Physicochemical Properties
| Molecular Formula | C19H18N2O3 |
| Molecular Weight | 322.36 |
| Exact Mass | 322.131 |
| CAS # | 1952264-34-4 |
| PubChem CID | 121414852 |
| Appearance | White to off-white solid powder |
| LogP | 1.8 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 24 |
| Complexity | 551 |
| Defined Atom Stereocenter Count | 0 |
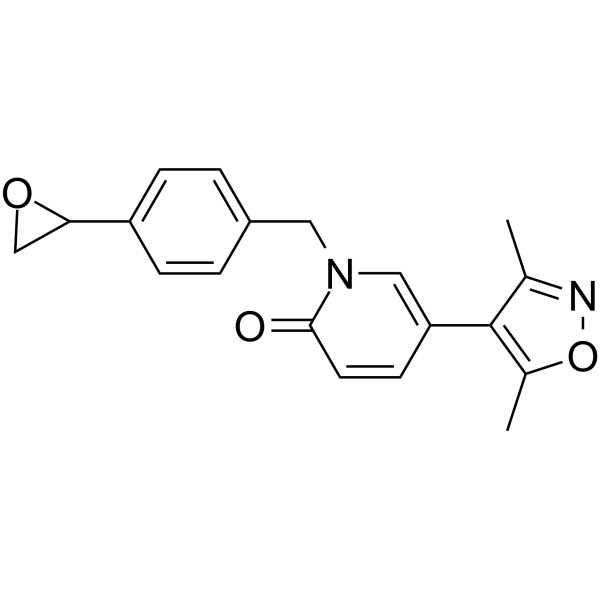
| SMILES | C1(=CC=C(CN2C=C(C3C(=NOC=3C)C)C=CC2=O)C=C1)C1OC1 |
| InChi Key | IELJHNIVCXDKQD-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C19H18N2O3/c1-12-19(13(2)24-20-12)16-7-8-18(22)21(10-16)9-14-3-5-15(6-4-14)17-11-23-17/h3-8,10,17H,9,11H2,1-2H3 |
| Chemical Name | 5-(3,5-dimethyl-1,2-oxazol-4-yl)-1-[[4-(oxiran-2-yl)phenyl]methyl]pyridin-2-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
1. BRD4(BD1) (targets Met149 residue in the bromodomain binding pocket, IC50 for BRD4(BD1) = 12 nM) [1] |
| ln Vitro |
1. Covalent binding activity: ZEN-3219 selectively forms a covalent bond with Met149 in BRD4(BD1) via its epoxide warhead; thermal denaturation assays confirmed that the compound increased the melting temperature of BRD4(BD1) by 12 °C, while no significant thermal shift was observed for other bromodomains (e.g., BRD2(BD1), BRD3(BD1)), indicating high target selectivity [1] 2. Bromodomain binding inhibition: ZEN-3219 inhibited the interaction between BRD4(BD1) and acetylated lysine (AcLys) peptides with an IC50 of 12 nM; MALDI-TOF mass spectrometry verified the formation of a covalent adduct between the compound and BRD4(BD1), with no detectable adducts formed with other BET family bromodomains [1] 3. Antiproliferative activity: In MV4-11 acute myeloid leukemia cells (BET-dependent cell line), ZEN-3219 exhibited durable antiproliferative effects; after a 24 h treatment with 50 nM of the compound followed by washout, cell proliferation remained inhibited by 78% at 72 h post-washout, whereas non-covalent BET inhibitors showed only 22% inhibition under the same conditions [1] 4. Transcriptional inhibition: ZEN-3219 suppressed the transcription of BRD4-dependent genes (e.g., MYC, BCL2) in MV4-11 cells; qRT-PCR results showed that 50 nM of the compound reduced MYC mRNA levels by 65% and BCL2 mRNA levels by 58% at 24 h, with the inhibitory effect persisting for 48 h post-compound washout [1] |
| Enzyme Assay |
1. Thermal denaturation assay: Purified BRD4(BD1) protein was incubated with serial concentrations of ZEN-3219 (0.1-1000 nM) in a buffer system for 1 h at room temperature. The sample was subjected to a temperature gradient (25-95 °C) in a thermal shift instrument, and the melting temperature (Tm) of the protein was calculated based on fluorescence signals from a dye that binds to unfolded protein. The change in Tm was used to evaluate the compound’s binding affinity and covalent interaction with BRD4(BD1) [1] 2. MALDI-TOF mass spectrometry assay for covalent adduct detection: Purified BRD4(BD1) was incubated with ZEN-3219 (100 nM) for 2 h at 37 °C, then desalted to remove unbound compound. The protein sample was analyzed via MALDI-TOF mass spectrometry, and the mass shift of BRD4(BD1) was calculated to confirm the formation of a covalent adduct (mass increase consistent with the molecular weight of ZEN-3219 minus the epoxide ring-opening moiety) [1] 3. X-ray crystallography assay: ZEN-3219 was co-crystallized with BRD4(BD1) protein, and the crystal structure was resolved at 1.8 Å resolution. The structure confirmed that the epoxide warhead of ZEN-3219 forms a covalent bond with the sulfur atom of Met149 in BRD4(BD1), while the core scaffold of the compound occupies the AcLys binding pocket, blocking the interaction with acetylated histones [1] |
| Cell Assay |
1. Cell proliferation assay: MV4-11 cells were seeded in 96-well plates at a density of 5×10³ cells per well and incubated for 12 h to attach. The cells were treated with serial dilutions of ZEN-3219 (1-1000 nM) for 24 h, followed by washing to remove unbound compound and further incubation for 48 h. A cell viability detection reagent was added to each well and incubated for 3 h at 37 °C, and absorbance was measured to calculate cell proliferation inhibition rates and evaluate the durability of the compound’s antiproliferative effect [1] 2. qRT-PCR for gene transcription detection: MV4-11 cells were treated with ZEN-3219 (50 nM) for 24 h, with a non-covalent BET inhibitor as a control (same concentration). Total RNA was extracted from the cells, reverse-transcribed into cDNA, and amplified using specific primers for MYC, BCL2, and reference genes. The relative mRNA expression levels were calculated via the 2^(-ΔΔCt) method to assess the compound’s effect on BRD4-dependent gene transcription [1] |
| References |
[1]. Design and Characterization of Novel Covalent Bromodomain and Extra-Terminal Domain (BET) Inhibitors Targeting a Methionine. J Med Chem. 2018 Sep 27;61(18):8202-8211. |
| Additional Infomation |
1. ZEN-3219 is a novel covalent BET inhibitor designed by attaching an epoxide warhead to a non-covalent BRD4(BD1) inhibitor scaffold, with the warhead oriented to target the Met149 residue in the BRD4(BD1) binding pocket [1] 2. The covalent binding mechanism of ZEN-3219 involves the epoxide group reacting with the sulfur-containing side chain of Met149, forming a stable covalent adduct that irreversibly blocks the AcLys binding pocket of BRD4(BD1), leading to durable inhibition of BRD4-mediated transcriptional regulation [1] 3. ZEN-3219 represents a new strategy for BET inhibitor development by targeting methionine residues for covalent modification, which offers improved target selectivity and durable pharmacodynamic effects compared with non-covalent BET inhibitors [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~25 mg/mL (~77.55 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1021 mL | 15.5106 mL | 31.0212 mL | |
| 5 mM | 0.6204 mL | 3.1021 mL | 6.2042 mL | |
| 10 mM | 0.3102 mL | 1.5511 mL | 3.1021 mL |