Physicochemical Properties
| Molecular Formula | C3H6N2.HCL |
| Molecular Weight | 106.55408 |
| Exact Mass | 106.03 |
| CAS # | 646-03-7 |
| PubChem CID | 12244473 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 1.361 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 6 |
| Complexity | 49.2 |
| Defined Atom Stereocenter Count | 0 |
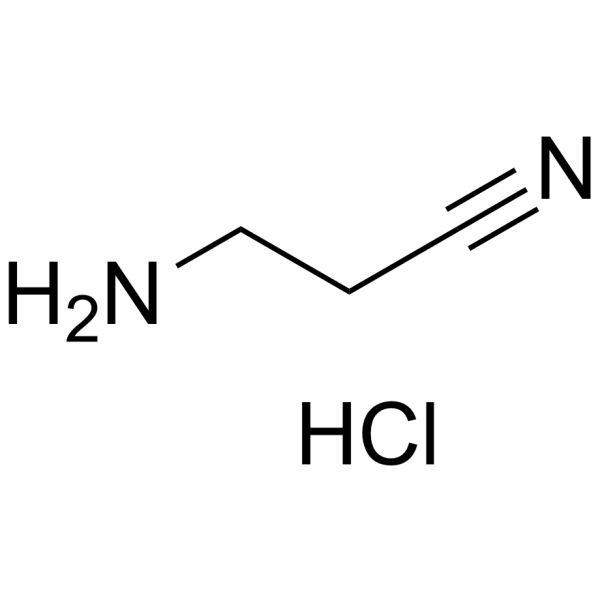
| SMILES | C(CN)C#N.Cl |
| InChi Key | SOWHACJNMKTTRC-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C3H6N2.ClH/c4-2-1-3-5;/h1-2,4H2;1H |
| Chemical Name | 3-aminopropanenitrile;hydrochloride |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | In an in vitro insulin resistance model, β-Aminopropionitrile (BAPN) increases glucose uptake and normalizes the expression of GLUT4 and adiponectin[1]. β-Aminopropionitrile (500 μM; 72 h) suppresses cervical cancer cells' ability to invade and migrate in vitro, as well as the morphological and marker protein alterations brought on by hypoxia-induced EMT[2]. |
| ln Vivo | In rats with diet-induced obesity, β-Aminopropionitrile (BAPN) (100 mg/kg/day; po; 6 weeks) improves the metabolic profile and decreases body weight gain[1]. C57BL/6 mice are given β-Aminopropionitrile monofumarate (1 g/kg/day; po; 4 weeks) to induce thoracic aortic dissection[3]. |
| Cell Assay |
Western Blot Analysis[1] Cell Types: 3T3-L1 adipocytes Tested Concentrations: 200 μM with 1.15 nM and 2.87 nM TNFα Incubation Duration: 72 h Experimental Results: TNFα decreased expression of GLUT4 and adiponectin, and increased SOCS3 protein levels in these cells. And these effects were prevented. Cell Invasion Assay[2] Cell Types: HeLa and SiHa cells Tested Concentrations: 500 μM Incubation Duration: 72 h Experimental Results: Dramatically decreased hypoxia- elicited cell invasion in both cell models. Cell Migration Assay [2] Cell Types: HeLa and SiHa cells Tested Concentrations: 500 μM Incubation Duration: 72 h Experimental Results: diminished hypoxia-induced migration from 180 and 240% to 60 and 70% in HeLa and SiHa cells, respectively. Western Blot Analysis[2] Cell Types: HeLa and SiHa cells Tested Concentrations: 500 μM Incubation Duration: 72 h Experimental Results: Effectively prevented hypoxia-induced downregulation of E-cadherin and strongly inhibited hypoxia-induced upregulation of α-SMA and vimentin. |
| Animal Protocol |
Animal/Disease Models: Male Wistar rats of 150 g, high-fat diet (HFD) model[1] Doses: 100 mg/kg/day Route of Administration: In the drinking water, 6 weeks Experimental Results: Dramatically prevented the rise in body weight in HFD rats, but not in animals that were fed a standard diet. decreased the increase in the weight of white adipose tissue (both epididymal and lumbar) in obese animals and attenuated their enhanced adiposity. Improved fasted glucose and insulin levels and consequently decreased HOMA index in the HFD group. Improved insulin signaling in adipose tissue from obese animals. Animal/Disease Models: C57BL/6 mice[3] Doses: 1 g/kg/day Route of Administration: In the drinking water, 4 weeks Experimental Results: Induce thoracic aortic dissection (TAD) in all mice with 24 h of Ang II infusion. Caused 87% of C57BL/6 mice to develop TAD without Ang II. |
| References |
[1]. The lysyl oxidase inhibitor β-aminopropionitrile reduces body weight gain and improves the metabolic profile in diet-induced obesity in rats. Dis Model Mech. 2015 Jun;8(6):543-51. [2]. Inactivation of lysyl oxidase by β-aminopropionitrile inhibits hypoxia-induced invasion and migration of cervical cancer cells. Oncol Rep. 2013 Feb;29(2):541-8. [3]. β-Aminopropionitrile monofumarate induces thoracic aortic dissection in C57BL/6 mice. Sci Rep. 2016 Jun 22;6:28149. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 9.3853 mL | 46.9263 mL | 93.8527 mL | |
| 5 mM | 1.8771 mL | 9.3853 mL | 18.7705 mL | |
| 10 mM | 0.9385 mL | 4.6926 mL | 9.3853 mL |