Physicochemical Properties
| Molecular Formula | C24H33N5O8S |
| Molecular Weight | 551.61 |
| Exact Mass | 551.205 |
| CAS # | 127985-74-4 |
| PubChem CID | 10123265 |
| Appearance | White to off-white solid powder |
| LogP | -7.2 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 14 |
| Rotatable Bond Count | 11 |
| Heavy Atom Count | 38 |
| Complexity | 863 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | UDOPJKHABYSVIX-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C24H33N5O8S/c30-21(31)13-26-5-6-27(14-22(32)33)9-10-29(16-24(36)37)20(12-28(8-7-26)15-23(34)35)11-18-1-3-19(4-2-18)25-17-38/h1-4,20H,5-16H2,(H,30,31)(H,32,33)(H,34,35)(H,36,37) |
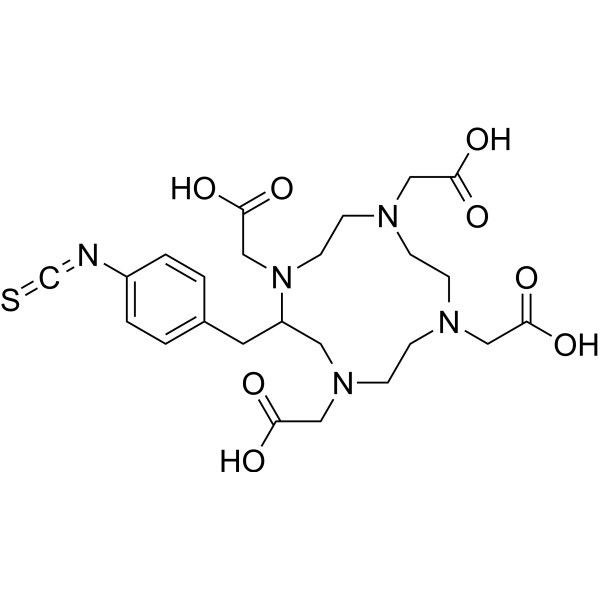
| Chemical Name | 2-[4,7,10-tris(carboxymethyl)-6-[(4-isothiocyanatophenyl)methyl]-1,4,7,10-tetrazacyclododec-1-yl]acetic acid |
| Synonyms | 127985-74-4; p-SCN-Bn-DOTA; (p-SCN-Bn)-dota; p-SCN-Bz-dota; 1,4,7,10-Tetraazacyclododecane-1,4,7,10-tetraaceticacid, 2-[(4-isothiocyanatophenyl)methyl]-; UNII-13KT123BYW; 13KT123BYW; 2-[4,7,10-tris(carboxymethyl)-6-[(4-isothiocyanatophenyl)methyl]-1,4,7,10-tetrazacyclododec-1-yl]acetic acid; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Chelating agent |
| ln Vitro |
Five distinct metal ions are complexed with p-SCN-Bn-DOTA: In(III), Y(III), Lu(III), Gd(III), and Cu(II)[2]. Nimotuzumab conjugates were obtained with high purity. Radiolabeling yield and specific activities ranged from 63.6% to 94.5% and from 748 to 1142 MBq/mg, respectively. The stability in DTPA excess and human serum was 95.9% and 93.2% after 10 days, respectively. The radioimmunoconjugate showed specific receptor binding in tumor cell lines. [1] |
| ln Vivo | Biodistribution in healthy animals showed the typical behavior of the immunoconjugates based on monoclonal antibodies. The study in xenografts mice demonstrated uptake of (177)Lu-Nimotuzumab in the tumor and reticuloendothelial organs. Conclusions: (177)Lu-Nimotuzumab was obtained with high purity and specific activities under optimal conditions without significant loss in immunoreactivity and might be a potential radioimmunoconjugate for radioimmunotherapy of tumors with epidermal growth factor receptor overexpression. [1] |
| Enzyme Assay |
Nimotuzumab was conjugated with S-2-(4-isothiocyanatobenzyl)-1,4,7,10-tetraazacyclododecane tetraacetic acid (p-SCN-Bn-DOTA), testing different molar ratios. The immunoconjugates were characterized. The radiolabeling with (177)Lu was optimized. Radioimmunoconjugates stability was tested in 2-[bis[2-[bis(carboxymethyl)amino]ethyl]amino]acetic acid (DTPA) excess and human serum. In vitro studies were performed in tumor model cell lines. Receptor-specific binding was tested by competitive inhibition.[1] Tenatumomab is an anti-tenascin murine monoclonal antibody previously used in clinical trials for delivering radionuclides to tumors by both pre-targeting (biotinylated Tenatumomab within PAGRIT) and direct 131Iodine labeling approaches. Here we present the synthesis and in vitro characterization of three Tenatumomab conjugates to bifunctional chelating agents (NHS-DOTA, NCS-DOTA and NCS-DTPA). Results indicate ST8198AA1 (Tenatumomab-DOTAMA, derived by conjugation of NHS-DOTA), as the most promising candidate in terms of conjugation rate and yield, stability, antigen immunoreactivity and affinity. Labeling efficiency of the different chelators was investigated with a panel of cold metals indicating DOTAMA as the best chelator. Labeling of Tenatumomab-DOTAMA was then optimized with several metals and stability performed confirms suitability of this conjugate for further development. ST8198AA1 represents an improvement of the previous antibody forms because the labeling with radionuclides like 177Lu or 64Cu would allow theranostic applications in patients bearing tenascin expressing tumors. [2] |
| Animal Protocol | (177)Lu-Nimotuzumab in vivo studies were conducted in healthy and xenograft animals. |
| References |
[1]. Preclinical evaluation of (177)lu-nimotuzumab: a potential tool for radioimmunotherapy of epidermal growth factor receptor-overexpressing tumors. Cancer Biother Radiopharm. 2011 Jun;26(3):287-97. [2]. Synthesis and preliminary in vitro evaluation of DOTA-Tenatumomab conjugates for theranostic applications in tenascin expressing tumors. Bioorg Med Chem. 2019 Aug 1;27(15):3248-3253. |
| Additional Infomation | The humanized monoclonal antibody Nimotuzumab (h-R3) has demonstrated an exceptional and better clinical profile than other monoclonal antibodies for immunotherapy of epidermal growth factor receptor-overexpressing tumors. This work deals with the preparation and radiolabeling optimization of (177)Lu-Nimotuzumab and their preclinical evaluation.[1] |
Solubility Data
| Solubility (In Vitro) | DMSO: 20.83 mg/mL (37.76 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (3.77 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (3.77 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (3.77 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8129 mL | 9.0644 mL | 18.1288 mL | |
| 5 mM | 0.3626 mL | 1.8129 mL | 3.6258 mL | |
| 10 mM | 0.1813 mL | 0.9064 mL | 1.8129 mL |