Physicochemical Properties
| Molecular Formula | C20H18BF2N3S |
| Molecular Weight | 381.25 |
| Appearance | Typically exists as solid at room temperature |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Fluorescent probe/dye |
| ln Vitro |
Cell Labeling Protocol (Guideline) Note: This protocol serves as a recommended guideline and should be adapted based on specific experimental requirements[1]. Procedure: 1. Cell Preparation o Seed cells in 20 mm confocal dishes at a density of 5 × 10⁴ cells/mL. o Incubate cells under standard culture conditions. 2. Probe Staining o For confocal imaging, add 5 μM meso-Benzothiazole-BODIPY 505/515 (Probe 1) to the culture medium. o Incubate cells with the probe for 30 minutes. 3. Imaging Parameters o Excitation wavelength: 488 nm o Emission collection range: 520–600 nm |
| Cell Assay |
Immunofluorescence[1] Cell Types: SH-SY5Y cells Tested Concentrations: 5 μM Incubation Duration: 30 min Experimental Results: demonstrated relatively weak fluorescence emissions in low viscos cells, but demonstrated strong fluorescence emissions when the SH-SY5Y cells were preincubated with LPS and nystatin. Cellular Viscosity Imaging and Autophagy Monitoring [1] The confocal imaging was carried out using 20 mm confocal dishes with SH-SY5Y cells of 5 × 104 cells/mL. For the subcellular imaging, 1 (5 μM) and Mito-tracker Blue or Lyso-tracker Blue (1 μM) in Dulbecco’s modified Eagle’s medium (DMEM) were used to culture the cells for 30 min. After the cells were washed with phosphate-buffered saline (PBS) three times, a Leica TCS SP8 confocal microscope was used to conduct subcellular imaging with a 100 × oil-immersion objective lens. For Mito-tracker Blue or Lyso-tracker Blue, the cells were excited at 405 nm and emissions were collected at the range of 425–500 nm, which was labeled as the blue channel. For 1, the cells were excited at 488 nm and emissions were collected at 520–600 nm, which was labeled as the red channel. The merged blue and red channels were also given. For the viscosity-responsive confocal imaging, the cells were first cultured with none, lipopolysaccharide (LPS), or nystatin (both at 20 μM) for 40 min. After washing with PBS three times, the freshly prepared stock solutions of 1 (5 μM in DMEM) were added to the above cell plates, which were further cultured at 37 °C for 30 min. The cells were excited at 488 nm and emissions were collected at 520–600 nm with a 63 × oil-immersion objective lens. For monitoring viscosity changes of 1 in living cells, low temperature and dexamethasone were used. The cells were first treated with 1 (10 μM) at 37 and 4 °C for 30 min, respectively. The other two groups were first incubated with 1 (10 μM) at 37 °C for 30 min, and then treated with dexamethasone (100 μM) or dimethylsulfoxide (DMSO, 10 μL) for another 10 min. After washing three times with PBS, the cells were excited at 488 nm and emissions were collected at 520–600 nm with a 100 × oil-immersion objective lens. For monitoring the autophagy process through the lysosomal viscosity changes, SH-SY5Y cells were incubated with 1 (10 μM) at 37 °C for 30 min and then were cultured in Hank’s balanced salt solution (HBSS), normal medium, or HBSS with the addition of 3-methyladenine (3-MA) (an autophagy inhibitor) to give the starvation conditions, rich-nutrient conditions, or autophagy-inhibited conditions for 2 h, respectively. The cells were excited at 488 nm and emissions were collected at 520–600 nm with a 100 × oil-immersion objective lens. |
| References |
[1]. Novel Meso-Benzothiazole-Substituted BODIPY-Based AIE Fluorescent Rotor for Imaging Lysosomal Viscosity and Monitoring Autophagy. Anal Chem. 2022 Oct 25;94(42):14707-14715. |
| Additional Infomation |
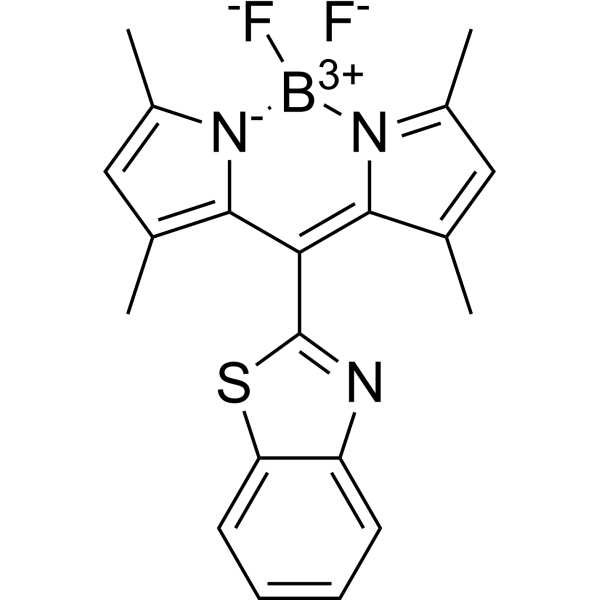
Meso-substituted boron dipyrromethenes (BODIPYs) provide a potential and innovative strategy for the synergistic construction of aggregation-induced emission (AIE) probes and fluorescent rotors for monitoring cellular viscosity changes, which play critical roles in understanding the function of viscosity in its closely associated diseases. Therefore, for the first time, a BODIPY-based fluorescent probe (1) with a rotatable meso-benzothiazole group was rationally designed and synthesized, showing both good viscosity-responsive and AIE properties. Probe 1 through direct linkage with the thiazole group, showed nearly no emission in low viscous solvents; however, a strong emission at 534 nm appeared and increased gradually with the increase in viscosity, attributing to the efficient restriction of the rotatable meso-benzothiazole group. The intensity (log I534) displayed a good linear relationship with viscosity (log η) in the viscous range of 0.59-945 cP in methanol/glycerol mixtures. Interestingly, 1 showed enhanced emission at 534 nm in 70% water compared to pure acetonitrile due to the aggregation-induced inhibited rotations. Cellular imaging suggested that 1 could successfully sense lysosomal viscosity changes induced by lipopolysaccharide, nystatin, low temperature, and dexamethasone in living cells, which could be further applied in autophagy monitoring by tracing viscosity changes. As a comparison, its analogue 2 directly linking with the phenyl group showed no viscosity-responsive or AIE properties. Therefore, for the first time, we reported a meso-benzothiazole-BODIPY-based fluorescent rotor with AIE and lysosomal viscosity-responsive properties in nervous cells, which was further applied in monitoring autophagy, and this work thus could provide an innovative strategy for the design of potential AIE and viscosity-responsive probes.[1] To summarize, for the first time, by incorporating a rotatable and lysosome-targeting benzothiazole group onto the BODIPY core, we herein reported a meso-benzothiazole BODIPY-based AIE fluorescent probe for imaging lysosomal viscosity and monitoring autophagy process in living cells depending on free rotation of the meso-benzothiazole group. Through the introduction of the meso-benzothiazole group with directly linking the thiazole part, the resulting probe 1 showed red-shifted absorption and emissions compared to probe 2 through direct linkage with the phenyl group. Furthermore, what is more interesting, even with slight difference in linkage, probe 1 displayed absolutely different properties compared to probe 2, such as viscosity-responsive and AIE properties. In low viscous environments, probe 1 displayed nearly no fluorescence emissions and when viscosity increased gradually, a strong fluorescence emission band at 534 nm appeared. Interestingly, this meso-benzothiazole-substituted probe showed good AIE at 534 nm, which further confirmed that the rotation of the meso-benzothiazole group could be efficiently restricted in both the aggregated state and high viscous environments. Further cellular experiments suggested that the meso-benzothiazole group could also serve as a lysosomal targeting group, which could be successfully applied in cellular viscosity monitoring by pretreatment with LPS, nystatin, low temperature, and dexamethasone. Moreover, the viscosity-responsive property of probe 1 was further applied in autophagy monitoring. Overall, for the first time, instead of the commonly used meso-phenyl-substitution, we introduced a meso-benzothiazole group onto the BODIPY core through direct linkage with a five-membered-ring, investigated its viscosity/AIE properties, and further extended to visualize its cellular viscosity changes and monitor autophagy process in lysosomes. Other related works based on meso-five-membered heterocyclic ring-substituted BODIPYs are undergoing to optimize their AIE/viscosity properties and investigate their structure–property relationships.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6230 mL | 13.1148 mL | 26.2295 mL | |
| 5 mM | 0.5246 mL | 2.6230 mL | 5.2459 mL | |
| 10 mM | 0.2623 mL | 1.3115 mL | 2.6230 mL |