Physicochemical Properties
| Molecular Formula | C74H86N10O25 |
| Molecular Weight | 1515.53 |
| Exact Mass | 1514.576558 |
| PubChem CID | 171713852 |
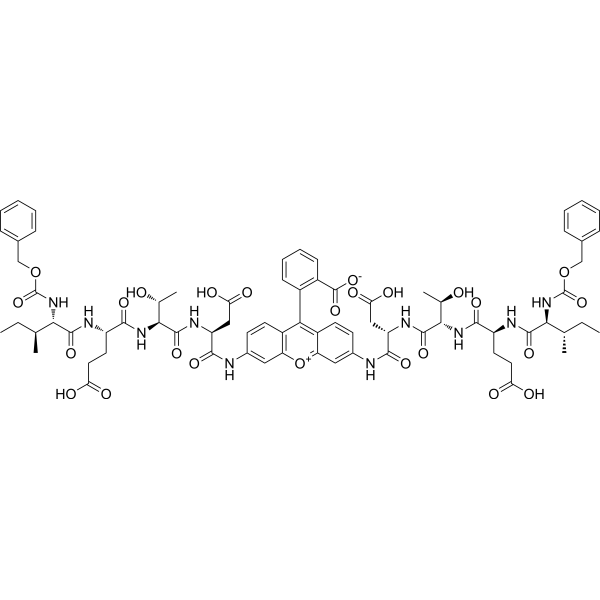
| Sequence | (Z-Ile-Glu-Thr-Asp)2-Rhodamine 110 |
| SequenceShortening | (Z-IETD)2-Rhodamine 110 |
| Appearance | Typically exists as solids at room temperature |
| Hydrogen Bond Donor Count | 16 |
| Hydrogen Bond Acceptor Count | 24 |
| Rotatable Bond Count | 41 |
| Heavy Atom Count | 109 |
| Complexity | 2960 |
| Defined Atom Stereocenter Count | 12 |
| SMILES | CC[C@H](C)[C@@H](C(=O)N[C@@H](CCC(=O)O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(=O)O)C(=O)NC1=CC2=C(C=C1)C(=C3C=CC(=CC3=[O+]2)NC(=O)[C@H](CC(=O)O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CCC(=O)O)NC(=O)[C@H]([C@@H](C)CC)NC(=O)OCC4=CC=CC=C4)C5=CC=CC=C5C(=O)[O-])NC(=O)OCC6=CC=CC=C6 |
| InChi Key | DSMSOMVESVLVDG-TVEQXDRTSA-N |
| InChi Code | InChI=1S/C74H86N10O25/c1-7-37(3)60(83-73(105)107-35-41-17-11-9-12-18-41)68(99)77-49(27-29-55(87)88)64(95)81-62(39(5)85)70(101)79-51(33-57(91)92)66(97)75-43-23-25-47-53(31-43)109-54-32-44(24-26-48(54)59(47)45-21-15-16-22-46(45)72(103)104)76-67(98)52(34-58(93)94)80-71(102)63(40(6)86)82-65(96)50(28-30-56(89)90)78-69(100)61(38(4)8-2)84-74(106)108-36-42-19-13-10-14-20-42/h9-26,31-32,37-40,49-52,60-63,85-86H,7-8,27-30,33-36H2,1-6H3,(H14-,75,76,77,78,79,80,81,82,83,84,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106)/t37-,38-,39+,40+,49-,50-,51-,52-,60-,61-,62-,63-/m0/s1 |
| Chemical Name | 2-[3,6-bis[[(2S)-3-carboxy-2-[[(2S,3R)-2-[[(2S)-4-carboxy-2-[[(2S,3S)-3-methyl-2-(phenylmethoxycarbonylamino)pentanoyl]amino]butanoyl]amino]-3-hydroxybutanoyl]amino]propanoyl]amino]xanthen-10-ium-9-yl]benzoate |
| Synonyms | Z-IETD-R110; Z-IETD R110; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Caspase-8 |
| ln Vitro | Marked caspase-8 activation was demonstrated in a minority of isolated pancreatic acinar cells by using a fluorescent probe-linked caspase-8 substrate (Z-IETD-R110). Menadione (30 μM) induced a significant increase (P < 0.00018) in the fluorescence of 15 ± 2% of acinar cells (Fig. 1B). Apoptosis-positive cells displayed a high increase in fluorescence (∼10.9 ± 0.8-fold) compared with control cells (Fig. 2A). Fluorescence of the caspase-8 substrate increased within 30 min after application of menadione (Fig. 2B). These data show that caspase-8 was activated in response to the menadione-induced oxidative stress. Confocal-microscopy images (Fig. 2C) show homogeneous distribution of fluorescence throughout the cell, suggesting predominantly cytosolic localization of activated caspase-8. [1] |
| Cell Assay |
Caspase activation. [1] Isolated pancreatic acinar cells were washed and suspended in calcium-free buffer solution (140 mM NaCl, 1.13 mM MgCl2, 4.7 mM KCl, 10 mM glucose, 0.1 M EDTA, and 10 mM HEPES, pH 7.2). Cells were then loaded with fluorescent indicator-linked substrates for activated caspase-2 (10 μM Z-VDAD-R110), caspase-8 (10 μM Z-IETD-R110), caspase-9 (10 μM Z-LEHD-R110), or general caspases (10 μM R110-aspartic acid amide) at room temperature for 20 min or for caspase-10 (50 μM AEVD-AFC) or caspase-12 (50 μM ATAD-AFC) at 37°C for 1 h. Caspase substrates (except for the general substrate) used in this study were specific for the relevant initiator caspases, as reported. To avoid activation of substrates by executioner caspases, all experiments were strictly limited to the first 30 min after induction of apoptosis by menadione. After loading, cells were washed and resuspended in calcium-free buffer solution. The isolated cells were placed on a Leica SP2 confocal microscope stage, and fluorescence was imaged over time (excitation 488 nm, emission 505–543 nm for caspase-2, -8, -9, or general caspase substrates; excitation 405 nm, emission 475–600 nm for caspase-10 and -12 substrates). Cells were then treated with 30 μM menadione. To examine colocalization of caspase activation and the position of mitochondria, cells were also loaded with MitoTracker Deep Red 633 (50 nM, excitation 633 nm, emission <650 nm) or tetramethyl rhodamine methyl ester (100 nM, excitation 543 nm, emission >600 nm) at 37°C for 15 or 20 min, respectively. Cells were then washed and resuspended in standard buffer solution. |
| References |
[1]. Baumgartner H K, et al. Caspase-8-mediated apoptosis induced by oxidative stress is independent of the intrinsic pathway and dependent on cathepsins[J]. American Journal of Physiology-Gastrointestinal and Liver Physiology, 2007, 293(1): G296-G307. |
| Additional Infomation | Cell-death programs executed in the pancreas under pathological conditions remain largely undetermined, although the severity of experimental pancreatitis has been found to depend on the ratio of apoptosis to necrosis. We have defined mechanisms by which apoptosis is induced in pancreatic acinar cells by the oxidant stressor menadione. Real-time monitoring of initiator caspase activity showed that caspase-9 (66% of cells) and caspase-8 (15% of cells) were activated within 30 min of menadione administration, but no activation of caspase-2, -10, or -12 was detected. Interestingly, when caspase-9 activation was inhibited, activation of caspase-8 was increased. Half-maximum activation (t0.5) of caspase-9 occurred within ∼2 min and was identified at or in close proximity to mitochondria, whereas t0.5 for caspase-8 occurred within ∼26 min of menadione application and was distributed homogeneously throughout cells. Caspase-9 but not caspase-8 activation was blocked completely by the calcium chelator BAPTA or bongkrekic acid, an inhibitor of the mitochondrial permeability transition pore. In contrast, caspase-8 but not caspase-9 activation was blocked by the destruction of lysosomes (preincubation with Gly-Phe β-naphthylamide, a cathepsin C substrate), loss of lysosomal acidity (bafilomycin A1), or inhibition of cathepsin L or D. Using pepstatin A-BODIPY FL conjugate, we confirmed translocation of cathepsin D out of lysosomes in response to menadione. We conclude that the oxidative stressor menadione induces two independent apoptotic pathways within pancreatic acinar cells: the classical mitochondrial calcium-dependent pathway that is initiated rapidly in the majority of cells, and a slower, caspase-8-mediated pathway that depends on the lysosomal activities of cathepsins and is used when the caspase-9 pathway is disabled.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.6598 mL | 3.2992 mL | 6.5984 mL | |
| 5 mM | 0.1320 mL | 0.6598 mL | 1.3197 mL | |
| 10 mM | 0.0660 mL | 0.3299 mL | 0.6598 mL |