Density | 1.7±0.1 g/cm3 |
| Boiling Point | 542.6±50.0 °C at 760 mmHg |
| Melting Point | 190 °C (dec.)(lit.) |
| Flash Point | 296.0±26.6 °C |
| Vapour Pressure | 0.0±3.1 mmHg at 25°C |
| Index of Refraction | 1.571 |
| LogP | 0.63 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 12 |
| Complexity | 251 |
| Defined Atom Stereocenter Count | 0 |
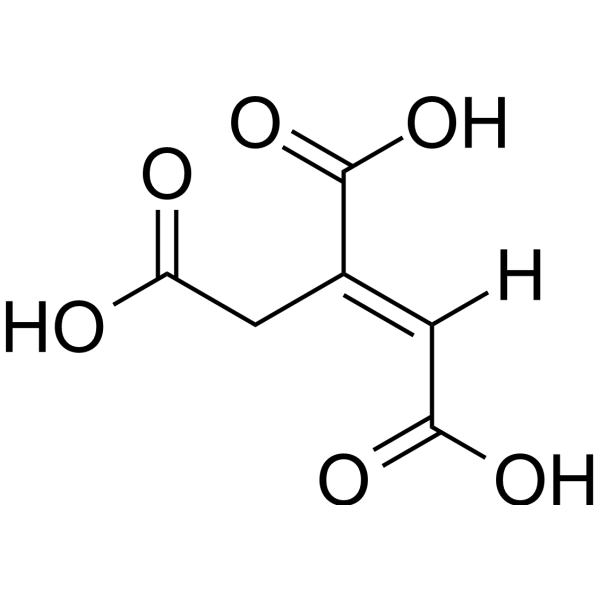
| SMILES | C(/C(=C\C(=O)O)/C(=O)O)C(=O)O |
| InChi Key | GTZCVFVGUGFEME-HNQUOIGGSA-N |
| InChi Code | InChI=1S/C6H6O6/c7-4(8)1-3(6(11)12)2-5(9)10/h1H,2H2,(H,7,8)(H,9,10)(H,11,12)/b3-1+ |
| Chemical Name | (E)-prop-1-ene-1,2,3-tricarboxylic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| References |
[1]. Quantitation of trans-aconitic acid in different stages of the sugar-manufacturing process. J Agric Food Chem. 2014 Aug 20;62(33):8314-8. |
| Additional Infomation |
Trans-aconitic acid is the trans-isomer of aconitic acid. It has a role as a fundamental metabolite. It is a conjugate acid of a trans-aconitate(3-). trans-Aconitic acid is a metabolite found in or produced by Escherichia coli (strain K12, MG1655). trans-Aconitic acid has been reported in Echinochloa phyllopogon, Populus tremula, and other organisms with data available. trans-Aconitic acid is a metabolite found in or produced by Saccharomyces cerevisiae. A tricarboxylic acid with the formula (COOH)-CH2-C(COOH)=CH-COOH. 1. Background of trans-Aconitic acid: trans-Aconitic acid is a natural organic acid present in sugarcane. It is a by-product of the tricarboxylic acid (TCA) cycle in plants and can affect the quality of sugar products (such as color and crystallization) during sugar manufacturing. The study aimed to establish a reliable method for quantifying trans-Aconitic acid to monitor its content changes across different sugar-manufacturing stages [1] 2. Quantitative analysis procedure of trans-Aconitic acid in sugar samples: a. Sample pretreatment: Sugar samples (including sugarcane juice, clarified juice, evaporator syrup, and molasses) were diluted with ultrapure water to an appropriate concentration. Then, 5 mL of the diluted sample was mixed with 1 mL of 0.1 M phosphoric acid (pH 2.5) and vortexed for 1 minute. The mixture was centrifuged at 10,000 × g for 15 minutes at 4°C, and the supernatant was filtered through a 0.22 μm hydrophilic polyethersulfone (PES) membrane to remove impurities [1] b. Chromatographic conditions: High-performance liquid chromatography (HPLC) was used with a C18 reversed-phase column (250 mm × 4.6 mm, 5 μm particle size). The mobile phase was a mixture of 0.1 M phosphoric acid (pH 2.5) and acetonitrile (98:2, v/v) with a flow rate of 1.0 mL/min. The column temperature was maintained at 30°C, and the detection wavelength was set to 210 nm. The injection volume was 20 μL [1] c. Method validation: A standard curve of trans-Aconitic acid was constructed with concentrations ranging from 5 μg/mL to 200 μg/mL, showing a linear correlation coefficient (R²) of 0.9998. The limit of detection (LOD) was 1.2 μg/mL, and the limit of quantification (LOQ) was 3.8 μg/mL. The recovery rate of trans-Aconitic acid in spiked sugar samples ranged from 92.3% to 97.8%, with a relative standard deviation (RSD) of 1.5%–3.2%, indicating good accuracy and precision [1] 3. Content distribution in sugar-manufacturing stages: The study detected trans-Aconitic acid content in different stages: sugarcane juice (12.5–18.3 μg/mL), clarified juice (10.2–15.7 μg/mL), evaporator syrup (85.6–102.4 μg/mL), and molasses (215.3–256.7 μg/mL). The content increased significantly during the evaporation and concentration stages, which was attributed to the reduction of water volume and the stability of trans-Aconitic acid under high-temperature conditions [1] |
Solubility Data
| Solubility (In Vitro) | H2O : ~100 mg/mL (~574.35 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 50 mg/mL (287.17 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.7435 mL | 28.7175 mL | 57.4350 mL | |
| 5 mM | 1.1487 mL | 5.7435 mL | 11.4870 mL | |
| 10 mM | 0.5743 mL | 2.8717 mL | 5.7435 mL |