Physicochemical Properties
| Molecular Formula | C13H21CLN6O4 |
| Molecular Weight | 360.799 |
| Exact Mass | 360.131 |
| Elemental Analysis | C, 43.28; H, 5.87; Cl, 9.83; N, 23.29; O, 17.74 |
| CAS # | 124832-27-5 |
| Related CAS # | Valacyclovir-d8 hydrochloride;1279033-32-7;Valacyclovir;124832-26-4;Valacyclovir-d4 hydrochloride;1331910-75-8;Valacyclovir hydrochloride hydrate;1218948-84-5; 124832-27-5 (HCl); 950189-66-9 |
| PubChem CID | 135398741 |
| Appearance | Typically exists as white to off-white solids at room temperature |
| Density | 1.55g/cm3 |
| Boiling Point | 588.4ºC at 760 mmHg |
| Melting Point | 170-172ºC |
| Flash Point | 309.7ºC |
| Vapour Pressure | 7.95E-14mmHg at 25°C |
| Index of Refraction | 1.673 |
| LogP | 1.285 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 24 |
| Complexity | 483 |
| Defined Atom Stereocenter Count | 1 |
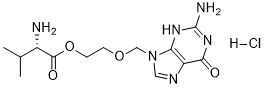
| SMILES | N[C@@H](C(C)C)C(OCCOCN1C=NC2=C1N=C(N)NC2=O)=O.Cl |
| InChi Key | ZCDDBUOENGJMLV-QRPNPIFTSA-N |
| InChi Code | InChI=1S/C13H20N6O4.ClH/c1-7(2)8(14)12(21)23-4-3-22-6-19-5-16-9-10(19)17-13(15)18-11(9)20;/h5,7-8H,3-4,6,14H2,1-2H3,(H3,15,17,18,20);1H/t8-;/m0./s1 |
| Chemical Name | 2-[(2-amino-6-oxo-1H-purin-9-yl)methoxy]ethyl (2S)-2-amino-3-methylbutanoate;hydrochloride |
| Synonyms | Zelitrex; 256U87; Vacyclovir; 256U; L-valyl ester; BW256U87; Valacyclovir Hydrochloride; Valtrex L-valylacyclovir; Valacyclovir HCl; Valtrex; Valaciclovir hydrochloride; Valaciclovir Hcl; 256U87 hydrochloride; BW-256U87; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | HSV-1 (IC50 = 2.9 μg/mL) |
| ln Vitro | At concentrations of 1.64 mM and 23.34 nmol/mg protein/5 min, respectively, the maximal rates of valacyclovir hydrochloride (valaciclovir hydrochloride, VACV) uptake are concentration-dependent and saturable. The in vitro intestinal transport properties of VACV are dominated by hPEPT1, as demonstrated by the very identical Km values observed in hPEPT1/CHO cells, rat and rabbit tissues, and Caco-2 cells [5]. |
| ln Vivo | A major comparative trial found that valacyclovir hydrochloride (1 g twice daily) for 10 days was just as effective as acyclovir (200 mg 5 times daily) for treating a first bout of genital herpes. Two trials found that valacyclovir (200 mg five times daily) was equally efficacious as acyclovir (200 mg five times daily) in a five-day treatment cycle for managing relapses. Valacyclovir at a dose of 1 g per day works just as well as 2 g per day. One dose of valacyclovir can be given every day [1]. Serum and cerebrospinal fluid acyclovir concentrations were assessed at steady state following six days of oral valacyclovir 1,000 mg three times a day [2]. PE and AC have EC50 values in 3T3 cells of 0.02 and 0.01 ug/ml, however in BHK cells they are 0.2 and 0.03 ug/ml. Immunosuppressed mice that were infected were treated with FA and VA (bid, 5.5 days) to eradicate otoparesis, ear lesions (vesicles, etc.), and death. The percentage of erythema was also reduced from 100% to 24% and 38%. By day six, the virus had vanished from the ears and brainstem, but in mice receiving VA treatment, it returned when the medication was stopped [3]. |
| Enzyme Assay | The in vitro 50% inhibitory concentration (IC50) of HSV-1 W strain was determined by using a plaque-reduction assay to verify its sensitivity to acyclovir. The IC50 for HSV-1 W was determined to be 2.9 µg/ml. [4]. |
| Animal Protocol | Forty-seven NZW rabbits latently infected with HSV-1 W strain were divided into four groups: I, 50 mg/kg/day valacyclovir; II, 100 mg/kg/day valacyclovir; III, 150 mg/kg/day valacyclovir; and IV, saline control. One half of the total dose of valacyclovir was delivered via intraperitoneal injections twice daily for 7 days beginning with one dose before excimer laser keratectomy. HSV-1 ocular shedding was determined from eye cultures for 7 days after treatment.The administration of both 100 mg/kg/day (group II) and 150 mg/kg/day (group III) of valacyclovir significantly reduced the number of eyes from which latent HSV-1 was recovered compared with the control group. There was no difference between the control group and group I (50 mg/kg/day valacyclovir). However, all three valacyclovir dosages significantly reduced the total number of HSV-1 shedding days compared with the control group, and 100% HSV-1 TG latency was demonstrated for all four groups.[4] |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation The dosage of acyclovir in milk after valacyclovir is less than 1% of a typical infant dosage and would not be expected to cause any adverse effects in breastfed infants. No special precautions are required when using valacyclovir during breastfeeding. In one study, administration of valacyclovir to mothers with concurrent herpes simplex type 2 and HIV infections reduced breastmilk shedding of the HIV virus in breastmilk at 6 and 14 weeks postpartum, but not later.[1] In another study in HIV-positive mothers, valacyclovir did not reduced breastmilk shedding of cytomegalovirus (CMV) or infant CMV acquisition.[2] ◉ Effects in Breastfed Infants In a study of pregnant women with concurrent HIV and Herpes simplex infections, mothers received zidovudine 300 mg daily from week of pregnancy until 12 months postpartum and nevirapine at delivery. Half of the women (n = 74) also received valacyclovir 500 mg orally twice daily from 34 weeks gestation until 12 months postpartum. At 6 weeks postpartum, all infants who received acyclovir in breastmilk had normal serum creatinine (<0.83 mg/dL). Their median serum creatinine and alanine aminotransferase (ALT) values, and growth were no different from those of unexposed infants, with the exception of one infant with an ALT level of 70.1 units/L. Infants whose mothers received valacyclovir generally had adverse effects that were similar to the placebo group, except that treated infants had a lower risk of eczema and oral thrush than infants in the placebo arm.[1][4] ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. |
| References |
[1]. Valacyclovir. New indication: for genital herpes, simpler administration. Can Fam Physician. 1999 Jul;45:1698-700, 1703-5. [2]. Acyclovir levels in serum and cerebrospinal fluid after oral administration of valacyclovir. Antimicrob Agents Chemother. 2003 Aug;47(8):2438-41. [3]. Comparison of efficacies of famciclovir and valaciclovir against herpes simplex virus type 1 in a murineimmunosuppression model. Antimicrob Agents Chemother. 1995 May;39(5):1114-9. [4]. Dhaliwal DK, Romanowski EG, Yates KA, Valacyclovir inhibits recovery of ocular HSV-1 after experimental reactivation by excimer laser keratectomy. Cornea. 1999 Nov;18(6):693-9. [5]. Guo A, Hu P, Balimane PV, Interactions of a nonpeptidic drug, valacyclovir, with the human intestinal peptide transporter (hPEPT1) expressed in a mammalian cell line.J Pharmacol Exp Ther. 1999 Apr;289(1):448-54. |
| Additional Infomation |
Valacyclovir hydrochloride is an organic molecular entity. Valacyclovir hydrochloride is an antiviral prescription medicine approved by the U.S. Food and Drug Administration (FDA) to: Treat and/or prevent certain types of herpes simplex virus (HSV) infections, including genital herpes (genital lesions) and cold sores (herpes labialis) Reduce the risk of transmitting genital herpes to other people Treat varicella zoster virus (VZV) infections, including chicken pox (primary varicella infection) and shingles (herpes zoster) HSV and VZV infections can be opportunistic infections (OIs) of HIV. An OI is an infection that occurs more frequently or is more severe in people with weakened immune systems—such as people with HIV—than in people with healthy immune systems. Valacyclovir Hydrochloride is the hydrochloride salt of valacyclovir. Valacyclovir is an acyclovir prodrug that, after metabolization, inhibits viral DNA replication. It is used in the management of herpes simplex and varicella zoster infections, as well as prophylactically for human cytomegalovirus infections. A prodrug of acyclovir that is used in the treatment of HERPES ZOSTER and HERPES SIMPLEX VIRUS INFECTION of the skin and mucous membranes, including GENITAL HERPES. See also: Acyclovir (has active moiety); Valacyclovir (has active moiety). |
Solubility Data
| Solubility (In Vitro) |
H2O : ~100 mg/mL (~277.16 mM) DMSO : ~43.33 mg/mL (~120.09 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.93 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.93 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.93 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 100 mg/mL (277.16 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7716 mL | 13.8581 mL | 27.7162 mL | |
| 5 mM | 0.5543 mL | 2.7716 mL | 5.5432 mL | |
| 10 mM | 0.2772 mL | 1.3858 mL | 2.7716 mL |