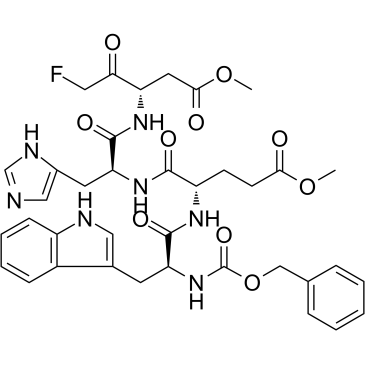
Z-WEHD-FMK is a novel, highly potent, cell-permeable and irreversible caspase-1/5 inhibitor. Z-WEHD-FMK also exhibits a robust inhibitory effect on cathepsin B activity (IC50=6 μM). Z-WEHD-FMK can be used to investigate cells for evidence of apoptosis.
Physicochemical Properties
| Molecular Formula | C37H42N7O10F |
| Molecular Weight | 763.76868 |
| Exact Mass | 763.298 |
| CAS # | 210345-00-9 |
| PubChem CID | 25108687 |
| Appearance | Light yellow to yellow solid powder |
| LogP | 4.205 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 23 |
| Heavy Atom Count | 55 |
| Complexity | 1330 |
| Defined Atom Stereocenter Count | 4 |
| SMILES | COC(=O)CC[C@@H](C(=O)N[C@@H](CC1=CN=CN1)C(=O)N[C@@H](CC(=O)OC)C(=O)CF)NC(=O)[C@H](CC2=CNC3=CC=CC=C32)NC(=O)OCC4=CC=CC=C4 |
| InChi Key | NLZNSSWGRVBWIX-KRCBVYEFSA-N |
| InChi Code | InChI=1S/C37H42FN7O10/c1-53-32(47)13-12-27(34(49)44-30(15-24-19-39-21-41-24)36(51)43-28(31(46)17-38)16-33(48)54-2)42-35(50)29(14-23-18-40-26-11-7-6-10-25(23)26)45-37(52)55-20-22-8-4-3-5-9-22/h3-11,18-19,21,27-30,40H,12-17,20H2,1-2H3,(H,39,41)(H,42,50)(H,43,51)(H,44,49)(H,45,52)/t27-,28-,29-,30-/m0/s1 |
| Chemical Name | methyl (4S)-5-[[(2S)-1-[[(3S)-5-fluoro-1-methoxy-1,4-dioxopentan-3-yl]amino]-3-(1H-imidazol-5-yl)-1-oxopropan-2-yl]amino]-4-[[(2S)-3-(1H-indol-3-yl)-2-(phenylmethoxycarbonylamino)propanoyl]amino]-5-oxopentanoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Nearly entirely blocks C is Z-WEHD-FMK (80 μM; 9 hours). golgin-84 cleavage brought on by trachomatis and raises GM130 expression in cells [1]. Z-WEHD-FMK successfully inhibited 0909I E. 30 minutes before to exposure to E. piscicida. pyroptosis morphology and piscicida-induced cytotoxicity in ZF4 cells. Moreover, it prevents cytotoxicity brought on by the administration of cytosolic LPS [2]. Significantly, Z-WEHD-FMK (20 μM; 18–24 hours after Cr3+, Ni2+, and Co2+) elicited a 76%–86% reduction in IL-1β production, with a similar reduction observed at 200–400 ppm Cr3+ 35%. Inhibitors of caspase-1 caused a reduction of 40% to 45% when Ni2+ concentrations were 48 ppm or higher. Ultimately, the caspase-1 inhibitor reduced the levels of Co2+ to below the detection threshold at 6 ppm, and in bone marrow-derived macrophages (BMDM) at 12 to 24 ppm, the reduction might range from 40% to 48%[3]. |
| Cell Assay |
Western Blot Analysis[1] Cell Types: Chlamydia trachomatis-infected or mock-infected HeLa cells Tested Concentrations: 80 μM Incubation Duration: 9 hrs (hours) Experimental Results: Increased expression of golgin-84 and GM130. Cell viability assay [2] Cell Types: Mycoplasma-free ZF4 cells Tested Concentrations: Incubation Duration: 30 minutes before exposure to E. piscicida Experimental Results: Inhibition of cytotoxicity and pyroptosis morphology of ZF4 cells. |
| References |
[1]. Caspase-4 and caspase-5, members of the ICE/CED-3 family of cysteine proteases, are CrmA-inhibitable proteases.Cell Death Differ. 1997 Aug;4(6):473-8. [2]. Sensing of cytosolic LPS through caspy2 pyrin domain mediates noncanonical inflammasome activation in zebrafish.Nat Commun. 2018 Aug 3;9(1):3052. [3]. Effects of metal ions on caspase-1 activation and interleukin-1β release in murine bone marrow-derived macrophages.PLoS One. 2018 Aug 23;13(8):e0199936. [4]. CA-074Me protection against anthrax lethal toxin.Infect Immun. 2009 Oct;77(10):4327-36. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~130.93 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (3.27 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (3.27 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (3.27 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.3093 mL | 6.5465 mL | 13.0929 mL | |
| 5 mM | 0.2619 mL | 1.3093 mL | 2.6186 mL | |
| 10 mM | 0.1309 mL | 0.6546 mL | 1.3093 mL |