Physicochemical Properties
| Molecular Formula | C10H10O5 |
| Molecular Weight | 210.1834 |
| Exact Mass | 210.053 |
| CAS # | 1081-71-6 |
| PubChem CID | 14124 |
| Appearance | White to yellow solid powder |
| Density | 1.374g/cm3 |
| Boiling Point | 402ºC at 760mmHg |
| Flash Point | 162.1ºC |
| Vapour Pressure | 3.49E-07mmHg at 25°C |
| Index of Refraction | 1.575 |
| LogP | 0.597 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 15 |
| Complexity | 250 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | YGQHQTMRZPHIBB-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C10H10O5/c1-15-9-5-6(2-3-7(9)11)4-8(12)10(13)14/h2-3,5,11H,4H2,1H3,(H,13,14) |
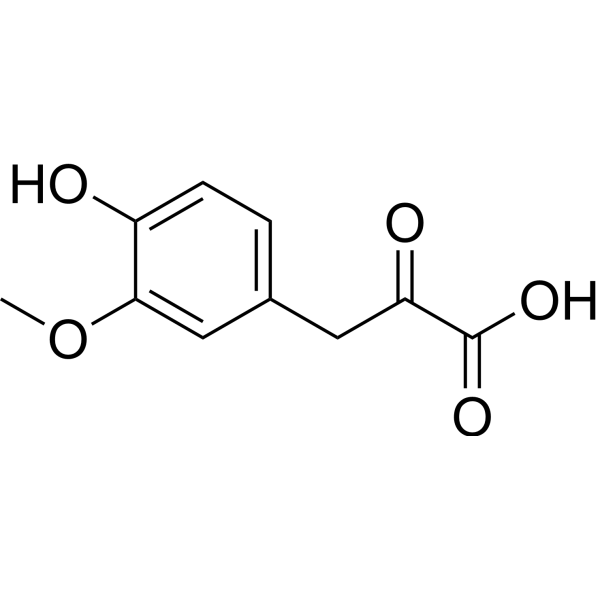
| Chemical Name | 3-(4-hydroxy-3-methoxyphenyl)-2-oxopropanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Catecholamines, dopamine, norepinephrine, and epinephrine are chemical neurotransmitters and hormones that regulate physiological processes as well as the onset and progression of neurological, psychiatric, endocrine, and cardiovascular illnesses [1]. The catecholamines, which include dopamine (3,4-dihydrophenylethylamine), norepinephrine (norepinephrine), and epinephrine (epinephrine), function as neurotransmitters or hormones both centrally and peripherally. Dopamine, the most prevalent monoamine neurotransmitter, is also found in non-neuronal tissues such as the gastrointestinal system and kidneys, where it regulates sodium balance [2]. |
| References |
[1]. Catecholamine metabolism: a contemporary view with implications for physiology and medicine. Pharmacol Rev. 2004 Sep;56(3):331-49. [2]. Liquid chromatographic methods for the quantification of catecholamines and their metabolites in several biological samples--a review. Anal Chim Acta. 2013 Mar 20;768:12-34. |
| Additional Infomation | Vanilpyruvic acid is a 2-oxo monocarboxylic acid that is 3,4-dihydroxyphenylpyruvic acid in which the hydroxy group at position 3 is substituted by a methoxy group. It has a role as a human urinary metabolite. It is a 2-oxo monocarboxylic acid, a monomethoxybenzene and a member of phenols. It is functionally related to a 3,4-dihydroxyphenylpyruvic acid. It is a conjugate acid of a vanilpyruvate. |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 27 mg/mL (~128.46 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (9.90 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.08 mg/mL (9.90 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.7578 mL | 23.7891 mL | 47.5783 mL | |
| 5 mM | 0.9516 mL | 4.7578 mL | 9.5157 mL | |
| 10 mM | 0.4758 mL | 2.3789 mL | 4.7578 mL |