Physicochemical Properties
| Molecular Formula | C17H17CLFN3O2S |
| Molecular Weight | 381.85218501091 |
| Exact Mass | 381.071 |
| CAS # | 1957202-44-6 |
| Related CAS # | Vonoprazan;881681-00-1;Vonoprazan Fumarate;881681-01-2 |
| PubChem CID | 141508909 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 25 |
| Complexity | 511 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | OJGGYCBXTRFIGZ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C17H16FN3O2S.ClH/c1-19-10-13-9-17(15-6-2-3-7-16(15)18)21(12-13)24(22,23)14-5-4-8-20-11-14;/h2-9,11-12,19H,10H2,1H3;1H |
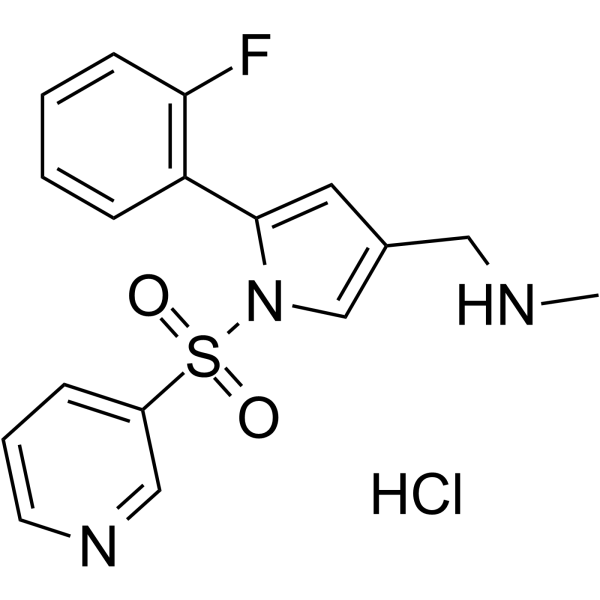
| Chemical Name | 1-[5-(2-fluorophenyl)-1-pyridin-3-ylsulfonylpyrrol-3-yl]-N-methylmethanamine;hydrochloride |
| Synonyms | TAK-438 HCl; TAK438 hydrochloride |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | H+/K+-ATPase (IC50 = 19 nM) |
| ln Vitro | The concentration-dependent pig stomach H+, K+-ATPase activity is demonstrated by vonoprazan (0.1 nM-10 μM; 30 minutes) [2]. Even at 500-fold higher concentrations than its IC50 value for gastric H+,K+-ATPase activity, vonoprazan does not impede Na+,K+-ATPase activity [2]. |
| ln Vivo | Rats who receive a dose of 4 mg/kg of vonoprazan (1-4 mg/kg; po) are entirely protected against stomach acid secretion triggered by basal and 2-deoxy-D-glucose (200 mg/kg; sc) [2]. |
| Enzyme Assay |
Proton Potassium Adenosine Triphosphatase (H+,K+-ATPase) Inhibitory Activity Test [1] Accordinpg to the method of Wallmark et al., a gastric mucosal membrane microsomal fraction was prepared from the stomach of swine. First, the stomach was removed, washed with tap water, and immersed in 3 mol/L brine, and the surface of the mucosal membrane was wiped with a paper towel. The gastric mucosal membrane was detached, chopped, and homogenized in a 0.25 mol/L saccharose solution (pH 6.8) containing 1 mmol/L EDTA and 10 mmol/L tris-hydrochloric acid using polytron (Kinematica). The obtained homogenate was centrifuged at 20000g for 30 min and the supernatant was centrifuged at 100000g for 90 min. The precipitate was suspended in 0.25 mol/L saccharose solution, superimposed on a 0.25 mol/L saccharose solution containing 7.5% Ficoll, and centrifuged at 100000g for 5 h. The fraction containing the interface between the both layers was recovered, and centrifugally washed with 0.25 mol/L saccharose solution. The obtained microsomal fraction was used as a proton, potassium adenosine triphosphatase standard product. To 40 μL of a 50 mmol/L HEPES-Tris buffer (5 mmol/L magnesium chloride, 10 mmol/L potassium chloride, 10 μmol/L valinomycin, pH 6.5) containing 2.5 μg/mL (based on the protein concentration) of the enzyme standard product was added a test compound (5 μL) dissolved in a 10% aqueous dimethyl sulfoxide solution, and the mixture was incubated at 37 °C for 30 min. The enzyme reaction was started by adding 5 μL of a 2 mmol/L adenosine triphosphate Tris salt solution (50 mmol/L HEPES-Tris buffer (5 mmol/L magnesium chloride, pH 6.5)). The enzyme reaction was carried out at 37 °C for 20 min, and 15 μL of a malachite green solution (0.12% malachite green solution in sulfuric acid (2.5 mol/L), 7.5% ammonium molybdate, and 11% Tween 20 were mixed at a ratio of 100:25:2) was added to quench the reaction. After the mixture was allowed to stand at room temperature for 15 min, the resulting reaction product of inorganic phosphorus with malachite green was colorimetrically determined at a wavelength of 610 nm. In addition, the amount of the inorganic phosphoric acid in the reaction solution free of potassium chloride was measured in the same manner, which was subtracted from the inorganic phosphoric acid amount in the presence of potassium chloride to determine the H+,K+-ATPase activity. The inhibitory rate (%) was determined from the activity value of the control and the activity values of various concentrations of the test compound, and the 50% inhibitory concentration (IC50) of the H+,K+-ATPase activity was determined. |
| Animal Protocol |
Animal/Disease Models: Male 7 or 8 week old SD (SD (Sprague-Dawley)) rat [2] Doses: 0.5 mg/kg, 1 mg/kg, 2 mg/kg, 4 mg/kg Route of Administration: Oral Experimental Results: Suppression of basal gastric acid at dose secreted in a dependent manner. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation
◉ Summary of Use during Lactation No information is available on the clinical use of vonoprazan during breastfeeding. Because of liver damage that occurred in nursing rodents, the manufacturer recommends that nursing mothers should pump and discard human milk while taking and for 2 days after the last dose. An alternate drug may be preferred. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding In healthy subjects, the plasma protein binding of vonoprazan ranges from 85% to 88%. At plasma concentrations between 0.1 and 10 mcg/mL, the plasma protein binding of vonoprazan is independent of concentration. |
| References |
[1]. Discovery of a novel pyrrole derivative 1-[5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamine fumarate (TAK-438) as a potassium-competitive acid blocker (P-CAB). J Med Chem, 2012, 55(9), 4446-4456. [2]. 1-[5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamine monofumarate (TAK-438), a novel and potent potassium-competitive acid blocker for the treatment of acid-related diseases. J Pharmacol Exp Ther, 2010, 335(1),. [3]. Role of Vonoprazan in Helicobacter pylori Eradication Therapy in Japan. Front Pharmacol. 2019 Jan 15;9:1560. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6188 mL | 13.0941 mL | 26.1883 mL | |
| 5 mM | 0.5238 mL | 2.6188 mL | 5.2377 mL | |
| 10 mM | 0.2619 mL | 1.3094 mL | 2.6188 mL |