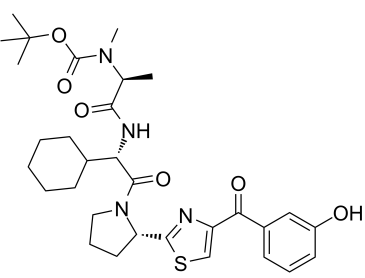
cIAP1 ligand 1 (E3 ligase Ligand 12, LCL-161-phenol) is a novel and potent IAP ligand based on the LCL161 analogue, used for synthesis of PROTAC degraders. cIAP1 ligand 1 can be connected to the ABL ligand for protein by a linker to form SNIPER.
Physicochemical Properties
| Molecular Formula | C31H42N4O6S |
| Molecular Weight | 598.753386974335 |
| Exact Mass | 598.282 |
| CAS # | 2095244-42-9 |
| PubChem CID | 138911355 |
| Appearance | White to off-white solid powder |
| LogP | 5.2 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 10 |
| Heavy Atom Count | 42 |
| Complexity | 983 |
| Defined Atom Stereocenter Count | 3 |
| SMILES | S1C=C(C(C2C=CC=C(C=2)O)=O)N=C1[C@@H]1CCCN1C([C@H](C1CCCCC1)NC([C@H](C)N(C(=O)OC(C)(C)C)C)=O)=O |
| InChi Key | SPXBMEKMIPOEMC-LQGLAIQGSA-N |
| InChi Code | InChI=1S/C31H42N4O6S/c1-19(34(5)30(40)41-31(2,3)4)27(38)33-25(20-11-7-6-8-12-20)29(39)35-16-10-15-24(35)28-32-23(18-42-28)26(37)21-13-9-14-22(36)17-21/h9,13-14,17-20,24-25,36H,6-8,10-12,15-16H2,1-5H3,(H,33,38)/t19-,24-,25-/m0/s1 |
| Chemical Name | tert-butyl N-[(2S)-1-[[(1S)-1-cyclohexyl-2-[(2S)-2-[4-(3-hydroxybenzoyl)-1,3-thiazol-2-yl]pyrrolidin-1-yl]-2-oxoethyl]amino]-1-oxopropan-2-yl]-N-methylcarbamate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
cIAP1 ligand 1 (a key component of AR-IAP PROTACs) targets Inhibitor of Apoptosis Protein (IAP) family members, including cIAP1, cIAP2, and XIAP. Key binding affinity data (Kd) from ITC assays: - cIAP1: Kd = 0.5 nM [1] - cIAP2: Kd = 1.2 nM [1] - XIAP: Kd = 45 nM [1] It functions as the IAP-recruiting moiety in PROTACs, which simultaneously bind androgen receptor (AR) via the AR ligand moiety, forming ternary complexes to induce AR degradation [1] |
| ln Vitro |
1. AR degradation activity (mediated by PROTACs containing cIAP1 ligand 1): - Prostate cancer cell lines (LNCaP, C4-2, 22Rv1): Treated with PROTACs (e.g., ARV-771, ARV-825) at 0.1–10 μM for 6–24 hours. Western blot analysis showed dose-dependent and time-dependent degradation of AR (full-length and splice variant AR-V7). AR degradation EC₅₀ values were 0.3 μM (LNCaP), 0.5 μM (C4-2), and 0.8 μM (22Rv1) for lead PROTAC ARV-825 [1] - Degradation specificity: PROTACs failed to degrade AR in AR-negative PC-3 cells, confirming AR-specific degradation [1] 2. Antiproliferative activity against prostate cancer cells: - LNCaP, C4-2, 22Rv1 cells: Treated with serial concentrations of PROTACs for 72 hours, cell viability measured by MTS assay. IC₅₀ values were 0.4 μM (LNCaP), 0.6 μM (C4-2), and 1.0 μM (22Rv1), while IC₅₀ > 10 μM in normal prostate epithelial cells (PrEC) [1] - CRPC (castration-resistant prostate cancer) relevance: PROTACs effectively inhibited 22Rv1 cells (AR-V7-positive, enzalutamide-resistant), with IC₅₀ 10-fold lower than enzalutamide [1] 3. Induction of apoptosis and cell cycle arrest: - LNCaP cells: 1 μM PROTAC (ARV-825) treatment for 24 hours induced PARP cleavage and caspase-3 activation (Western blot), with apoptotic rate increasing to 35% (Annexin V/PI staining, vs. 4% in vehicle control) [1] - Cell cycle analysis: 1 μM ARV-825 caused G2/M phase arrest (42% vs. 21% in control) in LNCaP cells [1] 4. Inhibition of AR transcriptional activity: - Luciferase reporter assay: LNCaP cells transfected with AR-responsive luciferase plasmid were treated with 0.1–1 μM PROTACs. AR transcriptional activity was inhibited by 65% (0.5 μM) and 88% (1 μM), consistent with AR protein degradation [1] |
| ln Vivo |
1. Antitumor efficacy in CRPC xenograft model: - Male nu/nu mice (6–8 weeks old) were subcutaneously injected with 2×10⁶ 22Rv1 cells into the right flank. When tumors reached 100–150 mm³, mice were randomized into 3 groups (n=6/group): vehicle control (10% DMSO + 40% PEG400 + 50% saline), PROTAC (ARV-825, containing cIAP1 ligand 1) 10 mg/kg, and 30 mg/kg [1] - Administration: Intraperitoneal injection once daily for 21 days. Tumor volume (measured with calipers every 3 days) and body weight (recorded daily) were monitored [1] - Efficacy: 10 mg/kg and 30 mg/kg groups showed tumor growth inhibition (TGI) of 62% and 85%, respectively, with no significant body weight loss (<5%) [1] 2. In vivo AR degradation validation: - Tumors were excised from mice treated with 30 mg/kg PROTAC for 24 hours. Tumor tissues were homogenized in RIPA buffer, and AR protein levels were detected by Western blot. Results showed >70% reduction in AR (full-length and AR-V7) compared to vehicle control [1] |
| Enzyme Assay |
1. ITC assay for IAP family protein binding: Purified recombinant cIAP1, cIAP2, or XIAP protein was dialyzed into assay buffer. cIAP1 ligand 1 was serially diluted and titrated into the protein solution at 25°C. Heat changes during binding were recorded, and binding affinity (Kd) was calculated by fitting the isotherm curve. Results showed high affinity for cIAP1 (Kd=0.5 nM) and cIAP2 (Kd=1.2 nM), and weak affinity for XIAP (Kd=45 nM) [1] 2. AR binding validation (for PROTACs): Surface Plasmon Resonance (SPR) was used to confirm PROTAC (containing cIAP1 ligand 1) binding to AR-LBD (ligand-binding domain). AR-LBD was immobilized on a sensor chip, and PROTAC solutions were injected at different concentrations. Binding responses were recorded, and equilibrium dissociation constants (KD) were derived, confirming specific AR binding [1] |
| Cell Assay |
1. AR degradation Western blot assay: - LNCaP/C4-2/22Rv1 cells were seeded in 6-well plates (5×10⁵ cells/well) and cultured overnight. Cells were treated with serial concentrations of PROTACs (0.01–10 μM) for 6–24 hours, or fixed concentration (1 μM) for 1–24 hours (time course). - Cells were lysed with RIPA buffer containing protease/phosphatase inhibitors, 30 μg protein separated by SDS-PAGE, transferred to PVDF membranes, and blocked with 5% non-fat milk. - Membranes were probed with anti-AR (targeting full-length AR and AR-V7), anti-cIAP1, and anti-β-actin (loading control) antibodies overnight at 4°C, followed by peroxidase-conjugated secondary antibodies. Bands were visualized with chemiluminescent reagents, and AR levels were quantified relative to β-actin [1] 2. Cell proliferation (MTS) assay: - Prostate cancer cells (LNCaP/C4-2/22Rv1) and PrEC cells were seeded in 96-well plates (3×10³ cells/well) and cultured overnight. - Serial concentrations of PROTACs were added, and cells were incubated for 72 hours at 37°C with 5% CO₂. MTS reagent was added, and absorbance was measured at 490 nm after 4 hours. IC₅₀ values were calculated by nonlinear regression [1] 3. Apoptosis and cell cycle assays: - Apoptosis: LNCaP cells were treated with 1 μM PROTAC for 24 hours, harvested, washed with PBS, stained with Annexin V-FITC and PI for 15 minutes in the dark, and analyzed by flow cytometry. - Cell cycle: Treated cells were fixed with 70% ethanol, stained with PI containing RNase A, and cell cycle distribution was analyzed by flow cytometry [1] 4. AR transcriptional activity (luciferase assay): - LNCaP cells were transfected with AR-responsive luciferase plasmid and Renilla luciferase plasmid (internal control) using transfection reagent. - After 24 hours, cells were treated with PROTACs (0.1–1 μM) for 16 hours. Luciferase activity was measured using a dual-luciferase assay kit, and relative activity was normalized to Renilla luciferase [1] |
| References |
[1]. Development of Protein Degradation Inducers of Androgen Receptor by Conjugation of Androgen Receptor Ligands and Inhibitor of Apoptosis Protein Ligands. J Med Chem. 2018 Jan 25;61(2):543-575. |
| Additional Infomation |
1. Mechanism of action: cIAP1 ligand 1 serves as the IAP-recruiting moiety in bifunctional PROTACs, which covalently link AR ligands (e.g., enzalutamide derivative) and cIAP1 ligand 1 via a PEG linker. The PROTAC binds to AR (via AR ligand moiety) and cIAP1 (via cIAP1 ligand 1), recruiting cIAP1 to the AR complex. cIAP1 acts as an E3 ubiquitin ligase, mediating AR ubiquitination and degradation via the ubiquitin-proteasome system [1] 2. Structural role: The linker length and exit vector between cIAP1 ligand 1 and AR ligand are critical for PROTAC activity. Optimal linker length (e.g., PEG6) enhances ternary complex formation (AR-PROTAC-cIAP1), maximizing AR degradation efficiency [1] 3. Therapeutic potential: PROTACs containing cIAP1 ligand 1 effectively target AR and AR-V7 (a key driver of enzalutamide resistance in CRPC), overcoming resistance to conventional AR inhibitors. They show potent antitumor activity in CRPC models with low normal cell toxicity, representing a promising therapeutic strategy for CRPC [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~167.01 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6701 mL | 8.3507 mL | 16.7015 mL | |
| 5 mM | 0.3340 mL | 1.6701 mL | 3.3403 mL | |
| 10 mM | 0.1670 mL | 0.8351 mL | 1.6701 mL |