Physicochemical Properties
| Molecular Formula | C14H12O3 |
| Molecular Weight | 228.24328 |
| Exact Mass | 228.079 |
| Elemental Analysis | C, 73.67; H, 5.30; O, 21.03 |
| CAS # | 52436-88-1 |
| PubChem CID | 5249188 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 2.362 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 17 |
| Complexity | 431 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CC1(CC2C(C(C3=CC=CC=C3C=2O1)=O)=O)C |
| InChi Key | DMSVAEFGRLMACV-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C14H12O3/c1-14(2)7-10-12(16)11(15)8-5-3-4-6-9(8)13(10)17-14/h3-6H,7H2,1-2H3 |
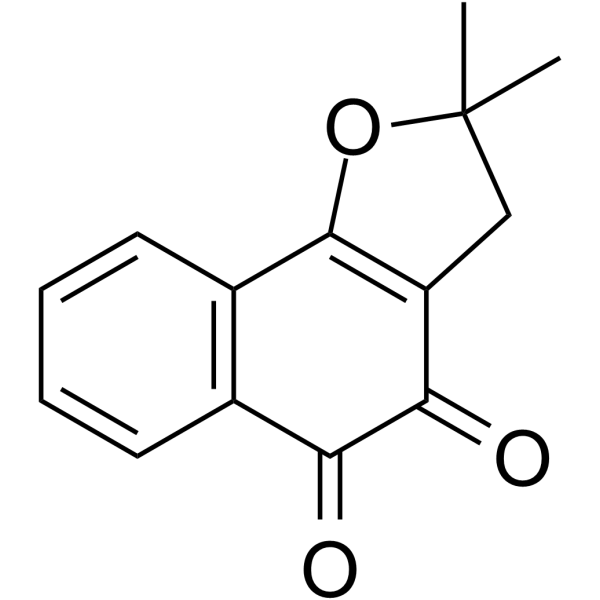
| Chemical Name | 2,2-dimethyl-3H-benzo[g][1]benzofuran-4,5-dione |
| Synonyms | nor-beta-lapachone; 52436-88-1; beta-Nor-lapachone; 2,2-dimethyl-3H-benzo[g][1]benzofuran-4,5-dione; beta--nor-Lapachone; nor-beta--Lapachone; Naphtho[1,2-b]furan-4,5-dione, 2,3-dihydro-2,2-dimethyl-; SCHEMBL107599; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Bacterial biofilm; Candida glabrata, ROS |
| ln Vitro | Infections caused particularly by Candida glabrata are hard to treat due to the development of antifungal resistance that occurs mainly through the production of efflux pumps and biofilm. Thus, a promising strategy to overcome infections caused by C. glabrata could be to use a substance able to inhibit efflux pumps and eradicate biofilms. Lapachones are natural naphthoquinones that possess a variety of pharmacological properties. Previous studies show that these substances inhibit the growth, virulence factors and efflux pumps of C. albicans. The aim of the present study was to evaluate whether lapachones are able to inhibit efflux pumps related to antifungal resistance in C. glabrata and either prevent biofilm formation or affect mature biofilms. Assays were performed with Saccharomyces cerevisiae strains that overexpress C. glabrata transporters (CgCdr1p and CgCdr2p). One C. glabrata clinical isolate that overexpresses CgCdr1p was also used. Both β-lapachone and β-nor-lapachone affected the growth of S. cerevisiae and C. glabrata when combined to fluconazole, and this action was inhibited by ascorbic acid. Both lapachones stimulated ROS production, inhibited efflux activity, adhesion, biofilm formation and the metabolism of mature biofilms of C. glabrata. Data obtained on the present study point to the potential use of β-lapachone and β-nor-lapachone as antibiofilm agents and adjuvants on the antifungal therapy related to resistant infections caused by C. glabrata. [1] |
| Enzyme Assay |
Antifungal activity of lapachones combined to fluconazole in liquid medium [1] The ability of lapachones of enhancing the fluconazole antifungal activity was assessed by the checkerboard method. Firstly, the S. cerevisiae strains (CgCdr1p + and CgCdr2p + ) were used in this assay. Yeasts (2 × 104 cells/ml) were added to YPD medium and incubated at 30 °C in the presence of combinations of serial concentrations of lapachones (100 µg/ml – 6.25 µg/ml) and fluconazole (500 µg/ml – 31.25 µg/ml) for 48 h with agitation (75 rpm). Cell growth was measured using a microplate reader at 600 nm. The fractional inhibitory concentration index (FICI) model was used to evaluate the interaction between lapachones and fluconazole. FICI is defined as the sum of the fractional inhibitory concentration (FIC) of each drug, while FIC is the ratio MIC combined / MIC alone. Synergistic, additive, indifferent, and antagonistic interactions were defined by FICI ≤ 0.5, 0.5 – 1.0, 1.0 – 4.0, and > 4.0, respectively. The compounds that improved the antifungal activity of fluconazole against the S. cerevisiae strains were therefore tested against the C. glabrata strains 109 and ATCC 2001. Yeasts (5 × 103 cells/ml) were added to RPMI-1640 medium and incubated at 37 °C in the presence of combinations of serial concentrations of lapachones (100 µg/ml – 6.25 µg/ml) and fluconazole (500 µg/ml – 3.9 µg/ml) for 48 h with agitation (75 rpm). Cell growth and interaction between the compounds were assessed as aforementioned. Spot assay [1] The spot method was performed in order to evaluate the antifungal activity of lapachones combined to fluconazole against different cellular concentrations, and to assess a possible role of ROS on the interaction between the compounds15. Briefly, 5-fold serial dilution of CgCdr1p+, CgCdr2p + and 109 (6 × 105 cells/ml) were spotted onto YPD agar in the presence or absence of fluconazole, lapachones, and 25 mM ascorbic acid. Plates were incubated at 30 °C (S. cerevisiae) or 37 °C (C. glabrata) for 48 h and photographed. In this assay, lapachones and fluconazole were used at the MIC combined concentration, obtained in the checkerboard assay. Nile red efflux assay [1] To evaluate whether the combined effect between lapachones and fluconazole was related to the inhibiton of efflux pumps, the fluorescent probe Nile red was used22. CgCdr1p+, CgCdr2p + and 109 cells (107 cells/ml) were harvested by centrifugation at 5000xg for 3 min and washed twice with cold 10 mM PBS. Then, cells were incubated for 1 h at 30 °C in the presence of the lapachones (at the MIC combined concentration), loaded with 7 µM Nile red and incubated for 30 min at 30 °C. Afterwards, cells were resuspended in PBS containing 0.2% glucose and incubated for 30 min at 30 °C. Fluorescence was measured at 485/538 nm (excitation/emission). Results were expressed as percentage of Nile red efflux in comparison to untreated cells. Preparation of plasma membranes [1] Since CgCdr1p and CgCdr2p are ABC transporters, they use ATP hydrolysis as energy source to extrude drugs from the cell. In order to evaluate if the compounds inhibit these efflux pumps by affecting their ATPase activity, plasma membranes enriched with the transporters were purified. S. cerevisiae and C. glabrata plasma membranes were obtained from Pdr5p-, CgCdr1p+, CgCdr2p+, 109, and ATCC 2001 strains as previously described23. Cells in the exponential growth phase were washed with 10 mM sodium azide. Then, yeast cell walls were digested with zymolase (4,1 mg for each 107 cells) and 2-mercaptoethanol (0,4% v/v). Remaining cells and organelles were removed by differential centrifugation, and plasma membranes were stored in liquid nitrogen. ATPase activity [1] The effect of lapachones on the ATPase activity of CgCdr1p and CgCdr2p was assessed by incubating purified membranes (0.013 mg/ml) obtained from CgCdr1p+, CgCdr2p + and 109 strains for 1 h at 37 °C in a reaction medium (100 mM Tris-HCl pH 7.5, 4 mM MgCl2, 75 mM KNO3, 7.5 mM NaN3, 0.3 mM (NH4)6Mo7O24, and 3 mM ATP) in the presence of lapachones at the MIC combined concentration. Then, the reaction was stopped by adding 1% sodium dodecyl sulfate (SDS). The inorganic phosphate generated was measured by the Fiske and Subbarrow method23. Purified plasma membranes from Pdr5p- and ATCC 2001 were used as negative control. Preformed biofilm [1] To evaluate the effect of the compounds on the metabolism of preformed biofilms, 24-hour mature biofilms of 109 and ATCC 2001 strains were built as described in section 2.11, in the absence of lapachones. Then, biofilms were incubated at 37 °C for 48 h in the presence or absence of serial concentrations of lapachones (200 – 12.5 µg/ml). Afterwards, the supernatant was removed and the wells were gently washed three times with PBS. The biofilm metabolic activity was measured as described in the section 2.10, and results were expressed as percentage in comparison to untreated cells. Membrane integrity of biofilm cells [1] The effect of lapachones on the integrity of plasma membranes of the cells within biofilms was assessed by using the fluorescent probe Sytox Green 25. Biofilms with a 24-hour maturation were treated with lapachones at 50 µg/ml and 200 µg/ml for 48 h at 37 °C and then incubated with 5 µM Sytox Green for 10 min at room temperature in the dark. Triton X-100 (1%) was used as positive control. Fluorescence was measured at 485/538 nm (excitation/emission), and results were expressed as mean fluorescence intensity. Reactive oxygen species measurement in biofilm cells In order to evaluate if the antibiofilm action of lapachones was related to the production of ROS, assays using the fluorescent probe DCFH-DA were performed. Biofilms with a 24-hour maturation were treated with lapachones at 200 µg/ml for 48 h at 37 °C, and then centrifuged at 5000xg for 3 min. Afterwards, cells were resuspended in PBS containing 10 µM DCFH-DA and incubated for 15 min at room temperature in the dark. Hydrogen peroxide (3%) was used as positive control. Fluorescence was measured at 485/538 nm (excitation/emission), and results were expressed as mean fluorescence intensity 21. Confocal laser scanning microscopy (CLSM) [1] In order to evaluate the effect of lapachones on biofilm integrity, CLSM analysis were performed. Biofilms formed in the presence of lapachones and mature biofilms treated with these compounds at 200 µg/ml were incubated in PBS containing 50 µg/ml Calcofluor White for 10 min at 37 °C in the dark. Then, biofilms were washed three times with PBS and analyzed by confocal laser scanning microscopy. Scanning electron microscopy (SEM) [1] SEM analysis were performed in order to assess whether the ultrastructure of preformed biofilms is affected by lapachones. Biofilms from 109 strain were built on poly lysine coated coverslips as described in section 2.11 and incubated with lapachones at 200 µg/ml or 2% DMSO at 37 °C for 48 h. Then, samples were fixed with 2.5% glutaraldehyde for 1 h at room temperature. Next, samples were post-fixed with 1% OsO4. Cells were washed with PBS, dehydrated in grades of ethanol, dried by the critical point method and set on metalic support. Antibiofilm activity of lapachones combined to fluconazole [1] In order to verify the antibiofilm activity of lapachones combined to fluconazole, the checkerboard method was carried out. Biofilms of 109 strain were built as described in 2.11 section in the presence of serial concentrations of lapachones (200 – 12.5 µg/ml) and fluconazole (1000 – 31.25 µg/ml). Preformed biofilms and high-density planktonic cells (107 cells/ml) were treated with combinations of lapachones (200 – 12.5 µg/ml) and fluconazole (1000 – 31.25 µg/ml). Cell viability was measured by MTT reduction assay, and the interaction between the compounds was assessed by the FICI index. |
| Cell Assay |
Reactive oxygen species measurement[1] The fluorescent probe DCFH-DA was used in order to measure the production of intracellular ROS induced by lapachones alone and combined to fluconazole 21. CgCdr1p+, CgCdr2p + and 109 cells (107 cells/ml) were incubated at 30 °C for 90 min in the presence of lapachones, fluconazole, lapachones + fluconazole, and lapachones + fluconazole + 25 mM ascorbic acid. Untreated cells were used as negative control. Then, cells were harvested by centrifugation at 5000xg for 3 min, resuspended in phosphate-buffered saline (PBS; 0.43g KH2PO4; 7.2g NaCl; 1.85 g Na2HPO4; pH 7.2; 990 ml distilled water) containing 10 µM DCFH-DA and incubated for 15 min at room temperature in the dark. Fluorescence was measured at 485/538 nm (excitation/emission), and results were expressed as mean fluorescence intensity. In this assay, lapachones and fluconazole were used at the MIC combined concentration, which values were obtained in the checkerboard assay. Human erythrocytes viability [1] The hemolytic effect of lapachones alone and combined to fluconazole was assessed as previously described. Erythrocytes were washed three times and resuspended in PBS to a final concentration of 2% v/v. Then, cells were incubated in the presence of lapachones alone and combined to fluconazole at MIC combined concentration for 1 h at 37 °C. Afterwards, erythrocytes were harvested by centrifugation at 3000xg for 5 min, and the absorbance of the hemoglobin present in the supernatant was measured at 540 nm. PBS and 1% Triton X-100 were used as 100% and 0% of hemolysis controls, respectively. . Adhesion to polystyrene [1] The effect of lapachones on the adhesion of C. glabrata to polystyrene was evaluated as described elsewhere. In a 96-well polystyrene flat-bottom plate, 109 and ATCC 2001 cell suspensions (107 cells/ml) were added to RPMI-1640 and incubated at 37 °C for 90 min with agitation (75 rpm) in the presence of lapachones at 50 µg/ml, 100 µg/ml, and 200 µg/ml. Then, the supernatant was discarded, and non-adhered cells were removed by gently washing the wells three times with PBS. Adhesion of C. glabrata to the wells was quantified by MTT reduction assay. Briefly, 3 mg/ml MTT and 0.4 mM menadione were added to the wells, and the plates were incubated at 37 °C for 3 h in the dark. Further, DMSO was added for 1 h in room temperature to dissolve the formazan crystals. The metabolic activity of cells was measured at 492 nm, and results were expressed as percentage in comparison to untreated cells. |
| References |
[1]. Effects of β-lapachone and β-nor-lapachone on multidrug efflux transporters and biofilms of Candida glabrata. Bioorg Med Chem. 2022;63:116749. |
| Additional Infomation | This study shows for the first time that β-lap and β-nor inhibit the most important efflux pumps related to the multidrug resistance phenotype in C. glabrata, allowing fluconazole to exert its antifungal activity. Data reveal that the interaction between the compounds is related to ROS production and ATPase activity inhibition. Also, both lapachones presented antibiofilm activity at different stages of biofilm development. It is important to highlight that, to our knowledge, it is the first time that the antifungal activity of lapachones against C. glabrata was evaluated. Moreover, this is the first report of the synergism between β-nor and an antimicrobial drug. In summary, data point to a potential use of β-lap and β-nor associated to fluconazole in the treatment of candidiasis caused by resistant C. glabrata isolates and also, as antibiofilm agents. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.3814 mL | 21.9068 mL | 43.8135 mL | |
| 5 mM | 0.8763 mL | 4.3814 mL | 8.7627 mL | |
| 10 mM | 0.4381 mL | 2.1907 mL | 4.3814 mL |