Zanubrutinib (formerly known as BGB-3111), an S-enantiomer, is a novel, highly selective, second generation BTK inhibitor, currently under clinical investigation in hematological cancers. BGB-3111 showed evidence of nanomolar BTK inhibition activity in both biochemical and cellular assays. BGB-3111 exhibited potent inhibition of cell proliferation in multiple MCL and DLBCL cell lines, as well as blocking downstream PLC-γ2 signaling and BTK autophosphorylation triggered by BCR aggregation. BGB-3111 demonstrated far more constrained off-target activities against a panel of kinases, including ITK, when compared to ibrutinib. Ibrutinib considerably reduced rituximab-induced NK cell IFN-γ secretion as well as in vitro cytotoxicity on mantle cell lymphoma cells; however, BGB-3111 was at least ten times less effective than ibrutinib in preventing rituximab-induced ADCC, which is in line with its feeble ITK inhibition activity.
Physicochemical Properties
| Molecular Formula | C27H29N5O3 | |
| Molecular Weight | 471.56 | |
| Exact Mass | 471.227 | |
| Elemental Analysis | C, 68.77; H, 6.20; N, 14.85; O, 10.18 | |
| CAS # | 1691249-45-2 | |
| Related CAS # | (±)-Zanubrutinib;1633350-06-7;(R)-Zanubrutinib;1691249-44-1;Zanubrutinib-d5 | |
| PubChem CID | 135565884 | |
| Appearance | White to off-white solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Boiling Point | 713.4±60.0 °C at 760 mmHg | |
| Flash Point | 385.2±32.9 °C | |
| Vapour Pressure | 0.0±2.3 mmHg at 25°C | |
| Index of Refraction | 1.680 | |
| LogP | 3.64 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 5 | |
| Rotatable Bond Count | 6 | |
| Heavy Atom Count | 35 | |
| Complexity | 756 | |
| Defined Atom Stereocenter Count | 1 | |
| SMILES | O=C(C=C)N1CCC(CC1)[C@]1([H])CCNC2=C(C(N)=O)C(C3C=CC(=CC=3)OC3C=CC=CC=3)=NN12 |
|
| InChi Key | RNOAOAWBMHREKO-QFIPXVFZSA-N | |
| InChi Code | InChI=1S/C27H29N5O3/c1-2-23(33)31-16-13-18(14-17-31)22-12-15-29-27-24(26(28)34)25(30-32(22)27)19-8-10-21(11-9-19)35-20-6-4-3-5-7-20/h2-11,18,22,29H,1,12-17H2,(H2,28,34)/t22-/m0/s1 | |
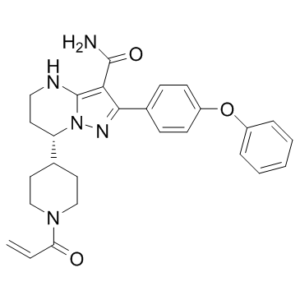
| Chemical Name | (7S)-2-(4-phenoxyphenyl)-7-(1-prop-2-enoylpiperidin-4-yl)-4,5,6,7-tetrahydropyrazolo[1,5-a]pyrimidine-3-carboxamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
BTK/Bruton tyrosine kinase BGB-3111 is more selective for BTK vs. EGFR, FGR, FRK, HER2, HER4, ITK, JAK3, LCK, BLK, and TEC than ibrutinib in biochemical and cellular assays[1]. BGB-3111 potently inhibits cell proliferation in multiple MCL and DLBCL cell lines by blocking downstream PLC-γ2 signaling and inhibiting BTK autophosphorylation triggered by BCR aggregation[2]. |
| ln Vitro |
BGB-3111 is more selective for BTK vs. EGFR, FGR, FRK, HER2, HER4, ITK, JAK3, LCK, BLK, and TEC than ibrutinib in biochemical and cellular assays[1]. BGB-3111 potently inhibits cell proliferation in multiple MCL and DLBCL cell lines by blocking downstream PLC-γ2 signaling and inhibiting BTK autophosphorylation triggered by BCR aggregation[2]. In preclinical studies, BGB-3111 showed more restricted off-target activities against a panel of kinases, including ITK, compared to ibrutinib. Due to its weaker activity on ITK, BGB-3111 was at least 10 times weaker than ibrutinib in inhibiting rituximab-induced antibody-dependent cellular cytotoxicity (ADCC) activity. [3] |
| ln Vivo |
BGB-3111 exhibits better oral bioavailability than ibrutinib in preclinical animal studies, achieving higher exposure and more complete target inhibition in the tissues[1]. BGB-3111 treatment causes a dose-dependent BTK occupancy in mouse BTK occupancy assays, and it exhibits approximately three times the potency of ibrutinib in target organs, such as the spleen and PBMC[2]. BGB-3111 exhibits better efficacy than ibrutinib and induces dose-dependent anti-tumor effects in both the REC-1 MCL and ABC subtype DLBCL (TMD-8) xenograft models. According to a rat toxicity study, BGB-3111 is highly well tolerated, and even at doses of up to 250 mg/kg/day, the MTD is not reached[3]. In the REC-1 mantle cell lymphoma (MCL) xenograft model and the ABC subtype diffuse large B-cell lymphoma (DLBCL, TMD-8) xenograft model, BGB-3111 induced dose-dependent antitumor effects and demonstrated superior efficacy in comparison with ibrutinib. [3] |
| Enzyme Assay |
Procedure for protein precipitation experiment:[4] human BTK protein were pre-incubated with BGB-3111 (31a, Zanubrutinib) or B43 (a reversible BTK inhibitor without the reactive chemical group) to form BTK/compound complexes (with BTK kinase in excess to ensure complete binding of compounds by BTK protein). The human BTK protein was then denatured and precipitated by centrifugation. The supernatants were analyzed for quantity of the original compound by LC-MS/MS or HPLC. B43 remained in the supernatant regardless of presence or absence of BTK protein during pre- S8 incubation, consistent with the reversible nature of its BTK binding (Figure 3B). In contrast, 31a was only recovered in the supernatant when pre-incubated without BTK protein. Upon preincubation with BTK, 31a largely disappeared from the supernatant (Figure 3A), consistent with the notion that 31a bound covalently/irreversibly to BTK protein thereby was removed from supernatant with precipitated BTK protein. The data also showed that the covalent bond formation between BTK and 31a was largely completed within 5 minutes[4]. In both biochemical and cellular assays, BGB-3111 demonstrated nanomolar BTK inhibition activity. In several MCL and DLBCL cell lines, BGB-3111 inhibited BCR aggregation-triggered BTK autophosphorylation, blocked downstream PLC-γ2 signaling, and potently inhibited cell proliferation. In comparison with ibrutinib, BGB-3111 showed much more restricted off-target activities against a panel of kinases, including ITK. While ibrutinib significantly inhibited rituximab-induced NK cell IFN-γ secretion and in vitro cytotoxicity on mantle cell lymphoma cells, BGB-3111 was at least 10-fold weaker than ibrutinib in inhibiting rituximab induced ADCC, consistent with its weak ITK inhibition activity.[2] Microsome intrinsic clearance: [4] Compounds (1 µM) were incubated in the liver microsomes at protein concentration of 0.5 mg/mL. The reactions were terminated S50 at 0, 2, 6, 10, 20, 30 min for human and 0, 5, 10, 20, 40, 60 min for dog, rat and mouse after the reactions were initiated. Compounds were analyzed using LC-MS/MS. Natural log of percentage remaining of compounds was plotted against the incubation time. The intrinsic clearance (CLint) was calculated with the following equation, where Cprotein was the microsomal protein concentration in the incubation system. CLint = -slope/Cprotein.[4] Cytochrome P450 Inhibition: [4] The human CYPs inhibition potential of compounds was evaluated based on the measurement of IC50 values in pooled human liver microsomes using 8 CYP probe substrates along with sample analysis by LC-MS/MS methods. Serial concentrations of compounds were incubated with each probe substrate in pooled human liver microsomes. Selective inhibitors were used as the positive controls to validate the incubation systems. Formation rate of the probe metabolites was monitored to calculate the IC50 values. The BTK biochemical assay used recombinant BTK protein. Compounds were pre-incubated with the enzyme for 1 hour at room temperature in assay buffer. The reaction was initiated by adding ATP (at its Km concentration) and a biotinylated peptide substrate. After 1 hour incubation, the reaction was stopped by adding a solution containing EDTA, a europium cryptate-conjugated anti-phosphotyrosine antibody, and streptavidin-labeled XL665. After a further 1-hour incubation, time-resolved fluorescence resonance energy transfer (TR-FRET) signals were measured [4] Assays for ITK, TEC, JAK3, EGFR, and HER2 were performed using a similar TR-FRET-based method [4] A broad kinase selectivity panel (342 kinases) was assessed using a filter-binding assay with ³³P-ATP. Compounds were pre-incubated with kinases for 1 hour, followed by addition of ATP to a final concentration of 1 μM. Reactions proceeded for 120 minutes before being spotted onto filter paper, washed, and quantified [4] |
| Cell Assay |
Cellular toxicity evaluation of BGB-3111 (31a, Zanubrutinib) (HEK293 and Ramos Proliferation Assay): The growthinhibitory activity of compounds in HEK293 and Ramos was determined using CellTiter-Glo luminescent cell viability assay. The number of cells seeded per well of a 96-well plate was optimized for each cell line to ensure logarithmic growth over 6 days treatment period (1000 cells/well for HEK293 cells, 6000 cells/well for Ramos cells). Cells were treated in triplicate with a 10-point dilution series. Following a 6-day exposure to the compound, a volume of CellTiter-Glo reagent equal to the volume of cell culture medium present in each well was added. Mixture was mixed on an orbital shaker for 5 minutes to allow cell lysis, followed by 10 minutes incubation at room temperature to allow development and stabilization of luminescent signal, which corresponded to quantity of ATP and thus the quantity of metabolically active cells. Luminescent signal was measured using PHERAstar FS reader. IC50 values for cell viability were determined with GraphPad Prism software.[4] The BTK pY223 cellular assay was conducted in Ramos cells. Cells were serum-starved overnight, treated with compounds for 3 hours, and then stimulated with pervanadate for 20 minutes to induce phosphorylation. Cells were lysed, and lysates were mixed with an HTRF detection antibody mixture (anti-BTK-d2 and anti-pBTK-K). After overnight incubation, fluorescence emission at 665 nm and 620 nm was measured to calculate the inhibition of phosphorylation [4] Cell viability assays for Rec-1 and OCI-LY10 cells were performed using a luminescent ATP detection assay. Cells were treated with compounds for 6 days. An equal volume of detection reagent was added to lyse cells and generate a luminescent signal proportional to viable cell number [4] The EGFR pY1068 cellular assay was performed in A431 cells. Cells were treated with compounds for 1 hour, stimulated with EGF for 10 minutes, lysed, and then analyzed using an HTRF detection kit specific for phosphorylated EGFR [4] The ITK p-PLCγ1 assay was performed in Jurkat cells. Cells were treated with compounds for 2 hours, stimulated with hydrogen peroxide, lysed, and analyzed by western blot using antibodies against total and phosphorylated PLCγ1 [4] The TEC p-TEC assay used TEC-overexpressing HEK293 cells. Serum-starved cells were treated with compounds for 3 hours, stimulated with pervanadate, lysed, and analyzed using an electrochemiluminescence-based immunoassay (Meso Scale Discovery) with plates coated with an anti-TEC capture antibody and a sulfotag-labeled anti-phosphotyrosine detection antibody [4] |
| Animal Protocol |
Pharmacokinetics (PK) study in rats: [4] Sprague-Dawley rats were housed in temperature (20-25°C) and humidity (40-70%) controlled facility with 12-hour light/dark cycle. The rats were sterilized water. The animals were fasted overnight prior to dosing until 4 hours post dosing. Water was not restricted during the study. Blood samples (0.15 mL) were collected via jugular vein cannula into dry heparinized (coated with 0.25% heparin in saline) tubes. For the IV administered groups, the sampling time points were pre-dose, 5, 15, 30, 45 min, 1, 2, 3, 4 and 6 h post-dose. For the oral administered groups, the sampling time points were pre-dose 15, 30 min, 1, 2, 3, 4, 6, 8, 12, and 24 h post-dose. Plasma samples were separated via centrifugation at 5500 rpm for 10 min, and stored in -30°C freezer until analysis. The plasma samples were analysis using LC-MS/MS with low limit of quantification (LLOQ) at 1 ng/mL. All PK parameters were calculated with non-compartment analysis using Phoenix WinNonlin 6.3. Plasma protein binding: [4] The plasma protein binding of compounds was measured using equilibrium dialysis with HTDialysis equilibrium dialysis chamber apparatus. In brief, an aliquot of compounds spiked plasma was added to the donor side of each designated well. An equal volume of phosphate buffer (0.002% Tween 80) was added to the receiver side. The plate was covered with adhesive sealing film to prevent evaporation and placed in a water bath at 37ºC for 5-8 h with a shaking speed at 80 rpm to reach protein binding equilibrium. Samples were taken from both sides, and analyzed using LC-MS/MS. The value of unbound fraction (fu) was determined by dividing the compounds concentration on the buffer side of the equilibrium dialysis chamber by the compounds concentration on the plasma side.[4] In mouse BTK occupancy assays, treatment with BGB-3111 resulted in a dose-dependent BTK occupancy and showed about 3-fold more potency than ibrutinib in target organs, including PBMC and spleen. BGB-3111 induced dose-dependent anti-tumor effects against REC-1 MCL xenografts engrafted either subcutaneously or systemically via tail vein injection in mice. In the subcutaneous xenografts, BGB-3111 at 2.5 mg/kg BID showed similar activity as ibrutinib at 50 mg/kg QD, its clinical relevant dose. In the systemic model, the median survival of BGB-3111 25 mg/kg BID group was significantly longer than those of both ibrutinib 50 mg/kg QD and BID groups. In an ABC-subtype DLBCL (TMD-8) subcutaneous xenograft model, BGB-3111 also demonstrated better anti-tumor activity than ibrutinib. Preliminary 14-day toxicity study in rats showed that BGB-3111 was very well tolerated and maximal tolerate dose (MTD) was not reached when it was dosed up to 250mg/kg/day.[2] For pharmacokinetic (PK) studies in rats, Zanubrutinib was administered intravenously as a 1 mg/kg solution or orally as a 5 mg/kg suspension in methylcellulose. Blood samples were collected at various time points for plasma concentration analysis [4] For pharmacodynamic (PD) studies in ICR mice, Zanubrutinib was administered orally via gavage as a solution or suspension in a volume of 10 mL/kg body weight. Mice were euthanized at specified times post-dose (e.g., 4 hours for dose-response, multiple time points for kinetics) to collect blood (for PBMCs and plasma) and spleen for BTK occupancy analysis [4] For the OCI-LY10 xenograft efficacy study, female NCG mice were inoculated subcutaneously with tumor cells. When tumors were palpable, mice were randomized into groups and treated orally with Zanubrutinib or vehicle, twice daily (BID) at doses of 2.5 or 7.5 mg/kg for 28 days. Tumor dimensions and body weights were measured regularly [4] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Following oral administration of zanubrutinib 160 mg twice daily and 320 mg once daily, the mean (%CV) zanubrutinib steady-state concentrations were 2,295 (37%) ng·h/mL and 2,180 (41%) ng·h/mL, respectively. The mean Cmax (%CV) was 314 (46%) ng/mL following 160 mg twice daily and 543 (51%) ng/mL following 320 mg once daily. The Cmax and AUC of zanubrutinib increase in a dose-proportional manner and there is minimal systemic accumulation after repeated dosing. The median Tmax is 2 hours. Following oral administration of 320 mg radiolabelled zanubrutinib, approximately 87% of the dose was excreted in the feces and about 8% of the dose was recovered in the urine, where less than 1% of the recovered drug comprised of unchanged parent drug. The geometric mean (%CV) apparent steady-state Vd is 881 (95%) L. The blood-to plasma ratio is about 0.7 to 0.8. The mean (%CV) apparent oral clearance (CL/F) of zanubrutinib is 182 (37%) L/h. Metabolism / Metabolites Zanubrutinib is predominantly metabolized by CYP3A4. Its metabolites have not been characterized. Biological Half-Life Following administration of a single oral dose of 160 mg or 320 mg of zanubrutinib, the mean half-life is approximately 2 to 4 hours. In rats following a 1 mg/kg IV dose, Zanubrutinib had a terminal half-life (T₁/₂) of 0.53 hours, a volume of distribution at steady state (Vss) of 2.32 L/kg, and a clearance (CL) of 76.8 mL/min/kg. Following a 5 mg/kg oral dose, it showed a time to maximum concentration (Tmax) of 0.33 hours, a maximum concentration (Cmax) of 235 ng/mL, an area under the curve (AUC) of 257 h·ng/mL, and an oral bioavailability (F) of 23.6%. Plasma protein binding free fraction (fu) was species-dependent: human 5.8%, dog 6.7%, rat 3.3%, mouse 5.1%. Microsomal intrinsic clearance was high in human (109 μL/min/mg) and rat (148 μL/min/mg), and moderate in dog (25.6 μL/min/mg) and mouse (67.0 μL/min/mg) [4] |
| Toxicity/Toxicokinetics |
Hepatotoxicity In the prelicensure clinical trials of zanubrutinib in patients with mantle cell lymphoma, liver test abnormalities were frequent although usually mild. ALT elevations arose in 28% and bilirubin levels in 24% of subjects, but were above 5 times the upper limit of normal (ULN) in less than 1%. In these trials that enrolled over 600 patients, there were no reports of clinically apparent liver injury, early discontinuations because of liver injury or liver related deaths. Nonetheless, other Bruton’s kinase inhibitors (ibrutinib, acalabrutinib) have been associated with rare cases of acute liver injury including acute liver failure. With those agents, the latency to onset of liver injury varied from several weeks to 9 months. The injury was typically hepatocellular and immunologic features were uncommon. While the pattern of injury was hepatocellular, the course was atypical of an acute hepatitis-like injury and more similar to acute hepatic necrosis with early onset of hepatic failure. Other Bruton’s kinase inhibitors have also been linked to several instances of reactivation of hepatitis B that can be severe and has been linked to fatal outcomes. Likelihood score: E* (unproven but suspected cause of clinically apparent liver injury including reactivation of hepatitis B in susceptible patients). Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation No information is available on the clinical use of zanubrutinib during breastfeeding. Because zanubrutinib is 94% bound to plasma proteins, the amount in milk is likely to be low. The manufacturer recommends that breastfeeding be discontinued during zanubrutinib therapy and for at least 2 weeks after the last dose. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding The plasma protein binding of zanubrutinib is approximately 94%. A toxicity study in rats indicated that BGB-3111 was very well tolerated, and the maximum tolerated dose (MTD) was not reached when it was dosed up to 250 mg/kg/day. [3] In an ongoing phase 1 first-in-human trial in patients with advanced B-cell malignancies, there were no drug-related adverse events (AEs) and no dose-limiting toxicities (DLT) reported at the time of the report. The MTD had not been reached. [3] |
| References |
[1]. Cancer Cell. 2015 Aug 10;28(2):225-39. [2]. Cancer Res (2015) 75 (15_Supplement): 2597. [3]. J Hematol Oncol . 2016 Sep 2;9(1):80. [4]. J Med Chem . 2019 Sep 12;62(17):7923-7940. |
| Additional Infomation |
Pharmacodynamics Zanubrutinib is an immunomodulating agent that decreases the survival of malignant B cells. It inhibits BTK by binding to its active site. It works to inhibit the proliferation and survival of malignant B cells to reduce the tumour size in mantle cell lymphoma. BGB-3111 is a second-generation, highly selective and potent BTK inhibitor under clinical development. [3] BGB-3111 is being developed as a potential treatment for B-cell malignancies. [3] A first-in-human, open-label phase 1 trial of BGB-3111 is ongoing, using a modified 3 + 3 dose-escalation design (40, 80, 160, 320 mg orally once daily; 160 mg orally twice daily) in patients with advanced B-cell malignancies. Preliminary results from 25 patients showed an objective response rate of 64% (16/25), including 1 complete response (CR) and 6 cases of stable disease (SD). [3] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.41 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.41 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.41 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1206 mL | 10.6031 mL | 21.2062 mL | |
| 5 mM | 0.4241 mL | 2.1206 mL | 4.2412 mL | |
| 10 mM | 0.2121 mL | 1.0603 mL | 2.1206 mL |