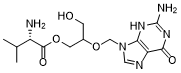
Valganciclovir (RS-79070-194; RS-079070-194) is the L-valyl ester prodrug of ganciclovir which is an antiviral agent. As a prodrug for ganciclovir, Valganciclovir exists as a mixture of two diastereomers. Unver in vivo conditions, the diastereomers are rapidly converted to ganciclovir by intestinal and hepatic esterases.
Physicochemical Properties
| Molecular Formula | C14H22N6O5 |
| Molecular Weight | 354.36168 |
| Exact Mass | 354.165 |
| CAS # | 175865-60-8 |
| Related CAS # | Valganciclovir hydrochloride;175865-59-5;Valganciclovir-d5 TFA;1402924-31-5 |
| PubChem CID | 135413535 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.6±0.1 g/cm3 |
| Boiling Point | 629.1±65.0 °C at 760 mmHg |
| Flash Point | 334.3±34.3 °C |
| Vapour Pressure | 0.0±1.9 mmHg at 25°C |
| Index of Refraction | 1.678 |
| LogP | -1.28 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 25 |
| Complexity | 528 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | N[C@@H](C(C)C)C(OCC(OCN1C=NC2=C1N=C(N)NC2=O)CO)=O |
| InChi Key | WPVFJKSGQUFQAP-GKAPJAKFSA-N |
| InChi Code | InChI=1S/C14H22N6O5/c1-7(2)9(15)13(23)24-4-8(3-21)25-6-20-5-17-10-11(20)18-14(16)19-12(10)22/h5,7-9,21H,3-4,6,15H2,1-2H3,(H3,16,18,19,22)/t8?,9-/m0/s1 |
| Chemical Name | [2-[(2-amino-6-oxo-1H-purin-9-yl)methoxy]-3-hydroxypropyl] (2S)-2-amino-3-methylbutanoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Is well absorbed from the gastrointestinal tract and the absolute bioavailability from valganciclovir tablets (following administration with food) is approximately 60% Valganciclovir. Is by renal excretion as ganciclovir through glomerular filtration and active tubular secretion The major route of elimination of valganciclovir. 0.703 ± 0.134 L/kg 3.07+/- 0.64 mL/min/kg [IV administration] 5.3 L/hr [Patient with creatinine clearance of 70.4 mL/min] Is by renal excretion as ganciclovir through glomerular filtration and active tubular secretion The major route of elimination of valganciclovir. Of intravenously administered ganciclovir was 3.07 + or - 0.64 mL/min/kg (n=68) while renal clearance was 2.99 + or - 0.67 mL/min/kg (n=16) Systemic clearance. Due to the rapid conversion of valganciclovir to ganciclovir, plasma protein binding of valganciclovir was not determined. Of ganciclovir is 1% to 2% over concentrations of 0.5 and 51 ug/mL Plasma protein binding. When ganciclovir was administered intravenously, the steady-state volume of distribution of ganciclovir was 0.703 + or - 0.134 L/kg (n=69). After administration of Valcyte tablets, no correlation was observed between ganciclovir AUC and reciprocal weight; oral dosing of Valcyte tablets according to weight is not required. When Valcyte tablets were administered with a high fat meal containing approximately 600 total calories (31.1 g fat, 51.6 g carbohydrates and 22.2 g protein) at a dose of 875 mg once daily to 16 HIV-positive subjects, the steady-state ganciclovir AUC increased by 30% (95% CI 12% to 51%), and the Cmax increased by 14% (95% CI -5% to 36%), without any prolongation in time to peak plasma concentrations (Tmax). Should be administered with food Valcyte. Of ganciclovir following oral administration of valganciclovir is about tenfold higher than that following oral administration of ganciclovir (60 versus 5.6%, respectively) The absolute bioavailability. From pharmacokinetic studies in adults indicate that oral administration of valganciclovir 900 mg once daily with food provides a mean area under the plasma concentration-time curve 0-24 hour (AUC0-24 hour) for ganciclovir comparable to that following IV ganciclovir 5 mg/kg once daily and exceeding that following oral ganciclovir 1 g 3 times daily with food Results. However, at these dosages, oral valganciclovir produces lower peak plasma ganciclovir concentrations than IV ganciclovir, and lower trough plasma ganciclovir concentrations than oral ganciclovir. If any, of these differences in peak and trough plasma drug concentrations with these 3 ganciclovir delivery systems has not been determined The clinical importance,. For more Absorption, Distribution and Excretion (Complete) data for Valganciclovir (7 total), please visit the HSDB record page. Metabolism / Metabolites Rapidly hydrolyzed in the intestinal wall and liver to ganciclovir. No other metabolites have been detected. Is an L-valyl ester (prodrug) of ganciclovir that exists as a mixture of two diastereomers Valganciclovir. After oral administration, both diastereomers are rapidly converted to ganciclovir by intestinal and hepatic esterases. ... Is rapidly hydrolyzed to ganciclovir; no other metabolites have been detected Valganciclovir. Of orally administered radiolabeled ganciclovir (1000 mg single dose) accounted for more than 1% to 2% of the radioactivity recovered in the feces or urine No metabolite. Biological Half-Life Approximately 4.08 hours. Increased in patients with renal function impairment. ...Ten patients were evaluated. Were 56.8 + or - 3.4 years old and had a mean creatinine clearance of 69 + or - 9 mL/min Patients. Of ganciclovir after administration of valganciclovir was 59%, and mean half-life was 3.73 + or - 1.15 hours Oral bioavailability. ... Of ganciclovir following oral administration of Valcyte tablets to either healthy or HIV-positive/CMV-positive subjects was 4.08 + or - 0.76 hours (n=73), and that following administration of intravenous ganciclovir was 3.81 + or - 0.71 hours (n=69) The terminal half-life. In heart, kidney, kidney-pancreas, and liver transplant patients, the terminal elimination half-life of ganciclovir following oral administration of Valcyte was 6.48 + or - 1.38 hours, and following oral administration of ganciclovir capsules was 8.56 + or - 3.62 hours. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Is rapidly converted to ganciclovir Valganciclovir. Might affect the decision to use valganciclovir in a nursing mother Several factors. Is available on the clinical use of ganciclovir or valganciclovir during breastfeeding No information. Can be transmitted to infants though breastmilk, with preterm and immunocompromised infants at greatest risk Cytomegalovirus (CMV). Is available on any changes in the risk of transmission if the mother is being treated with ganciclovir or valganciclovir No information. Although the manufacturer recommends avoiding breastfeeding during valganciclovir use because of the risk of infant drug toxicity, neonates with CMV infections are often treated directly with ganciclovir or valganciclovir. If the mother has a concurrent infection with HIV, breastfeeding is not recommended in the United States and other developed countries. ◉ Effects in Breastfed Infants Was not found as of the revision date Relevant published information. ◉ Effects on Lactation and Breastmilk Was not found as of the revision date Relevant published information. Protein Binding Of ganciclovir is 1% to 2% over concentrations of 0.5 and 51 mg/mL Plasma protein binding. Interactions Potential pharmacologic interaction (additive hematologic toxicity (neutropenia, anemia)) /with concomitant use of valganciclovir and zidovudine. Potential pharmacokinetic interaction /with concomitant use of valganciclovir and probenecid/ (decreased renal clearance and increased AUC of ganciclovir); monitor for ganciclovir toxicity. Potential pharmacologic interactions (additive hematologic toxicity) /with concomitant use of valganciclovir and myelosuppressive agents or irradiation/. Potential pharmacokinetic interaction /with concomitant use of valganciclovir and mycophenolate mofetil/ in patients with renal impairment (increased plasma concentrations of the metabolites of both drugs). For more Interactions (Complete) data for Valganciclovir (6 total), please visit the HSDB record page. |
| References |
[1]. Transport of valganciclovir, a ganciclovir prodrug, via peptide transporters PEPT1 and PEPT2. J Pharm Sci. 2000 Jun;89(6):781-9. [2]. Oral valganciclovir versus ganciclovir as delayed pre-emptive therapy for patients after allogeneic hematopoietic stem cell transplant: a pilot trial (04-0274) and review of the literature. Transpl Infect Dis. 2012 Jun;14(3):259-67. [3]. Valganciclovir prevents cytomegalovirus reactivation in patients receiving alemtuzumab-based therapy. Blood. 2008 Feb 15;111(4):1816-9. |
| Additional Infomation |
Therapeutic Uses Antiviral Agents Are used for initial (induction) treatment and maintenance treatment (secondary prophylaxis) of cytomegalovirus (CMV) retinitis in adults with human immunodeficiency virus (HIV) infection, including those with acquired immunodeficiency syndrome (AIDS) Valganciclovir hydrochloride tablets. /Included in US product labeling/ Also recommend use of oral valganciclovir for treatment and secondary prophylaxis of CMV retinitis in HIV-infected older children and adolescents who can receive adult dosage The US Centers for Disease Control and Prevention (CDC), National Institutes of Health (NIH), and Infectious Diseases Society of America (IDSA). /NOT included in US product labeling/ Are used for prevention of cytomegalovirus (CMV) disease in adult kidney, heart, and kidney-pancreas transplant recipients considered at high risk for the disease (CMV-seronegative recipient of an organ from a CMV-seropositive donor) Valganciclovir hydrochloride tablets. /Included in US product labeling/ Is the most common viral infection after solid organ transplantation (SOT) Cytomegalovirus (CMV). That decrease its incidence after SOT are essential for long-term graft survival Safe and effective prophylactic regimens. Although valganciclovir is not Food and Drug Administration-approved for CMV prophylaxis in liver transplant recipients, postmarketing studies have shown valganciclovir to be as effective as ganciclovir in high-risk adult patients undergoing SOT. Currently, data are lacking for pediatric liver transplantation. Was to compare the efficacy and safety of valganciclovir and ganciclovir for CMV infection prophylaxis in pediatric liver transplant recipients The purpose of this study. Was a retrospective study of 56 pediatric liver transplant recipients who were prescribed either oral ganciclovir (n = 37) or valganciclovir (n = 19) This. Were followed until 200 days after transplantation or death Patients. Compared the rates of early-onset CMV infection and CMV disease in the 2 medication groups The primary outcome measure. Identified patient-specific factors that contributed to CMV acquisition and the incidence of late-onset CMV infection or disease Secondary outcome measures. Were also evaluated The rates of adverse drug effects and discontinuation. Was documented in 0% of valganciclovir patients and in 5.4% of ganciclovir patients (P = 0.54) Early-onset CMV disease. There were no statistically significant differences in the secondary outcomes. Was seen in the valganciclovir group versus the ganciclovir group (22.2% versus 8.1%, P = 0.23) An increased incidence of late-onset CMV disease. Were reported No differences in adverse events. In conclusion, no statistically significant differences were found in the incidence of CMV infection or disease between patients receiving oral valganciclovir and patients receiving oral ganciclovir. Drug Warnings /BOXED WARNING/ WARNING: HEMATOLOGIC TOXICITY, CARCINOGENICITY, TERATOGENICITY, AND IMPAIRMENT OF FERTILITY. Of Valcyte, which is metabolized to ganciclovir, includes granulocytopenia, anemia, and thrombocytopenia Clinical toxicity. In animal studies, ganciclovir was carcinogenic, teratogenic, and caused aspermatogenesis. Of valganciclovir, which is metabolized to ganciclovir, includes granulocytopenia, anemia, and thrombocytopenia Toxicity. Have been reported in patients receiving valganciclovir or ganciclovir Severe leukopenia, neutropenia, anemia, thrombocytopenia, pancytopenia, bone marrow aplasia, and aplastic anemia. May occur at any time and the degree of cytopenia may increase with continued valganciclovir therapy Cytopenia. Usually begin to return to baseline 3-7 days after discontinuance of the drug Cell counts. Should be performed frequently, especially in those with baseline neutrophil counts less than 1000/cu mm and in those who have experienced leukopenia while receiving ganciclovir or other nucleoside analogs Complete blood cell counts (CBCs) and platelet counts. May be warranted if therapy is changed from oral ganciclovir to valganciclovir (because of comparatively increased plasma ganciclovir concentrations with valganciclovir) More frequent monitoring for cytopenias. Should not be used in patients with an absolute neutrophil count less than 500/cu mm, a platelet count less than 25,000/cu mm, or a hemoglobin concentration less than 8 g/dL Valganciclovir. Use with caution in patients with preexisting cytopenias and in those who have received or are receiving concomitant myelosuppressive drugs or irradiation. Indicate that ganciclovir is carcinogenic, mutagenic, teratogenic, and causes aspermatogenesis Animal data. Is converted to ganciclovir and is expected to have carcinogenic and reproductive toxic effects similar to those of ganciclovir Valganciclovir. Can be considered a potential carcinogen in humans and may be teratogenic or embryotoxic at usual therapeutic doses Valganciclovir. Is considered likely that valganciclovir will produce temporary or permanent inhibition of spermatogenesis and also may suppress fertility in females It. Should be advised to use an effective method of contraception during and for at least 30 days after valganciclovir therapy Women of childbearing potential. Should be advised to use a reliable method of barrier contraception during and for at least 90 days after valganciclovir therapy Men. May occur in geriatric patients (with or without renal impairment), patients receiving potentially nephrotoxic drugs, and inadequately hydrated patients Acute renal failure. Should be maintained in all patients Adequate hydration. Use caution and adjust valganciclovir dosage based on creatinine clearance. Use caution in patients receiving concomitant therapy with potentially nephrotoxic drugs. For more Drug Warnings (Complete) data for Valganciclovir (16 total), please visit the HSDB record page. Pharmacodynamics Is an antiviral medication used to treat cytomegalovirus infections Valganciclovir. As the L-valyl ester of ganciclovir, it is actually a prodrug for ganciclovir. After oral administration, it is rapidly converted to ganciclovir by intestinal and hepatic esterases. After this, it (being an analogue of guanosine) gets incorporated into DNA and thus cannot be properly read by DNA polymerase. Results in the termination of the elongation of viral DNA This. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8220 mL | 14.1099 mL | 28.2199 mL | |
| 5 mM | 0.5644 mL | 2.8220 mL | 5.6440 mL | |
| 10 mM | 0.2822 mL | 1.4110 mL | 2.8220 mL |