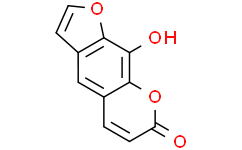
Physicochemical Properties
| Molecular Formula | C₁₁H₆O₄ |
| Molecular Weight | 202.16 |
| Exact Mass | 202.026 |
| CAS # | 2009-24-7 |
| PubChem CID | 65090 |
| Appearance | White to off-white solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 428.1±45.0 °C at 760 mmHg |
| Melting Point | 250ºC |
| Flash Point | 212.7±28.7 °C |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.711 |
| LogP | 0.72 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 15 |
| Complexity | 312 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | JWVYQQGERKEAHW-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C11H6O4/c12-8-2-1-6-5-7-3-4-14-10(7)9(13)11(6)15-8/h1-5,13H |
| Chemical Name | 9-hydroxy-7H-furo[3,2-g][1]benzopyran-7-one |
| Synonyms | 8-HydroxypsoralenNSC 401269Xanthotoxol Xanthotol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Enzyme Assay |
Human liver microsomal metabolism assay of Xanthotoxol: The incubation system was composed of human liver microsomes, Xanthotoxol (final concentrations: 0.5-20 μM), NADPH-generating system (as coenzyme), and phosphate buffer. The mixture was pre-incubated at 37°C for 5 minutes, then the reaction was initiated by adding the NADPH-generating system and incubated at 37°C for 0-60 minutes. The reaction was terminated by adding ice-cold acetonitrile containing an internal standard. After centrifugation, the supernatant was analyzed by HPLC-MS/MS to quantify Xanthotoxol and identify its metabolites. [1] CYP450 isoform inhibition assay for metabolic pathway identification: Selective inhibitors of major CYP450 isoforms (CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP3A4) were separately added to the human liver microsomal incubation system at their respective specific concentrations. After pre-incubating with microsomes for 15 minutes, Xanthotoxol and the NADPH-generating system were added to initiate the reaction. The residual amount of Xanthotoxol was detected by HPLC-MS/MS to evaluate the contribution of each CYP450 isoform to Xanthotoxol metabolism. [1] Metabolic inhibition kinetic assay: Xanthotoxol (0.5-10 μM) was incubated with human liver microsomes and NADPH-generating system in the presence of serial concentrations of specific CYP450 inhibitors (e.g., ketoconazole for CYP3A4). The reaction was terminated and analyzed as described above. The inhibition constant (Ki) and IC₅₀ value were calculated by fitting the data to appropriate kinetic models to determine the type and potency of inhibition. [1] |
| ADME/Pharmacokinetics |
In vitro metabolism in human liver microsomes: Xanthotoxol was rapidly metabolized in human liver microsomes, with a metabolic half-life (t₁/₂) of 18.3 minutes. Two major metabolites were identified: M1 (a monohydroxylated derivative) and M2 (a glucuronide conjugate), with M1 being the predominant metabolite (accounting for ~65% of total metabolites). [1] CYP450 isoform contribution: CYP3A4 was the major isoform mediating Xanthotoxol metabolism (contribution ~58%), followed by CYP2C9 (~22%) and CYP2C19 (~12%); CYP1A2 and CYP2D6 had negligible contributions (<5% each). [1] Metabolic kinetics parameters: The Michaelis-Menten constant (Km) for Xanthotoxol metabolism in human liver microsomes was 8.7 μM, and the maximum reaction rate (Vmax) was 125.6 pmol/min/mg protein. [1] Metabolic inhibition potential: Xanthotoxol exhibited competitive inhibition of CYP3A4 with an IC₅₀ value of 12.4 μM and a Ki value of 4.2 μM; it had no significant inhibitory effect on CYP1A2, CYP2C9, CYP2C19, or CYP2D6 (IC₅₀ > 50 μM). [1] |
| References |
[1]. Metabolism and Metabolic Inhibition of Xanthotoxol in Human Liver Microsomes. Evid Based Complement Alternat Med. 2016;2016:5416509. |
| Additional Infomation |
Xanthotoxol is an 8-hydroxyfurocoumarin. Xanthotoxol has been reported in Angelica japonica, Pleurospermum rivulorum, and other organisms with data available. Xanthotoxol is a natural furanocoumarin compound isolated from plants of the Zanthoxylum genus (e.g., Zanthoxylum bungeanum Maxim.), which has been used in traditional Chinese medicine for its anti-inflammatory, analgesic, and anti-tumor potential. [1] The study focused on the in vitro metabolism of Xanthotoxol in human liver microsomes, clarifying its metabolic pathways, key metabolizing enzymes, and metabolic inhibition characteristics, which provides important experimental basis for predicting its in vivo pharmacokinetic behavior and potential drug-drug interactions in clinical applications. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~494.66 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (12.37 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (12.37 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.9466 mL | 24.7329 mL | 49.4658 mL | |
| 5 mM | 0.9893 mL | 4.9466 mL | 9.8932 mL | |
| 10 mM | 0.4947 mL | 2.4733 mL | 4.9466 mL |