Physicochemical Properties
| Molecular Formula | C25H47N5O6S |
| Molecular Weight | 545.73558 |
| Exact Mass | 545.325 |
| CAS # | 149385-65-9 |
| PubChem CID | 10816380 |
| Appearance | White to off-white solid powder |
| Density | 1.137 g/cm3 |
| Boiling Point | 866.382ºC at 760 mmHg |
| LogP | 3.124 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 18 |
| Heavy Atom Count | 37 |
| Complexity | 766 |
| Defined Atom Stereocenter Count | 6 |
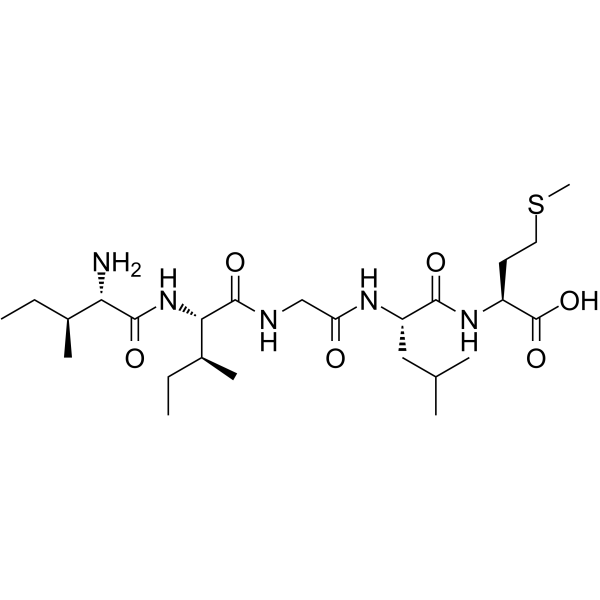
| SMILES | CC[C@H](C)[C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(=O)O)N |
| InChi Key | ZMDGLWRNBGRYQB-ZKHIMWLXSA-N |
| InChi Code | InChI=1S/C25H47N5O6S/c1-8-15(5)20(26)23(33)30-21(16(6)9-2)24(34)27-13-19(31)28-18(12-14(3)4)22(32)29-17(25(35)36)10-11-37-7/h14-18,20-21H,8-13,26H2,1-7H3,(H,27,34)(H,28,31)(H,29,32)(H,30,33)(H,35,36)/t15-,16-,17-,18-,20-,21-/m0/s1 |
| Chemical Name | (2S)-2-[[(2S)-2-[[2-[[(2S,3S)-2-[[(2S,3S)-2-amino-3-methylpentanoyl]amino]-3-methylpentanoyl]amino]acetyl]amino]-4-methylpentanoyl]amino]-4-methylsulfanylbutanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). This product is not stable in solution, please use freshly prepared working solution for optimal results.(2). Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | β-Amyloid (31-35) is the functional cytotoxic domain of the Aβ peptide. β-Amyloid(31-35) promotes the phosphorylation of biotinylated Aβ(1-40), enhances CDK-1 activity, and inhibits the binding of Aβ to cyclin B1. β-Amyloid(31-35) is cytotoxic, and olomoucine suppresses this action in neurons of the differentiated human teratocarcinoma cell line Ntera 2/cl-D1 (NT-2) [1]. Recommendations for Beta-Amyloid Aggregation (This is our suggested approach; it should be adjusted to suit your particular need as it simply offers guidelines). 1. Solid Aβ peptide should be dissolved in cold hexafluoro-2-propanol (HFIP). To achieve monomerization and randomization of the structure, incubate the peptides for a minimum of one hour at room temperature. 2. The HFIP is removed by evaporation and the resultant peptide is kept as a thin film at -20 or -80 °C. 3. The resultant membrane should be dissolved in 5 mM of anhydrous DMSO, vortexed, and diluted with buffer (serum- and phenol-red-free media) to the proper concentration. 4. After that, let the solution age for 48 hours at 4–8°C. The materials were then centrifuged at 14,000g for 10 minutes at 4-8°C; soluble oligomers were in the supernatant. For the studies, the supernatant was diluted 10-200 times. Different approaches are used based on the final application. |
| ln Vitro |
β-Amyloid (31-35) is the functional cytotoxic domain of the Aβ peptide. β-Amyloid(31-35) promotes the phosphorylation of biotinylated Aβ(1-40), enhances CDK-1 activity, and inhibits the binding of Aβ to cyclin B1. β-Amyloid(31-35) is cytotoxic, and olomoucine suppresses this action in neurons of the differentiated human teratocarcinoma cell line Ntera 2/cl-D1 (NT-2) [1]. Recommendations for Beta-Amyloid Aggregation (This is our suggested approach; it should be adjusted to suit your particular need as it simply offers guidelines). 1. Solid Aβ peptide should be dissolved in cold hexafluoro-2-propanol (HFIP). To achieve monomerization and randomization of the structure, incubate the peptides for a minimum of one hour at room temperature. 2. The HFIP is removed by evaporation and the resultant peptide is kept as a thin film at -20 or -80 °C. 3. The resultant membrane should be dissolved in 5 mM of anhydrous DMSO, vortexed, and diluted with buffer (serum- and phenol-red-free media) to the proper concentration. 4. After that, let the solution age for 48 hours at 4–8°C. The materials were then centrifuged at 14,000g for 10 minutes at 4-8°C; soluble oligomers were in the supernatant. For the studies, the supernatant was diluted 10-200 times. Different approaches are used based on the final application. β-Amyloid 31-35 (at 20 µM) increased the activity of recombinant CDK-1/cyclin B1, as measured by enhanced incorporation of ³²P from ATP into a biotinylated Histone H1 peptide substrate.[1] β-Amyloid 31-35 (at 20 µM) increased phosphoserine (pS) levels in Histone Type III-SS incubated with recombinant CDK-1, and increased phosphothreonine (pT) levels in a synthetic CDK-1 substrate (CSH 103) incubated with immunoprecipitated CDK-1 from neuronal cells.[1] β-Amyloid 31-35 (at 20 µM) did not affect the phosphorylation activity of recombinant or immunoprecipitated mitogen-activated protein kinase (MAPK p42) or protein kinase C δ (PKCδ) under the tested conditions.[1] β-Amyloid 31-35 (at 25 µM) exhibited cytotoxicity towards differentiated human NT-2 neurons after 24 hours of incubation, as assessed by the MTT reduction assay. This cytotoxicity was prevented by co-incubation with 50 µM of the CDK inhibitor olomoucine.[1] |
| Enzyme Assay |
Cyclin B1 Binding Assay (ELISA): Enzyme-linked immunosorbent assay plates were coated with recombinant human cyclin B1. Binding of biotinylated Aβ peptides (1-40 or 25-35) was detected. To test for competitive inhibition, unlabelled Aβ peptides (including Aβ 31-35) at 10-20 µM were added simultaneously with the biotinylated peptides. Binding was quantified, and affinity constants were calculated.[1] CDK-1 Kinase Activity Assay: The phosphorylation activity of recombinant CDK-1/cyclin B1 or immunoprecipitated CDK-1 was measured. Substrates included Histone Type III-SS, a synthetic Histone H1 peptide fragment (PKTPKKAKKL), or a synthetic CDK-1 substrate (CSH 103). Reactions were performed in the presence of [γ-³²P]ATP to measure ³²P incorporation into the substrate, or the reaction products were analyzed by immunoassay to detect increases in immunoreactive phosphoserine (pS) and phosphothreonine (pT) levels. Aβ peptides (including Aβ 31-35 at 20 µM) were added to the reaction mixtures to test their effects on kinase activity.[1] MAPK and PKC Activity Assays: For MAPK p42 assays, plates were coated with myelin basic protein as a substrate. For PKCδ assays, plates were coated with Histone Type III-SS. Assays were performed with recombinant kinases or kinases immunoprecipitated from cell extracts, in the presence of [γ-³²P]ATP or using immunoassays for pS/pT. Aβ peptides (including Aβ 31-35 at 20 µM) were tested for their ability to affect kinase activity.[1] |
| Cell Assay |
Neuronal Cytotoxicity Assay (MTT): Differentiated human NT-2 neurons were propagated in culture. Cells were incubated with test peptides (including β-Amyloid 31-35 at 25 µM) for 24 hours. In some experiments, the CDK inhibitor olomoucine (50 µM) was co-incubated with the peptide. After the incubation period, cell viability was assessed using the MTT reduction assay. The amount of formazan dye produced, which correlates with mitochondrial activity and cell viability, was measured.[1] |
| Toxicity/Toxicokinetics | β-Amyloid 31-35 (at 25 µM) is cytotoxic to differentiated human NT-2 neurons after 24-hour exposure in vitro.[1] |
| References |
[1]. The amyloid-beta peptide binds to cyclin B1 and increases human cyclin-dependent kinase-1 activity. Neurosci Lett. 2002 Apr 5;322(2):131-3. [2]. Fragment 31-35 of beta-amyloid peptide induces neurodegeneration in rat cerebellar granule cells via bax gene expression and caspase-3 activation. A crucial role for the redox state of methionine-35 residue. Neurochem Int. 2006 Oct;49(5):. [3]. Synthesis and biological properties of beta-turned Abeta(31-35) constrained analogues. Bioorg Med Chem Lett. 2008 Mar 15;18(6):2078-82. |
| Additional Infomation |
β-Amyloid 31-35 is a fragment of the amyloid-β peptide, which is associated with Alzheimer's disease pathology. It is considered the minimal sequence required for the cytotoxicity of the full-length Aβ peptide.[1] The proposed mechanism involves β-Amyloid 31-35 binding to cyclin B1, which is a regulatory component of the CDK-1 complex. This binding increases the kinase activity of CDK-1 towards its substrates. The resulting aberrant phosphorylation events may contribute to neuronal dysfunction and death, linking amyloid toxicity to cell cycle dysregulation observed in Alzheimer's disease.[1] The cytotoxicity of β-Amyloid 31-35 can be prevented by olomoucine, a broad-spectrum CDK inhibitor, supporting the role of CDK activation in its toxic mechanism.[1] The study suggests that the direct interaction between Aβ and cyclin B1/CDK-1 may provide a link between the amyloid hypothesis and the tau phosphorylation abnormalities characteristic of Alzheimer's disease.[1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~5 mg/mL (~9.16 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (3.81 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: 2.08 mg/mL (3.81 mM) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8324 mL | 9.1619 mL | 18.3237 mL | |
| 5 mM | 0.3665 mL | 1.8324 mL | 3.6647 mL | |
| 10 mM | 0.1832 mL | 0.9162 mL | 1.8324 mL |