Y-39983 (also called Y-33075) is a novel, potent and selective inhibitor of Rho-associated coiled coil-forming protein kinase( ROCK ) with IC50 values of 3.6 nM, 0.42 μM and 0.81 μM for ROCK, PKC and CaMKII, respectively. Y-39983 acts by downregulating RhoA/Rho-associated kinase expression during its promotion of axonal regeneration. Y-39983 also promotes regeneration of crushed axons of retinal ganglion cells into the optic nerve of adult cats. Y-39983 has been reported to relax the ciliary arteries of precontracted isolated rabbit in vitro. In addition, Y-39983 has been found to increase optic-nerve-head blood flow by laser speckle flowmetry. Moreover, Y-39983 lowered the intraocular pressure ( IOP) in a dose-dependent fashion in the eyes of rabbits and monkeys.
Physicochemical Properties
| Molecular Formula | C16H16N4O | |
| Molecular Weight | 280.33 | |
| Exact Mass | 280.132 | |
| Elemental Analysis | C, 68.55; H, 5.75; N, 19.99; O, 5.71 | |
| CAS # | 199433-58-4 | |
| Related CAS # | Y-33075 dihydrochloride;173897-44-4;Y-33075 hydrochloride;471843-75-1 | |
| PubChem CID | 9810884 | |
| Appearance | White to off-white solid powder | |
| Density | 1.32 | |
| Boiling Point | 444.644ºC at 760 mmHg | |
| Flash Point | 222.713ºC | |
| LogP | 3.608 | |
| Hydrogen Bond Donor Count | 3 | |
| Hydrogen Bond Acceptor Count | 3 | |
| Rotatable Bond Count | 3 | |
| Heavy Atom Count | 21 | |
| Complexity | 367 | |
| Defined Atom Stereocenter Count | 1 | |
| SMILES | O=C(NC1=C2C(NC=C2)=NC=C1)C3=CC=C(C=C3)[C@H](N)C |
|
| InChi Key | JTVBXQAYBIJXRP-SNVBAGLBSA-N | |
| InChi Code | InChI=1S/C16H16N4O/c1-10(17)11-2-4-12(5-3-11)16(21)20-14-7-9-19-15-13(14)6-8-18-15/h2-10H,17H2,1H3,(H2,18,19,20,21)/t10-/m1/s1 | |
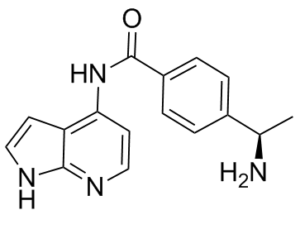
| Chemical Name | 4-[(1R)-1-aminoethyl]-N-(1H-pyrrolo[2,3-b]pyridin-4-yl)benzamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | ROCK (IC50 = 3.6 nM); PKC (IC50 = 420 nM); CaMKII (IC50 = 810 nM) | ||
| ln Vitro | At 3.6 nM for IC50, Y-33075, also known as Y-39983, is a strong inhibitor of ROCK. More potently than Y-27632, Y-33075 inhibits PKC and CaMKII. In contrast, Y-27632 and Y-33075 have IC50s of 26 μM and 0.81 μM against CaMKII and 9.0 μM and 0.42 μM, respectively, against PKC. Comparing Y-27632 and Y-33075 against PKC, their IC50s are 82 and 117 times greater than ROCK, respectively, but against CaMKII, they are 236 and 225 times greater than ROCK, respectively [1]. In comparison to neurites in RGCs not receiving Y-39983 therapy, Y-33075 (Y-39983, 10 μM) lengthens them in RGCs [2]. Rabbit ciliary artery segments in a Ca2+-free solution are not able to contract due to histamine when exposed to Y-33075 (Y-39983, 1 μM). In solutions with high potassium (high K), Y-33075 (10 μM) does not affect the rise in [Ca2+]i [3]. | ||
| ln Vivo |
Y-39983 (≥0.01%) dramatically reduces intraocular pressure (IOP) in rabbits two hours after topical application. IOP significantly decreases in monkeys' treated eyes with Y-39983 (0.05%) between two and seven hours after topical application. management[1]. Rat eyes with retinal ganglion cells (RGCs) have more regenerating axons when exposed to 100 μM Y-39983[2]. A biochemical assay showed that Y-39983 inhibited ROCK more potently than Y-27632. In rabbits, topical administration of Y-39983 significantly increased conventional outflow by 65.5%, followed by significant, dose-dependent reduction in IOP. Maximum IOP reduction was 13.2 +/- 0.6 mm Hg (mean +/- SE) at 0.1% Y-39983 in rabbits. In monkeys, at 3 hours after topical administration of 0.05% Y-39983, maximum reduction of IOP was 2.5 +/- 0.8 mm Hg. No serious side effects were observed in ocular tissues except sporadic punctate subconjunctival hemorrhage during long-term topical administration of Y-39983 four times a day (at 2-hour intervals) in rabbits or monkeys. However, punctate subconjunctival hemorrhage was not observed with administration twice daily (at a 6-hour interval) or three times a day (at 5-hour intervals).[1] Topical administration of 0.05% Y-39983 solution significantly increased blood flow in ONH compared with the vehicle group in rabbits. Maximum increase in blood flow in the 0.05% Y-39983 group was 122.84 ± 5.98 % (Mean ± S.E.) at 90 minutes after administration compared with before administration. Neurites in rat RGCs treated with 10 μM Y-39983 were extended compared with those without Y-39983 treatment of RGCs in vitro. Y-39983 dose-dependently increased the number of RGCs with regenerating axons in vivo. The numbers of RGCs with regenerating axons in 10 and 100 μM Y-39983-treated rats were 99.3 ± 10.5 and 169.5 ± 43.3 cells/mm(2) (Mean ± S.D.), respectively, and significantly increased compared with those in saline-treated rats (43.3 ± 6.0 cells/mm(2)).[2] |
||
| Enzyme Assay | Recombinant ROCK (ROK α/ROCK II) and purified protein kinase C (PKC: mixture of α, β, γ isoforms) were purchased from Upstate Biotechnology. Recombinant calmodulin-dependent protein kinase II (CaMK II) was purchased from Daiichi Pure Chemical. ROCK (0.2 U/mL) was incubated with 1 μM [γ-32P] ATP and 10 μg/mL histone as substrates in the absence or presence of various concentrations of Y-27632, Y-39983, or staurosporine at room temperature for 20 minutes in 20 mM MOPS (3-(N-morpholino)propanesulfonic acid) buffer (pH 7.2) containing 0.1 mg/mL bovine serum albumin (BSA), 5 mM dithiothreitol [DTT], 10 mM β-glycerophosphate, 50 μM Na3VO4, and 10 mM MgCl2 in a total volume of 100 μL. PKC (10 ng/mL) was incubated with 1 μM [γ-32P] ATP and 20 μM PKC substrate in the absence or presence of various concentrations of Y-27632, Y-39983, or staurosporine at room temperature for 30 minutes in 20 mM MOPS buffer (pH 7.5) containing 0.1 mg/mL BSA, 10 mM DTT, 10 mM β-glycerophosphate, 50 μM Na3VO4, 2 mM CaCl2, 20 μg/mL phosphatidyl-l-serine, and 10 mM MgCl2 in a total volume of 100 μL. CaMK II (125 U/mL) was incubated with 1 μM [γ-32P] ATP, 10 μM calmodulin, and 20 μM CaMK II substrate, in the absence or presence of various concentrations of Y-27632, Y-39983, or staurosporine at room temperature for 30 minutes in 20 mM MOPS buffer (pH 7.5) containing 0.2 mg/mL BSA, 0.5 mM DTT, 0.1 mM β-glycerophosphate, 50 μM Na3VO4, 1 mM CaCl2, and 5 mM MgCl2 in a total volume of 100 μL. Incubation was terminated by the addition of 100 μL of 0.7% phosphoric acid. A 160 μL portion of the mixture was transferred to Multiscreen-PH plate (Millipore, MA). A positively charged phosphocellulose filter absorbed the substrate that bound 32P (Multiscreen-Vacuum manifold; Millipore). The filter was washed with 300 μL of 0.5% phosphoric acid and then twice with purified water and then dried. The radioactivity of the dried filter was measured with a liquid scintillation counter. Results are presented as 50% inhibitory concentrations and 95% confidence intervals (CIs).[1] | ||
| Cell Assay | Human umbilical venous endothelial cells (HUVECs) were purchased from Dainippon Pharmaceutical (Osaka, Japan). HUVECs were cultured in CS-C medium (Dainippon Pharmaceutical) and maintained in a 95% air-5% CO2 atmosphere at 37°C and passaged using the trypsin-EDTA method. HUVECs were seeded into 24-well plates. After seeding, HUVECs were incubated in medium containing 1 μM Y-39983 for 15 or 30 minutes and observed by phase-contrast microscopy. Medium was then removed, and HUVECs were incubated in medium without Y-39983 for 1 hour to evaluate recovery from the morphologic changes induced by Y-39983[1]. | ||
| Animal Protocol |
|
||
| References |
[1]. Effects of Topical Administration of Y-39983, a Selective Rho-Associated Protein Kinase Inhibitor, on Ocular Tissues in Rabbits and Monkeys Invest. Ophthalmol. Vis. Sci. July 2007 vol. 48no. 7 3216-3222. [2]. Effects of Y-39983, a selective Rho-associated protein kinase inhibitor, on blood flow in optic nerve head in rabbits and axonal regeneration of retinal ganglion cells in rats. Curr Eye Res. 2011 Oct;36(10):964-70. [3]. Effects of Rho-associated protein kinase inhibitors Y-27632 and Y-39983 on isolated rabbit ciliary arteries.Jpn J Ophthalmol. 2011 Jul;55(4):411-7. Epub 2011 Jun 11. |
||
| Additional Infomation |
4-[(1R)-1-aminoethyl]-N-(1H-pyrrolo[2,3-b]pyridin-4-yl)benzamide is a pyrrolopyridine. See also: Y-39983 (annotation moved to). |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.92 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (8.92 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (8.92 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: Saline: 30 mg/mL (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.5672 mL | 17.8361 mL | 35.6722 mL | |
| 5 mM | 0.7134 mL | 3.5672 mL | 7.1344 mL | |
| 10 mM | 0.3567 mL | 1.7836 mL | 3.5672 mL |