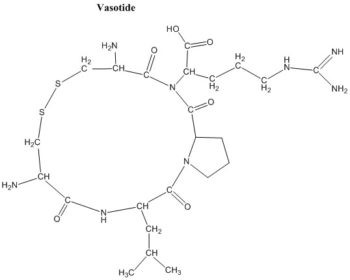
Vasotide, D(Cys-Leu-Pro-Arg-Cys) [abbreviated as D(CLPRC)], is a novel and potent peptidomimetic compound that targets two retinal VEGF receptors (VEGFR-1 and VEGFR-2) and reduces pathological angiogenesis in murine and nonhuman primate models of retinal disease. Blood vessel growth from preexisting vessels (angiogenesis) underlies many severe diseases including major blinding retinal diseases such as retinopathy of prematurity (ROP) and aged macular degeneration (AMD). This observation has driven development of antibody inhibitors that block a central factor in AMD, named vascular endothelial growth factor (VEGF), from binding to its receptors VEGFR-1 and VEGFR-2. However, some patients are insensitive to current anti-VEGF drugs or develop resistance, and the required repeated intravitreal injection of these large molecules is costly and clinically problematic. Vasotide inhibits retinal angiogenesis by binding selectively to the VEGF receptors, VEGFR-1 and Neuropilin-1 (NRP-1). Delivery of Vasotide in eye drops or via intraperitoneal injection in a laser-induced monkey model of human wet AMD, a mouse genetic knockout model of the AMD subtype called retinal angiomatous proliferation (RAP), and a mouse oxygen-induced model of retinopathy of prematurity (ROP) markedly decreased retinal angiogenesis in all three animal models. This prototype drug candidate is a promising new dual receptor inhibitor of the VEGF ligand with potential for translation into safer, less invasive applications to combat pathological angiogenesis in retinal disorders.
Physicochemical Properties
| Molecular Formula | C₂₇H₂₉CL₂N₇O₂S |
| Molecular Weight | 586.54 |
| Appearance | Typically exists as solid at room temperature |
| SMILES | O=C(NCC1=CC=C(S(C2=CC(F)=CC(F)=C2)(=O)=O)C=C1)C3=CN4C(C=C3)=NC=C4.Cl |
| Synonyms | Vasotide; D(Cys-Leu-Pro-Arg-Cys); D(CLPRC) |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
VEGF receptors Researchers have evaluated a small cyclic retro-inverted peptidomimetic, D(Cys-Leu-Pro-Arg-Cys) [D(CLPRC)], and hereafter named Vasotide, that inhibits retinal angiogenesis by binding selectively to the VEGF receptors VEGFR-1 and neuropilin-1 (NRP-1). [1] |
| ln Vitro |
Researchers have evaluated a small cyclic retro-inverted peptidomimetic, D(Cys-Leu-Pro-Arg-Cys) [D(CLPRC)], and hereafter named Vasotide, that inhibits retinal angiogenesis by binding selectively to the VEGF receptors VEGFR-1 and neuropilin-1 (NRP-1). [1] The cyclic retro-inverted peptidomimetic derivative of CPQPRPLC, named Vasotide (p(CLPRC)), was shown to be a stronger ligand for VEGFR-1 and NRP-1 than the original linear peptide sequence and had enhanced stability in vivo. [1] NMR analyses identified Arg-Pro-Leu (RPL) as a minimal necessary and sufficient tripeptide motif for dual ligand-binding to VEGFR-1 and NRP-1 receptors. [1] |
| ln Vivo |
Vasotide reduces vascular tuft formation in ROP mice. Vasotide reduced angiogenesis in a laser-induced monkey model of AMD. Vasotide administered i.p. reduced angiogenesis in the mouse vldlr model of RAP. The VEGF receptor-targeted prototype peptidomimetic Vasotide can reduce pathological angiogenesis in two preclinical murine models and one non-human primate model of human retinal diseases: the mouse ROP model, the vldlr knockout mouse model of retinal angiogenesis proliferation (RAP), and the laser-induced monkey model of wet AMD. Each disease type displays both common and unique angiogenic features. This suggests that, like the currently FDA-approved anti-VEGF therapeutics, Vasotide may be valuable for a range of human retinal diseases involving angiogenesis. Indeed, our results with Vasotide are roughly similar to the therapeutic response to bevacizumab previously reported in eyes of animals of the same primate colony comparably lesioned by laser-induced photocoagulation. More elaborate pharmacokinetic/therapeutic analysis of Vasotide is required to identify additional characteristics such as duration of efficacy, safety, convenience and cost. [1] Delivery of Vasotide via either eye drops or intraperitoneal injection in a laser-induced monkey model of human wet AMD, a mouse genetic knockout model of the AMD subtype called retinal angiomatous proliferation (RAP), and a mouse oxygen-induced model of ROP decreased retinal angiogenesis in all three animal models. This prototype drug candidate is a promising new dual receptor inhibitor of the VEGF ligand with potential for translation into safer, less-invasive applications to combat pathological angiogenesis in retinal disorders. [1] In a mouse oxygen-induced retinopathy (OIR) model of retinopathy of prematurity (ROP), administration of Vasotide either intraperitoneally (i.p.) or via eye drops during the hypoxic phase (P12-P18) significantly decreased the formation of pathological vascular tufts projecting into the vitreous chamber at P19, compared to control peptide-treated mice. The retinal avascular areas were not significantly affected. [1] In a laser-induced choroidal neovascularization (CNV) model in African green monkeys (a model for wet age-related macular degeneration, AMD), topical eye drop administration of Vasotide (6 mg in 50 µl GelTears vehicle, twice daily for 5 days, then once daily for 16 days) significantly reduced the incidence and size of clinically relevant CNV lesions (grade III/IV) and decreased late-phase fluorescein leakage at day 29 post-laser, compared to vehicle-treated controls. Histological analysis at day 34 showed a dramatic reduction in inflammatory cell infiltration (macrophages and granulocytes), CNV, and fibroplasia. [1] In a vldlr knockout mouse model of retinal angiomatous proliferation (RAP, an AMD subtype), i.p. injection of Vasotide (40 µg/g body weight, daily at specified periods) significantly reduced pathological angiogenesis. This included a reduction in vascular density in both the inner and outer (normally avascular) retinal halves, decreased number and size of vascular tufts, and prevention of blood vessel growth into the photoreceptor cell layers and across the retinal pigment epithelium compared to control peptide-treated mice. The therapeutic effect was sustained across multiple treatment periods up to P270. [1] |
| Enzyme Assay |
A subtractive bacteriophage display-library screening strategy was initially used to identify the peptide CPQPRPLC as a selective ligand to VEGFR-1 and NRP-1. [1] Subsequent structural analyses (e.g., NMR) were conducted to define the minimal binding motif. [1] |
| Cell Assay |
Immunohistochemical and immunofluorescence analyses were performed on retinal tissues. Blood vessels were stained with fluorescently conjugated isolectin IB4 (which binds to endothelial cells). Co-staining with antibodies against CD31 (endothelial cell marker), CD68 or F4/80 (macrophage markers), and GR1 (granulocyte marker) was used to identify specific cell types and assess inflammation. Tissues were imaged using fluorescence microscopy and confocal microscopy. [1] For retinal whole mounts, eyes were fixed, retinas were dissected and stained with fluorescent IB4, often made transparent with an organic iodine agent, and then flat-mounted for imaging. Image stacks were captured through the full retinal thickness for three-dimensional analysis. [1] |
| Animal Protocol |
Eye drop administration of drugs to monkeys [1] A topical formulation of the Vasotide peptide was administered at a concentration of 120 mg/ml. Following laser photocoagulation on day 1, the test article, Blink Gel Tears, referred to in this text as GelTears, was administered topically by pipette onto both right and left eyes (50 µl) twice daily for 5 days, then once daily for 16 days. The vehicle alone (50 µl) was delivered in an identical fashion. Treatment regimen was as defined in Table S1. Monkeys were dosed while in a supine position. Mouse vldlr-null (knockout) model of the human Retinal Angiomatous Proliferation (RAP) disorder [1] Breeding pairs of mutant mice with targeted deletion of the vldlr gene (B6;129S7-Vldlrtm1Her/J; vldlr KO) were maintained and bred in standardized conditions. Age-matched C57BL/6J mice were used as normal wildtype controls. Vldlr KO mice were randomly divided into three groups: vldlr KO without treatment, vldlr KO given control peptide D(CAPAC), and vldlr KO given the therapeutic peptide Vasotide. We injected intraperitoneally (i.p.) 20 µg peptide/µl PBS, 40 µg/g body weight, daily at P12-P18, P48-P54, P108-P114 and P208-P214. Mice were maintained according to the Association for Research in Vision and Ophthalmology statement on animal usage in ophthalmic research. Mouse OIR Model: C57BL/6J mouse pups with their mothers were placed in 75% oxygen from postnatal day 7 (P7) to P12 to suppress normal retinal vascular development. On P12, they were returned to room air (21% O2), initiating a hypoxic phase that promotes pathological angiogenesis. Drug treatment was administered during P12-P18. Vasotide or control peptide was delivered either by intraperitoneal injection (8 µg/g or 40 µg/g body weight, daily) or via topical eye drops (200 µg in 2 µl Systane vehicle, three times daily). Retinas were analyzed at P19. [1] Monkey Laser-Induced CNV Model: Adult male African green monkeys received laser photocoagulation (six spots per eye) to rupture Bruch's membrane and induce CNV. Starting day 1 post-laser, monkeys received topical eye drops containing either Vasotide (120 mg/ml, 50 µl per eye) in a GelTears vehicle or vehicle alone. Dosing was twice daily for 5 days, then once daily for days 6-21. Ophthalmic imaging (fluorescein angiography, optical coherence tomography) was performed at day 29, and histopathology was assessed at day 34. [1] Mouse vldlr Knockout RAP Model: Vldlr-null mice and wildtype controls were used. Vasotide or control peptide D(CAPAC) was administered via intraperitoneal injection (40 µg/g body weight in PBS) daily during specified treatment periods: P12-P18, P48-P54, P108-P114, and P208-P214. Retinas were collected and analyzed at multiple time points from P16 to P270. [1] |
| ADME/Pharmacokinetics |
Preliminary data from prior studies indicated that the linear tripeptide p(LPR) (a precursor) administered in eye drops could transfer into the vitreous chamber of the mouse eye and directly contact the inner retinal surface where pathological tufts form. [1] The cyclic retro-inverted peptidomimetic Vasotide was designed for enhanced stability in vivo compared to its linear counterparts. [1] The manuscript notes that more elaborate pharmacokinetic/therapeutic analysis is required to identify additional characteristics such as duration of efficacy. [1] |
| Toxicity/Toxicokinetics |
In the monkey model, minor clouding of the inferior cornea was observed in both treatment and vehicle groups, attributed to the low pH of the topical formulation. This did not impair fundoscopy or image analysis. [1] |
| References |
[1]. The peptidomimetic Vasotide targets two retinal VEGF receptors and reduces pathological angiogenesis in murine and nonhuman primate models of retinal disease. Sci Transl Med . 2015 Oct 14;7(309):309ra165. [2]. The potential of anti-VEGF (Vasotide) by eye drops to treat proliferative retinopathies. Ann Transl Med . 2016 Oct;4(Suppl 1):S41. d |
| Additional Infomation |
A suggested new treatment for blinding retinal diseases
This paper describes a novel small drug candidate named Vasotide™, made of engineered amino acids, that blocks abnormal overgrowth of blood vessels in the eye’s retina, leading to loss of vision. A distinctive feature of this drug is its delivery in simple eye drops. Vasotide uniquely prevents a blood vessel growth-promoting molecule called VEGF from attaching to two different receptor molecule types on endothelial cells, which line the inside of blood vessels, thereby inhibiting the pathology in animal models of major retinal diseases called aged macular degeneration, retinopathy of prematurity, and possibly the untested but similarly caused diabetic retinopathy. [1] Blood vessel growth from preexisting vessels (angiogenesis) underlies many severe diseases including major blinding retinal diseases such as retinopathy of prematurity (ROP) and aged macular degeneration (AMD). This observation has driven development of antibody inhibitors that block a central factor in AMD, vascular endothelial growth factor (VEGF), from binding to its receptors VEGFR-1 and mainly VEGFR-2. However, some patients are insensitive to current anti-VEGF drugs or develop resistance, and the required repeated intravitreal injection of these large molecules is costly and clinically problematic.[1] Collectively, the findings presented by Sidman and colleagues, indicate the potential of agents such as Vasotide, which inhibit VEGFR-1 and NRP-1 for the treatment of a variety of ocular diseases featuring neovascularization. Vasotide was not directly compared to current United States Food and Drug Administration (FDA)-approved agents such as aflibercept, ranibizumab and bevacizumab, which target VEGF-A and VEGFR-2. This assessment is important for not only understanding the relative efficacy of Vasotide, but also which components of the VEGF pathway are most critical for attenuating ocular neovascularization, findings that may be particularly relevant to patients resistant to current anti-VEGF agents. Perhaps one of the most promising aspects for the future treatment of ocular neovascularization and vascular permeability is topical administration. If penetrance to the retina and choroid can be confirmed and then optimized, and robust pharmacokinetic and safety data obtained, eye drops would be preferable to currently monthly intravitreal injections and may result in significant economic savings.[2] Vasotide is a small cyclic retro-inverted peptidomimetic, p(Cys-Leu-Pro-Arg-Cys) or p(CLPRC). It is a prototype drug candidate designed to inhibit pathological angiogenesis by blocking VEGF ligand binding to VEGFR-1 and NRP-1. [1] Its proposed mechanism of action is distinct from current anti-VEGF-A therapies (e.g., ranibizumab, bevacizumab) which primarily target VEGF-A binding to VEGFR-2. Vasotide may be beneficial for patients unresponsive or resistant to current therapies. [1] The study suggests Vasotide may also influence inflammatory and immune mechanisms involved in angiogenic retinopathies, as evidenced by reduced inflammatory infiltrates in treated monkeys. [1] Potential routes of administration include topical eye drops and intraperitoneal injection, with eye drops offering a potentially safer, less invasive, and more convenient alternative to repeated intravitreal injections. [1] The possibility of combination therapy with other anti-angiogenic agents acting via different mechanisms is discussed. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7049 mL | 8.5246 mL | 17.0491 mL | |
| 5 mM | 0.3410 mL | 1.7049 mL | 3.4098 mL | |
| 10 mM | 0.1705 mL | 0.8525 mL | 1.7049 mL |