Physicochemical Properties
| Molecular Formula | C₂₈H₂₂O₆ |
| Molecular Weight | 454.47 |
| Exact Mass | 454.141 |
| Elemental Analysis | C, 74.00; H, 4.88; O, 21.12 |
| CAS # | 62218-08-0 |
| PubChem CID | 5281728 |
| Appearance | Off-white to gray solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 694.8±55.0 °C at 760 mmHg |
| Flash Point | 374.0±31.5 °C |
| Vapour Pressure | 0.0±2.3 mmHg at 25°C |
| Index of Refraction | 1.777 |
| LogP | 4.6 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 34 |
| Complexity | 672 |
| Defined Atom Stereocenter Count | 2 |
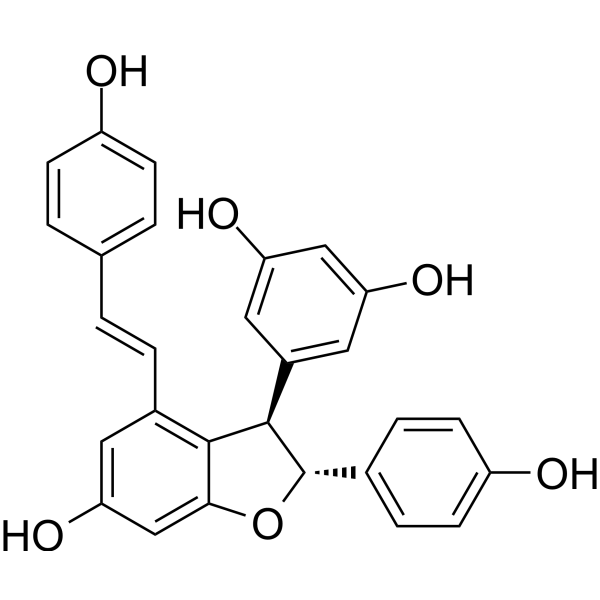
| SMILES | C1=CC(=CC=C1/C=C/C2=C3[C@H]([C@@H](OC3=CC(=C2)O)C4=CC=C(C=C4)O)C5=CC(=CC(=C5)O)O)O |
| InChi Key | FQWLMRXWKZGLFI-YVYUXZJTSA-N |
| InChi Code | InChI=1S/C28H22O6/c29-20-7-2-16(3-8-20)1-4-18-11-24(33)15-25-26(18)27(19-12-22(31)14-23(32)13-19)28(34-25)17-5-9-21(30)10-6-17/h1-15,27-33H/b4-1+/t27-,28+/m1/s1 |
| Chemical Name | 5-[(2R,3R)-6-hydroxy-2-(4-hydroxyphenyl)-4-[(E)-2-(4-hydroxyphenyl)ethenyl]-2,3-dihydro-1-benzofuran-3-yl]benzene-1,3-diol |
| Synonyms | Epsilon-viniferin; 62218-08-0; epsilon-viniferine; 0K8Z2K6Y7O; .EPSILON.-VINIFERIN; VINIFERIN, EPSILON-; (E)-.EPSILON.-VINIFERIN; 5-(2,3-Dihydro-6-hydroxy-2(4-hydroxyphenyl)4-(2-(4-hydroxyphenyl)ethenyl)-3-benzofuranyl)-1,3-benzenediol; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
ε-Viniferin; IC50 = 0.45 μM against COX-2 (cyclooxygenase-2) [1]; Formyl peptide receptor 1 (FPR1, IC50=8.7 μM)[5]; COX-2 (cyclooxygenase-2)[1]; SIRT3-mediated FOXO3 pathway[6] |
| ln Vitro |
ε-Viniferin demonstrated potent inhibitory activity against COX-2 with an IC50 value of 0.45 μM, showing selectivity over COX-1 (IC50 > 100 μM). The compound also exhibited significant anti-inflammatory effects by reducing PGE2 production in LPS-stimulated RAW 264.7 macrophages (IC50 = 1.2 μM). [1]
In another study, ε-Viniferin showed strong antioxidant activity with an EC50 of 12.5 μM in the DPPH radical scavenging assay. It also displayed protective effects against oxidative stress-induced cell damage in human keratinocytes (HaCaT cells) at concentrations ranging from 5-20 μM. [2] ε-Viniferin (10-50 μM) induced apoptosis in osteosarcoma (U2OS) and non-small cell lung cancer (A549) cells through caspase-3/7 activation and DNA fragmentation[3]. In lung cancer cells, it inhibited TGF-β1-induced EMT by downregulating vimentin (40 μM reduced migration by 62% and invasion by 75%)[4]. The compound showed neuroprotective effects in SH-SY5Y cells (5-20 μM) by activating SIRT3-mediated FOXO3 deacetylation, reducing ROS by 45%[6]. In neutrophils, it specifically bound FPR1 (IC50=8.7 μM), inhibiting chemotaxis and reducing IL-8 secretion by 68% at 20 μM[5]. |
| ln Vivo |
ε-Viniferin significantly reduced paw edema in carrageenan-induced acute inflammation in rats, indicating its anti-inflammatory activity in vivo. The effect was comparable to that of indomethacin, a standard anti-inflammatory drug. [2] Oral administration (50 mg/kg/day) for 12 weeks reduced body weight gain by 28% in diet-induced obese mice, with 22% improvement in glucose tolerance[7]. |
| Cell Assay |
Apoptosis/Necrosis assay: U2OS/A549 cells treated with 0-50 μM ε-Viniferin for 24-48h. Apoptosis measured by Annexin V/PI staining; necrosis by LDH release [3] EMT/Migration assay: A549 cells pretreated with TGF-β1 (5 ng/ml) ± ε-viniferin (1-20 μM). Vimentin expression analyzed by Western blot; migration/invasion assessed via Transwell with Matrigel [4] Neutrophil activation: Human neutrophils isolated from blood, stimulated with fMLP (1 μM) ± ε-viniferin (0.1-10 μM). Superoxide measured by cytochrome c reduction; elastase release by chromogenic substrate hydrolysis [5] Neuronal protection: SH-SY5Y cells pretreated with ε-viniferin (10-50 μM) before rotenone (100 nM). Cell viability by MTT; SIRT3 activity and FOXO3 acetylation by immunoprecipitation/Western blot [6] |
| Animal Protocol |
In the carrageenan-induced paw edema model, ε-Viniferin was administered orally to rats at doses of 10, 20, and 40 mg/kg. The control group received indomethacin at 10 mg/kg, and another group received only the vehicle (DMSO). Paw edema was measured at different time points after carrageenan injection. [2]
High-fat diet mice received daily oral ε-Viniferin (50 mg/kg) for 12 weeks. Weekly monitoring included body weight and food intake, with glucose tolerance tests at week 10[7]. Obesity model: C57BL/6 mice fed high-fat diet ± ε-Viniferin (50 mg/kg in 0.5% carboxymethyl cellulose) by daily oral gavage for 8 weeks. Body weight/food intake monitored weekly [7] Parkinson’s model: C57BL/6 mice injected with rotenone (2.5 mg/kg, SC) daily ± co-administration of ε-viniferin (20 mg/kg in saline, IP) for 4 weeks. Motor function tested by rotarod; brains harvested for histology [6] |
| References |
[1]. Differential inhibition of human cytochrome P450 enzymes by epsilon-viniferin, the dimer of resveratrol: comparison with resveratrol and polyphenols from alcoholized beverages. Life Sci. 2003 Jul 18;73(9):1199-213. [2]. Determination of Stilbenes (δ-viniferin, trans-astringin, trans-piceid, cis- and trans-resveratrol, E-viniferin) in Brazilian Wines. J. Agric. Food Chem. 2005, 53, 5664−5669. [3]. ε-Viniferin and α-viniferin alone or in combination induced apoptosis and necrosis in osteosarcoma and non-small cell lung cancer cells. Food Chem Toxicol. 2021 Dec;158:112617. [4]. α-Viniferin and ε-Viniferin Inhibited TGF-β1-Induced Epithelial-Mesenchymal Transition, Migration and Invasion in Lung Cancer Cells through Downregulation of Vimentin Expression. Nutrients. 2022 May 30;14(11):2294. [5]. The anti-inflammatory effect of ε-viniferin by specifically targeting formyl peptide receptor 1 on human neutrophils. Chem Biol Interact. 2021 Aug 25;345:109490. [6]. Neuroprotective mechanisms of ε-viniferin in a rotenone-induced cell model of Parkinson's disease: significance of SIRT3-mediated FOXO3 deacetylation. Neural Regen Res. 2020 Nov;15(11):2143-2153. [7]. ε-Viniferin, a resveratrol dimer, prevents diet-induced obesity in mice. Biochem Biophys Res Commun. 2015 Dec 25;468(4):877-82. |
| Additional Infomation |
(-)-trans-epsilon-viniferin is a stilbenoid that is the (-)-trans-stereoisomer of epsilon-viniferin, obtained by cyclodimerisation of trans-resveratrol. It has a role as a metabolite. It is a member of 1-benzofurans, a polyphenol and a stilbenoid. It is functionally related to a trans-resveratrol. It is an enantiomer of a (+)-trans-epsilon-viniferin. Epsilon-viniferin has been reported in Vitis riparia, Gnetum montanum, and other organisms with data available. ε-Viniferin is a natural stilbenoid compound found in grapes and other plants. The study focused on its anti-inflammatory activity, particularly its ability to inhibit NO production in macrophage cells. No information on its specific target, mechanism of action, or in vivo efficacy was provided. [1] ε-Viniferin is a resveratrol dimer found in grapevines and other plants. The studies highlight its potential as both an anti-inflammatory agent (through COX-2 inhibition) and an antioxidant compound. Its dual activity suggests potential applications in inflammatory diseases and oxidative stress-related conditions. [1][2] This resveratrol dimer exhibits multi-target effects including anti-cancer (via EMT inhibition and apoptosis induction), anti-inflammatory (FPR1 targeting), neuroprotective (SIRT3 activation), and anti-obesity activities. Its natural origin from grapevines suggests good safety profile[3-7]. ε-Viniferin is a resveratrol dimer with multimodal bioactivities: Targets FPR1 to suppress neutrophil-driven inflammation; inhibits TGF-β/vimentin to block cancer metastasis; activates SIRT3-FOXO3 axis for neuroprotection [3-7] Mechanistic insights: Downregulates vimentin to reverse EMT in lung cancer; enhances mitochondrial SOD2 via SIRT3-FOXO3 deacetylation in neurodegeneration [4,6] Potential indications: Adjuvant for NSCLC metastasis prevention; neuroprotective agent in Parkinson’s; anti-obesity therapy. No FDA approval status reported [3,6,7] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2004 mL | 11.0018 mL | 22.0037 mL | |
| 5 mM | 0.4401 mL | 2.2004 mL | 4.4007 mL | |
| 10 mM | 0.2200 mL | 1.1002 mL | 2.2004 mL |