Physicochemical Properties
| Molecular Formula | C34H43N7O2 |
| Molecular Weight | 581.750927209854 |
| Exact Mass | 581.347 |
| CAS # | 1637443-98-1 |
| Related CAS # | 1637443-98-1 |
| PubChem CID | 131704486 |
| Appearance | White to off-white solid |
| LogP | 4.8 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 13 |
| Heavy Atom Count | 43 |
| Complexity | 813 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | VIBJAHJNWHZSJP-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C34H43N7O2/c1-4-40(5-2)24-25-43-27-16-14-26(15-17-27)35-33-29-11-7-9-13-31(29)37-34(38-33)28-10-6-8-12-30(28)36-32(42)18-19-41-22-20-39(3)21-23-41/h6-17H,4-5,18-25H2,1-3H3,(H,36,42)(H,35,37,38) |
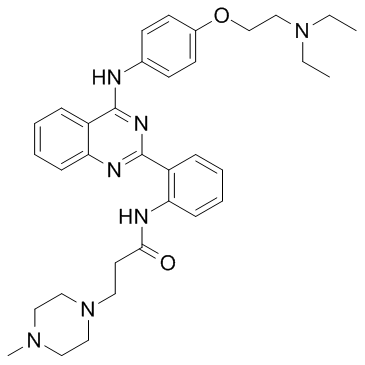
| Chemical Name | N-[2-[4-[4-[2-(diethylamino)ethoxy]anilino]quinazolin-2-yl]phenyl]-3-(4-methylpiperazin-1-yl)propanamide |
| Synonyms | hVEGF-IN-1 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
VEGFR hVEGF-IN-1 (compound 1) (1 nM-100 μM; 5 min) exhibits a binding affinity to both IRES-A (WT) and IRES-A mutant RNA oligomer (IRES-MU1), with Kds of 1.29 and 13.4 μM, respectively, as determined by microscale thermophoresis (MST) measurements[1]. hVEGF-IN-1 (0.375-3 μM; 0-24 h) decreases MDA-MB-231 cell migration by about 25% at the concentration of 3 μM[1]. hVEGF-IN-1 (0.1875-3 μM; 48 h) lowers the amount of VEGF-A protein in MCF-7 cells[1]. hVEGF-IN-1 (0.375-3 μM; 48 h) cuts the migrated MCF-7 cells' relative wound closure by about 35% at a concentration of 3 μM[1]. hVEGF-IN-1 (1.25-10 μM) decreases the stability of the IRES-A G-Quadruplex in a dose-dependent manner[1]. |
| ln Vitro |
hVEGF-IN-1 (compound 1) (1 nM-100 μM; 5 min) exhibits a binding affinity to both IRES-A (WT) and IRES-A mutant RNA oligomer (IRES-MU1), with Kds of 1.29 and 13.4 μM, respectively, as determined by microscale thermophoresis (MST) measurements[1]. hVEGF-IN-1 (0.375-3 μM; 0-24 h) decreases MDA-MB-231 cell migration by about 25% at the concentration of 3 μM[1]. hVEGF-IN-1 (0.1875-3 μM; 48 h) lowers the amount of VEGF-A protein in MCF-7 cells[1]. hVEGF-IN-1 (0.375-3 μM; 48 h) cuts the migrated MCF-7 cells' relative wound closure by about 35% at a concentration of 3 μM[1]. hVEGF-IN-1 (1.25-10 μM) decreases the stability of the IRES-A G-Quadruplex in a dose-dependent manner[1]. Compound 1 down-regulated cap-independent translation mediated by the hVEGF-A IRES-A region in a dual-luciferase reporter assay in MCF-7 cells. At 1.5 µM, it reduced MetLuc activity (driven by the IRES-A) by approximately 80%. This effect was specific to the wild-type IRES sequence and not observed with mutant controls (MU2, MU3). In MCF-7 cells, compound 1 decreased the secretion of VEGF-A protein by about 75% at 1.5 µM, as measured by ELISA. Western blot analysis also confirmed a dose-dependent reduction in cellular VEGF-A protein levels (tested up to 3 µM). Compound 1 did not affect the transcription of the hVEGF-A gene or the IRES-A containing reporter gene, as shown by RT-PCR and qRT-PCR analyses. Compound 1 (at 3 µM) significantly inhibited the migration of MCF-7 breast cancer cells in a scratch wound healing assay under chemically induced hypoxic conditions (100 µM CoCl₂), reducing relative wound closure to ~35%, comparable to the effect of VEGF-A siRNA knockdown. This inhibitory effect on migration could be partially rescued by adding exogenous VEGF-A protein. In a trans-well migration assay using the more invasive MDA-MB-231 breast cancer cell line, compound 1 (3 µM) reduced cell migration to approximately 25% of the control. Real-time cell analysis (RTCA) confirmed that compound 1 suppressed MDA-MB-231 cell migration in a dose-dependent manner over 24 hours. [1] |
| ln Vivo |
hVEGF-IN-1 (compound 1) (7.5 mg/kg; i.p. once daily for 20 d) inhibits the growth of tumors in a human breast tumor xenograft[1]. In a human breast tumor xenograft model established with MCF-7 cells in athymic nude mice, administration of compound 1 at 7.5 mg/kg intraperitoneally (i.p.) once daily for 20 days significantly inhibited tumor growth. The average tumor volume in the treated group was less than 300 mm³ compared to over 700 mm³ in the saline control group after 20 days. The final tumor weight was reduced by approximately 60.1% (0.18 g vs 0.45 g in control). Immunohistochemistry (IHC) analysis of tumor tissues from the compound 1-treated group showed a marked decrease in VEGF-A protein expression compared to the control group. [1] |
| Enzyme Assay |
Surface Plasmon Resonance (SPR): Biotin-labeled RNA oligomers (IRES-A WT, IRES-A MU1) and a control hairpin DNA were immobilized on a GLH chip. Compound 1 and other library compounds were serially diluted in running buffer (50 mM Tris, 150 mM KCl, 0.005% Tween-20, pH 7.4) and injected over the chip surface at a flow rate of 50 µL/min. The association phase lasted 300 seconds, followed by a 200-second dissociation phase at 25°C. Binding responses were recorded, and dissociation constants (K_D) were calculated using the Langmuir fitting model. [1] Microscale Thermophoresis (MST): For direct binding measurements, the target RNA oligomer (IRES-A WT, MU1, or MU2) was labeled with a fluorescent dye (FAM). A constant concentration of labeled RNA was incubated with a serial dilution of compound 1 in 10 mM Tris-HCl buffer (pH 7.4) containing 100 mM KCl and 0.1 mM EDTA. After incubation, samples were loaded into glass capillaries, and thermophoresis movement was measured using an MST instrument. The change in normalized fluorescence was plotted against compound concentration to determine the K_D value. For competition binding with the BG4 antibody, the BG4 protein was labeled with an NT-647 dye. A constant concentration of labeled BG4 was incubated with a serial dilution of in vitro transcribed IRES-A mRNA in the presence or absence of a fixed concentration of compound 1. MST measurements were performed to determine the shift in binding affinity. [1] Competition Dialysis: Various nucleic acid structures, including the IRES-A WT RNA G-quadruplex, other RNA/DNA G-quadruplexes, duplex DNA (CT DNA), and control RNAs (hairpin, random RNA), were placed in separate dialysis cassettes after annealing. All cassettes were submerged in a common reservoir containing a fixed concentration of compound 1 in 10 mM Tris-HCl buffer (pH 7.4, 100 mM NaCl) and dialyzed for 24 hours at 25°C. The amount of compound bound to each nucleic acid was quantified spectrophotometrically after dialysis to assess binding selectivity. [1] Circular Dichroism (CD) Spectroscopy: RNA oligomers were annealed in 10 mM Tris-HCl buffer (pH 7.4) containing 100 mM KCl. CD spectra were recorded at 25°C in the absence and presence of increasing concentrations of compound 1. Changes in the characteristic peaks (e.g., ~260 nm for parallel G-quadruplex) were monitored to assess structural perturbation. For melting experiments, the CD signal at 262 nm was monitored as the temperature was increased from 25°C to 95°C at a controlled rate, both in the absence and presence of compound 1 (5 µM). The melting temperature (T_m) was determined from the fitted melting curves. [1] Electrophoretic Mobility Shift Assay (EMSA): Annealed IRES-A RNA oligomers (WT and MU2) were incubated with increasing concentrations of compound 1 in a buffer containing 100 mM KCl. The mixtures were then loaded onto a 16% native polyacrylamide gel prepared with a running buffer containing 100 mM KCl. Electrophoresis was performed at a constant voltage. The gel was stained with a fluorescent nucleic acid stain and visualized to observe shifts in RNA mobility or changes in band intensity corresponding to different structural states. [1] |
| Cell Assay |
Cell Line: MCF-7 cells Concentration: 0.1875, 0.375, 0.75, 1.5, 3 μM Incubation Time: 48 hours Result: Down-regulated hVEGF-A expression. Dual-Luciferase Reporter Assay: MCF-7 cells were co-transfected with two plasmids: one expressing Renilla luciferase (pRL-TK) as an internal control, and another where the Metridia luciferase (MetLuc) gene was placed downstream of the IRES-A sequence (WT or mutant) within a reporter construct. After 4 hours of transfection, cells were treated with compound 1 at various concentrations (0.1875 to 1.5 µM) for 24 hours. Cell culture supernatants were collected, and secreted MetLuc activity was measured using a chemiluminescent assay. Renilla luciferase activity from cell lysates was measured separately for normalization. The ratio of MetLuc to Renilla luciferase activity was calculated to determine the effect on IRES-mediated translation. [1] Enzyme-Linked Immunosorbent Assay (ELISA): MCF-7 cells were treated with compound 1 at concentrations ranging from 0 to 1.5 µM for 24 hours. The cell culture supernatant was collected and used to quantify secreted VEGF-A protein levels using a commercial ELISA kit specific for human VEGF-A, following the manufacturer's protocol. Absorbance was measured, and protein concentration was determined from a standard curve. [1] Western Blot: MCF-7 cells treated with compound 1 (0 to 3 µM) for 24-48 hours were lysed. Total protein was quantified, separated by SDS-PAGE, and transferred to a membrane. The membrane was probed with primary antibodies against VEGF-A and β-actin (loading control), followed by appropriate secondary antibodies conjugated to horseradish peroxidase. Protein bands were visualized using chemiluminescence. [1] RNA Immunoprecipitation (RIP): MCF-7 cells were transfected with a plasmid expressing FLAG-tagged BG4 antibody. After 24 hours, cells were treated with compound 1 (2.5 or 5 µM) or DMSO for another 24 hours. Cells were cross-linked with formaldehyde, lysed, and the lysate was incubated with anti-FLAG magnetic beads overnight at 4°C. Beads were washed, and bound RNA was eluted, followed by reverse cross-linking and proteinase K digestion. The purified RNA was analyzed by dot blotting, RT-PCR, and qRT-PCR using primers specific for the IRES-A region to quantify BG4-bound RNA. [1] Cell Scratch (Wound Healing) Assay: MCF-7 cells were grown to confluence in 6-well plates. A scratch was made using a pipette tip. Cells were then treated with 100 µM CoCl₂ to mimic hypoxia and with varying concentrations of compound 1 (0.375 to 3 µM) or transfected with VEGF-A siRNA (as a control) for 48 hours. In some wells, after 48 hours of compound treatment, exogenous VEGF-A protein was added for an additional 12 hours. The scratch area was photographed at 0 and 48 hours (or 60 hours for VEGF-A rescue) using phase-contrast microscopy. The wound closure area was quantified using image analysis software. [1] Trans-well Migration Assay: MDA-MB-231 cells were suspended in serum-free medium with or without compound 1 (0.375 to 3 µM) and seeded into the upper chamber of a trans-well insert with a porous membrane. The lower chamber contained medium with 10% fetal bovine serum (FBS) and 100 µM CoCl₂ as a chemoattractant. Cells were allowed to migrate for 24 hours. Non-migrated cells on the upper surface of the membrane were removed. Migrated cells on the lower surface were fixed, stained with MTT, dissolved in DMSO, and quantified by measuring absorbance at 570 nm. [1] Real-Time Cell Analysis (RTCA) for Migration: MDA-MB-231 cells were serum-starved for 24 hours. The lower chamber of a CIM-16 plate was filled with medium containing FBS and CoCl₂, with or without compound 1 (0.5 to 8 µM). The upper chamber was filled with serum-free medium containing the same concentration of compound. After background measurement, serum-starved cells were seeded into the upper chamber. The plate was placed in an RTCA instrument, and cell migration (represented as Cell Index) across the microporous membrane was monitored automatically every 15 minutes for 24 hours. [1] |
| Animal Protocol |
BALB/c female nude mice were implanted MCF-7 cells 7.5 mg/kg I.p. once daily for 20 days Xenograft Tumor Model and Treatment: Female BALB/c nude mice were subcutaneously implanted with MCF-7 human breast cancer cells. When tumors reached approximately 1000 mm³, tumor tissues were excised, divided, and re-implanted into the underarm regions of experimental mice. Mice were randomly divided into three groups: negative control (saline), positive control (doxorubicin, 1 mg/kg), and treatment (compound 1, 7.5 mg/kg). Compounds or saline were administered via intraperitoneal (i.p.) injection once daily for 20 consecutive days. The injection volume was 150 µL. Tumor diameters (length and width) and body weights were measured daily. After 20 days, mice were euthanized, tumors were excised and weighed, and tumor tissues were processed for immunohistochemistry (IHC) analysis of VEGF-A expression. [1] |
| ADME/Pharmacokinetics |
The provided literature does not contain specific ADME or pharmacokinetics data (e.g., half-life, clearance, bioavailability) for compound 1. [1] |
| Toxicity/Toxicokinetics |
In MCF-7 cells, the cytotoxicity IC₅₀ of compound 1 was reported to be 12.5 µM. In the MCF-7 xenograft mouse model, no significant change in body weight was observed in the group treated with compound 1 (7.5 mg/kg, i.p., daily for 20 days), indicating minimal systemic toxicity at this dose and regimen under the experimental conditions. [1] |
| References |
[1]. Discovery of Small Molecules for Repressing Cap-Independent Translation of Human Vascular Endothelial Growth Factor (hVEGF) as Novel Antitumor Agents. J Med Chem. 2017 Jul 13;60(13):5306-5319. |
| Additional Infomation |
Compound 1 is a novel quinazoline derivative, chemically designated as N-(2-(4-(N-(4-(2-diethylaminoethoxy)phenyl)amino)quinazolin-2-yl)phenyl)-3-(4-methylpiperazin-1-yl)propanamide. It was discovered through SPR-based screening of a library of 144 natural products and derivatives for binding to the G-rich sequence in the hVEGF-A IRES-A region. Its proposed mechanism of action is the selective binding and destabilization of a specific RNA G-quadruplex structure located in the 5′-UTR of hVEGF-A mRNA. This disrupts cap-independent translation initiation, leading to reduced VEGF-A protein synthesis, which in turn inhibits tumor cell migration and tumor growth. |
Solubility Data
| Solubility (In Vitro) |
DMSO: ~25 mg/mL (~43 mM) Ethanol: ~50 mg/mL (~86 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.30 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.30 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7190 mL | 8.5948 mL | 17.1895 mL | |
| 5 mM | 0.3438 mL | 1.7190 mL | 3.4379 mL | |
| 10 mM | 0.1719 mL | 0.8595 mL | 1.7190 mL |