Physicochemical Properties
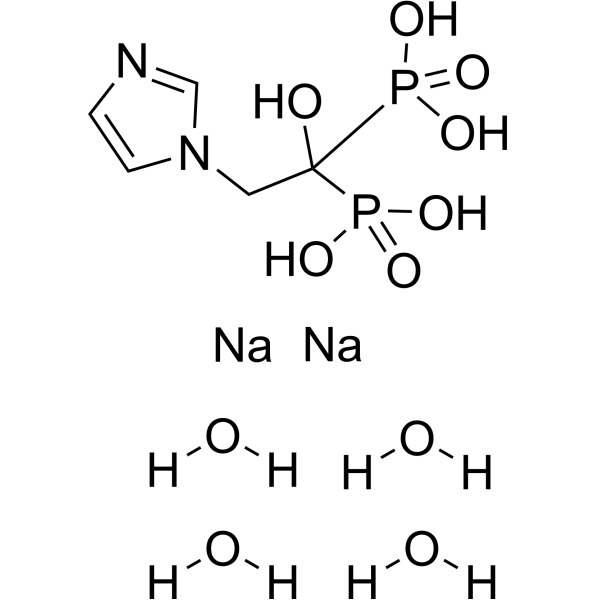
| Molecular Formula | C5H8N2O7P2-2.2[NA+].4[H2O] |
| Molecular Weight | 388.11454 |
| Exact Mass | 388.002 |
| Elemental Analysis | C, 15.47; H, 4.16; N, 7.22; Na, 11.85; O, 45.34; P, 15.96 |
| CAS # | 165800-07-7 |
| Related CAS # | Zoledronic Acid;118072-93-8;Zoledronic acid monohydrate;165800-06-6; Zoledronic Acid;118072-93-8;Zoledronic acid disodium tetrahydrate;165800-07-7; 131654-46-1 (disodium); 165800-08-8 (trisodium hydrate); 827573-11-5 (trisodium); 165800-07-7 (disodium hydrate); |
| PubChem CID | 23649597 |
| Appearance | Typically exists as solid at room temperature |
| Density | 2.13g/cm3 |
| Boiling Point | 764ºC at 760mmHg |
| Melting Point | 305-307ºC |
| Flash Point | 415.8ºC |
| Vapour Pressure | 1.53E-24mmHg at 25°C |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 22 |
| Complexity | 322 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O.O.O.O.OC(P(=O)([O-])[O-])(P(O)(O)=O)CN1C=CN=C1.[Na+].[Na+] |
| InChi Key | IEJZOPBVBXAOBH-UHFFFAOYSA-L |
| InChi Code | InChI=1S/C5H10N2O7P2.2Na.4H2O/c8-5(15(9,10)11,16(12,13)14)3-7-2-1-6-4-7;;;;;;/h1-2,4,8H,3H2,(H2,9,10,11)(H2,12,13,14);;;4*1H2/q;2*+1;;;;/p-2 |
| Chemical Name | Phosphonic acid, (1-hydroxy-2-(1H-imidazol-1-yl)ethylidene)bis-, disodium salt, tetrahydrate |
| Synonyms | Sodium (1-hydroxy-2-(1H-imidazol-1-yl)-1-Phosphonoethyl)phosphonate tetrahydrate; Zoledronic Acid, Disodium Salt, Tetrahydrate; Phosphonic acid, P,P'-[1-hydroxy-2-(1H-imidazol-1-yl)ethylidene]bis-, sodium salt, hydrate (1:2:4); disodium;(1-hydroxy-2-imidazol-1-yl-1-phosphonatoethyl)phosphonic acid;tetrahydrate; DISODIUM TETRAHYDRATE 1-HYDROXY-2-(IMIDAZOL-1-YL)-1-PHOSPHONOETHYLPHOSPHONATE; Sodium(1-hydroxy-2-(1H-imidazol-1-yl)-1-Phosphonoethyl)phosphonatetetrahydrate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Bisphosphonate (BP), with potent anti-resorptive; RANKL |
| ln Vitro | In osteocyte-like MLO-Y4 cells, zoledronic acid disodium tetrahydrate (0.1–1 µM; 48 hours) enhances the expression of sclerostin and receptor activator of nuclear factor kB ligand (RANKL) mRNA [2]. In MLO-Y4 cells, zoledronic acid disodium tetrahydrate can upregulate the expression of factors supporting osteoclastogenesis [2]. Through the IL-6/JAK2/STAT3 pathway, zoledronic acid disodium tetrahydrate increases RANKL expression in MLO-Y4 cells [2]. By controlling the NF-κB and JNK signaling pathways, zoledronic acid disodium tetrahydrate suppresses osteoclast differentiation and function [3]. Zoledronic Acid disodium tetrahydrate (10-100 µM; 1-7 days) dramatically lowers the viability of MC3T3-E1 cells and causes them to undergo apoptosis [4]. Because it induces apoptosis, zoledronic acid disodium tetrahydrate (10–100 µM; 4 days) reduces cell viability [4]. At concentrations less than 1 µM, zoledronic acid disodium tetrahydrate prevents MC3T3-E1 cells from differentiating and maturing [4]. |
| ln Vivo | Zoledronic acid disodium tetrahydrate (0.05 mg/kg; intraperitoneally; weekly; for 3 weeks) improves bone mineral density and content [5]. Zoledronic acid disodium tetrahydrate (0.5-1 mg/kg; i.p.; weekly; for 3 weeks) suppresses osteoclast and osteoblast function and bone remodeling in vivo, interfering with bone mechanical characteristics [5]. |
| Cell Assay |
Cell Viability Assay[4] Cell Types: MC3T3-E1 Cell Tested Concentrations: 0.01 µM, 0.1 µM, 1 µM, 10 µM, 100 µM Incubation Duration: 1 day, 3 days, 5 days, 7 days Experimental Results: Cell viability diminished at 10 µM and 100 µM. Apoptosis analysis[4] Cell Types: MC3T3-E1 Cell Tested Concentrations: 0.01 µM, 0.1 µM, 1 µM, 10 µM, 100 µM Incubation Duration: 1 Day, 4 Days, 7 Days Experimental Results: Early Apoptotic Cells and Late Apoptosis The number of apoptotic or necrotic cells increases in a dose-dependent and time-dependent manner (high concentration). Western Blot Analysis[4] Cell Types: MC3T3-E1 Cell Tested Concentrations: 0.01 µM, 0.1 µM, 1 µM, 10 µM, 100 µM Incubation Duration: 4 days Experimental Results: The protein level of inactive caspase-3 was down-regulated and up-regulated at 10 and Regulation of protein levels of active caspase-3 at 100 µM concentration. |
| Animal Protocol |
Animal/Disease Models: Fiveweeks old C57BL6 mice [5] Doses: 0.05 mg/kg, 0.5 mg/kg, 1 mg/kg Route of Administration: intraperitoneal (ip) injection, once a week for 3 weeks Experimental Results: Inhibition of osteoclasts and osteoblasts Osteocyte function and bone remodeling 0.5 mg/kg and 1 mg/kg. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Because no information is available on the use of zoledronic acid during breastfeeding, an alternate drug may be preferred, especially while nursing a newborn or preterm infant. However, absorption of zoledronic acid by a breastfed infant is unlikely. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. |
| References |
[1]. Various pathways of zoledronic acid against osteoclasts and bone cancer metastasis: a brief review. BMC Cancer. 2020; 20: 1059. [2]. Zoledronate Enhances Osteocyte-Mediated Osteoclast Differentiation by IL-6/RANKL Axis. Int J Mol Sci. 2019 Mar; 20(6): 1467. [3]. Zoledronic acid inhibits osteoclast differentiation and function through the regulation of NF-κB and JNK signalling pathways. Int J Mol Med. 2019 Aug;44(2):582-592. [4]. Dose-dependent inhibitory effects of zoledronic acid on osteoblast viability and function in vitro. Mol Med Rep. 2016 Jan; 13(1): 613-622. [5]. High-dose zoledronic acid impacts bone remodeling with effects on osteoblastic lineage and bone mechanical properties. Clin Cancer Res. 2009 Sep 15;15(18):5829-39. [6]. Oral Zoledronic acid bisphosphonate for the treatment of chronic low back pain with associated Modic changes: A pilot randomized controlled trial. J Orthop Res. 2022 Feb 23. |
| Additional Infomation |
Zoledronate Disodium is the disodium salt form of zoledronate, a synthetic imidazole, third generation bisphosphonate analog of pyrophosphate with antiresorptive activity. Zoledronate binds to hydroxyapatite crystals in the bone matrix and inhibits farnesyl pyrophosphate (diphosphate) synthase, thereby preventing protein prenylation within the mevalonate pathway. This leads to the loss of downstream metabolites essential for osteoclast function, leading to the induction of apoptosis and eventually, osteoclast-cell death. By preventing osteoclast-mediated bone resorption, zoledronate decreases bone turnover and stabilizes the bone matrix. An imidobisphosphonate inhibitor of BONE RESORPTION that is used for the treatment of malignancy-related HYPERCALCEMIA; OSTEITIS DEFORMANS; and OSTEOPOROSIS. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5766 mL | 12.8829 mL | 25.7659 mL | |
| 5 mM | 0.5153 mL | 2.5766 mL | 5.1532 mL | |
| 10 mM | 0.2577 mL | 1.2883 mL | 2.5766 mL |