diABZI STING agonist-3 is a potent and selective stimulator of interferon genes (STING) receptor agonist with an EC50s of 130 for human PBMCs. It is able to activate STING and stimulate the secretion of IFNβ, IL-6, TNF, and KC/GROα, exhibiting durable anti-tumour effects.
Physicochemical Properties
| Molecular Formula | C42H51N13O7 |
| Molecular Weight | 849.9372 |
| Exact Mass | 849.403 |
| Elemental Analysis | C, 59.35; H, 6.05; N, 21.42; O, 13.18 |
| CAS # | 2138498-18-5 |
| Related CAS # | diABZI STING agonist-1;2138299-33-7;diABZI STING agonist-1 trihydrochloride;2138299-34-8 |
| PubChem CID | 131986624 |
| Appearance | White to light yellow solid powder |
| LogP | 1.9 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 18 |
| Heavy Atom Count | 62 |
| Complexity | 1570 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O1C([H])([H])C([H])([H])N(C([H])([H])C1([H])[H])C([H])([H])C([H])([H])C([H])([H])OC1=C([H])C(C(N([H])[H])=O)=C([H])C2=C1N(C(N([H])C(C1=C([H])C(C([H])([H])[H])=NN1C([H])([H])C([H])([H])[H])=O)=N2)C([H])([H])/C(/[H])=C(\[H])/C([H])([H])N1C(N([H])C(C2=C([H])C(C([H])([H])[H])=NN2C([H])([H])C([H])([H])[H])=O)=NC2C([H])=C(C(N([H])[H])=O)C([H])=C(C1=2)OC([H])([H])[H] |
| InChi Key | JGLMVXWAHNTPRF-CMDGGOBGSA-N |
| InChi Code | InChI=1S/C42H51N13O7/c1-6-54-31(19-25(3)49-54)39(58)47-41-45-29-21-27(37(43)56)23-33(60-5)35(29)52(41)12-8-9-13-53-36-30(46-42(53)48-40(59)32-20-26(4)50-55(32)7-2)22-28(38(44)57)24-34(36)62-16-10-11-51-14-17-61-18-15-51/h8-9,19-24H,6-7,10-18H2,1-5H3,(H2,43,56)(H2,44,57)(H,45,47,58)(H,46,48,59)/b9-8+ |
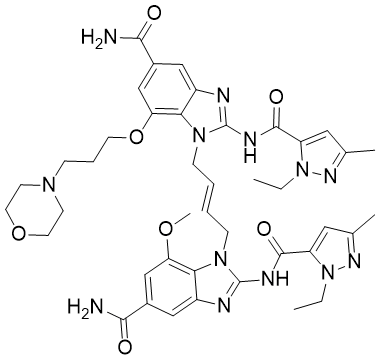
| Chemical Name | 1-[(E)-4-[5-carbamoyl-2-[(2-ethyl-5-methylpyrazole-3-carbonyl)amino]-7-(3-morpholin-4-ylpropoxy)benzimidazol-1-yl]but-2-enyl]-2-[(2-ethyl-5-methylpyrazole-3-carbonyl)amino]-7-methoxybenzimidazole-5-carboxamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Stimulator of interferon genes (STING) |
| ln Vitro |
The selective interferon gene (STING) receptor agonist stimulator diABZI STING agonist-1 (tautomer) has EC50 values of 130 and 186 nM for humans and mice, respectively. Compound 3, diABZI STING agonist-1, shows excellent selectivity for more than 350 tested kinases at 1 μM [1].
- Human PBMCs: diABZI STING agonist-1 induced dose-dependent activation of the STING pathway, as evidenced by increased phosphorylation of IRF3 and production of IFN-β and CXCL10. The EC₅₀ for IFN-β induction was 130 nM. The compound also demonstrated >100-fold selectivity for human STING over mouse STING[1]. - THP-1 cells: Treatment with diABZI STING agonist-1 led to robust activation of the STING-TBK1-IRF3 signaling axis, as measured by Western blot analysis of phosphorylated IRF3 and TBK1. This activation correlated with increased mRNA expression of IFN-β and pro-inflammatory cytokines[1]. |
| ln Vivo | Type I interferons and proinflammatory cytokines are activated in vivo in a STING-dependent manner by diABZI STING agonist-1 (tautomer) (subcutaneous injection; 2.5 mg/kg) [1]. The diABZI STING Agonist-1 (tautomer) (iv; 3 mg/kg) exhibited a half-life of 1.4 hours for systemic administration and attained systemic concentrations higher than the mouse STING's half-maximum effective concentration (EC50) of 200 ng/ml [1]. By the end of the trial, eight mice remained tumor-free on day 43 after treatment with diABZI STING agonist-1 (tautomer) (iv; 1.5 mg/kg; 43 days) significantly suppressed tumor growth and significantly increased survival (P < 0.001) in 10 mice [1]. |
| Enzyme Assay | Recombinant human STING binding assay: The binding affinity of diABZI STING agonist-1 to human STING was evaluated using surface plasmon resonance (SPR). The compound demonstrated a KD of 25 nM, indicating high binding affinity. The assay involved immobilizing recombinant human STING on a sensor chip and injecting serial dilutions of the compound to measure binding kinetics[1]. |
| Cell Assay | - Human PBMC activation assay: PBMCs were isolated from healthy donors and treated with diABZI STING agonist-1 at concentrations ranging from 0.1 to 1000 nM. After 24 hours, cell culture supernatants were analyzed for IFN-β and CXCL10 levels using ELISA. Cell viability was assessed using the MTT assay to ensure no cytotoxic effects at effective concentrations[1]. - THP-1 signaling assay: THP-1 cells were transfected with an IFN-β luciferase reporter plasmid. Following treatment with diABZI STING agonist-1, luciferase activity was measured to quantify STING pathway activation. Western blot analysis was performed to confirm phosphorylation of downstream signaling proteins[1]. |
| Animal Protocol |
Animal/Disease Models: wild and Sting−/− C57Blk6 mice Doses: 2.5 mg/kg Route of Administration: subcutaneous injection; 2.5 mg/kg Experimental Results: The secretion of IFNβ, IL-6, TNF and CXCL1 was activated in wild-type mice, But this is not the case in Sting−/− mice. Animal/Disease Models: BALB/c mouse colorectal tumor syngeneic mouse model (CT-26) [1] Doses: 3 mg/kg Route of Administration: intravenous (iv) (iv)injection; 200mg/kg. 3 mg/kg Experimental Results: The half-life is 1.4 hrs (hrs (hours)), and the systemic concentration is higher than the EC50 of mouse STING (200 ng/ml). Animal/Disease Models: BALB/c mouse colorectal tumor syngeneic mouse model (CT-26) [1] Doses: 1.5 mg/kg Route of Administration: intravenous (iv) (iv)injection; 200mg/kg. 1.5 mg/kg; 43-day Experimental Results: Dramatically inhibited tumor growth and improved survival rate. |
| References |
[1]. Design of amidobenzimidazole STING receptor agonists with systemic activity. Nature. 2018 Dec;564(7736):439-443. |
| Additional Infomation | - Mechanism of action: diABZI STING agonist-1 is a small-molecule STING agonist that binds to the ligand-binding domain of STING, triggering conformational changes that activate the TBK1-IRF3 signaling pathway, leading to the production of type I interferons and pro-inflammatory cytokines[1]. - Development rationale: The compound was designed to overcome limitations of cyclic dinucleotide (CDN) STING agonists, such as poor stability and systemic delivery challenges. Its amidobenzimidazole scaffold provides enhanced potency, selectivity, and oral bioavailability[1]. - Preclinical efficacy: diABZI STING agonist-1 demonstrated robust antitumor activity in multiple syngeneic mouse models, both as monotherapy and in combination with immune checkpoint inhibitors[1]. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.1766 mL | 5.8828 mL | 11.7655 mL | |
| 5 mM | 0.2353 mL | 1.1766 mL | 2.3531 mL | |
| 10 mM | 0.1177 mL | 0.5883 mL | 1.1766 mL |