Physicochemical Properties
| Molecular Formula | C22H14NO3F3 |
| Molecular Weight | 397.34666 |
| Exact Mass | 397.093 |
| CAS # | 1208222-39-2 |
| PubChem CID | 45281794 |
| Appearance | Light yellow to orange solid powder |
| LogP | 5.046 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 29 |
| Complexity | 609 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | SPBGRXOPAXZSER-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C22H14F3NO3/c23-22(24,25)29-17-10-11-19-18(12-17)20(27)21(28)26(19)13-14-6-8-16(9-7-14)15-4-2-1-3-5-15/h1-12H,13H2 |
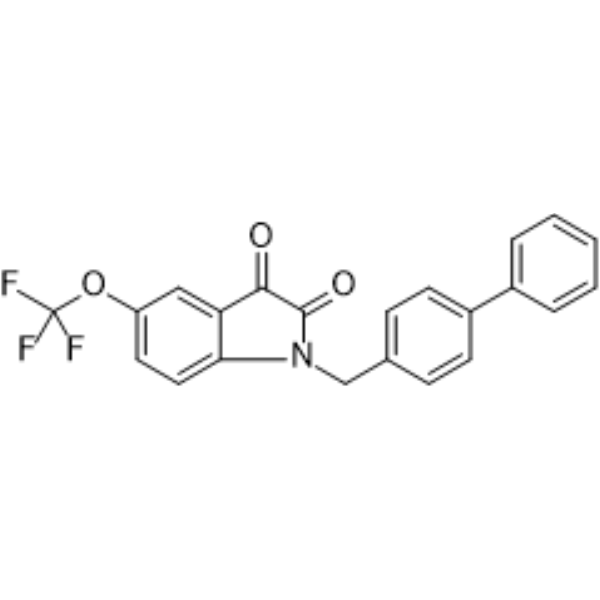
| Chemical Name | 1-[(4-phenylphenyl)methyl]-5-(trifluoromethoxy)indole-2,3-dione |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
VU 0365114 is a highly selective positive allosteric modulator (PAM) for the muscarinic acetylcholine receptor subtype M5 (M5 mAChR). (M5 EC50 = 2.7 µM, >30 µM vs. M1-M4). [1] Muscarinic acetylcholine receptor M5 (M5 receptor) positive allosteric modulator (PAM). (EC50 = 2.7 µM, % ACh Max = 85%). [3] The compound is selective for M5 over M1, M3, M4 receptors (M1 EC50 > 30 µM). [3] |
| ln Vitro |
Human beta cells activated with ACh secrete more insulin when exposed to 10 μM of VU 0365114 [2]. Human beta cells' ACh-stimulated somatostatin production is unaffected by VU 0365114 (10 μM) [2]. VU 0365114 (also referred to as compound 4) is a potent and selective M5 PAM. In M5-CHO cells, it potentiated acetylcholine (ACh)-induced intracellular calcium mobilization with an EC50 of 2.7 µM. It exhibited >30 µM activity at M1, M2, M3, and M4 mAChR subtypes, indicating high selectivity. In an acetylcholine fold-shift assay at a standard 30 µM concentration, VU 0365114 elicited a 10-fold leftward shift of the ACh concentration-response curve. [1] In human pancreatic islets, VU 0365114 (10 μmol/L) amplified the insulin secretion stimulated by acetylcholine (10 μmol/L). [2] VU 0365114 (10 μmol/L) further increased insulin secretion at a basal glucose concentration (3 mmol/L) in the absence of exogenous acetylcholine, indicating potentiation of endogenous cholinergic signaling. This insulin response to VU 0365114 was blocked by atropine. [2] VU 0365114 (10 μmol/L) did not alter somatostatin secretion in response to acetylcholine or at basal glucose concentration. [2] VU0365114 was identified as a highly selective M5 positive allosteric modulator (PAM). In concentration-response curves (CRCs) using M5-CHO cells, it exhibited an M5 EC50 of 2.7 µM and potentiated the acetylcholine (ACh) response to 85% of the maximum ACh response. [3] VU0365114 showed significant selectivity versus M1 and M3 receptors. At 30 µM, it only modestly activated M3 and was inactive at M1 (EC50 > 30 µM). [3] In M5 fold-shift experiments, VU0365114 at 30 µM elicited a greater than 50-fold leftward shift of the ACh concentration-response curve, indicating strong positive allosteric modulation. [3] The compound also displayed moderate intrinsic allosteric agonism at 30 µM in the absence of ACh. [3] |
| Cell Assay |
The functional activity of VU 0365114 as an M5 PAM was determined using an intracellular calcium mobilization assay in Chinese Hamster Ovary (CHO) cells expressing the human M5 muscarinic receptor. Cells were loaded with a calcium-sensitive fluorescent dye. Increases in intracellular calcium in response to acetylcholine (ACh) were measured fluorometrically in the presence or absence of the test compound. The assay was used to determine the compound's ability to potentiate the response to a submaximal concentration (EC20) of ACh (for initial screening) and to generate full concentration-response curves for calculating EC50 values and maximal potentiation (ACh Max %). The fold-shift of the ACh concentration-response curve was also assessed at a fixed compound concentration. [1] Hormone secretion from human pancreatic islets was dynamically measured using a high-capacity, automated perfusion system. A peristaltic pump pushed HEPES-buffered solution (containing 125 mmol/L NaCl, 5.9 mmol/L KCl, 2.56 mmol/L CaCl2, 1 mmol/L MgCl2, 25 mmol/L HEPES, and 0.1% BSA, pH 7.4) at a rate of 100 μL/min through a column containing 100 human islets immobilized in gel. The column and solutions were maintained at 37°C. The perfusate was collected every minute into a cooled fraction collector. [2] For experiments, the glucose concentration was adjusted to 3 mmol/L. Drugs, including acetylcholine and VU 0365114, were applied via the perfusion buffer. Insulin in the collected fractions was quantified using a multiplex immunoassay kit. Somatostatin secretion was determined using a fluorescent enzyme-linked immunoassay kit. [2] Functional activity (potentiation of an EC20 concentration of acetylcholine) was assessed in Chinese Hamster Ovary (CHO) cells stably expressing human muscarinic acetylcholine receptor subtypes (M1, M3, M5). For M2 and M4 receptor assays, which couple to Gαi/o proteins, the cells were co-transfected with a chimeric Gαq/i5 protein to redirect signaling to the phospholipase C pathway, allowing detection via intracellular calcium mobilization. [3] Intracellular calcium mobilization was used as the functional readout for receptor activation. Compound activity was initially triaged in a single-point screen at 10 µM for its ability to potentiate an EC20 of ACh in M5-CHO cells. [3] Selected compounds, including VU0365114, were further characterized in full 8-point concentration-response curves to determine EC50 values and the percentage of maximal ACh response (% ACh Max). [3] Fold-shift experiments were performed by evaluating the acetylcholine concentration-response curve in the presence of a fixed concentration (30 µM) of the test compound (VU0365114) to determine the magnitude of leftward shift. [3] |
| ADME/Pharmacokinetics |
The analogs in this series, including VU0365114, displayed moderate to poor pharmacokinetic (PK) properties in rats. [3] Limited brain exposure was observed, with a brain-to-plasma AUC ratio (AUCBrain/AUCPlasma) of approximately 0.25. This was attributed to the presence of the bis-carbonyl group in the isatin moiety of the compound. [3] It was noted that brain exposure was not examined when a DMSO-containing vehicle was used, which might potentially improve brain levels. [3] |
| References |
[1]. Heterobiaryl and heterobiaryl ether derived M5 positive allosteric modulators. Bioorg Med Chem Lett. 2010 Oct 1;20(19):5617-22. [2]. Control of insulin secretion by cholinergic signaling in the human pancreatic islet. Diabetes. 2014 Aug;63(8):2714-26. [3]. Chemical lead optimization of a pan G(q) mAChR M(1), M(3), M(5) positive allosteric modulator (PAM) lead. Part I: Development of the first highly selective M(5) PAM. Bioorg Med Chem Lett. 2010 Jan 15;20(2):558-62. |
| Additional Infomation |
1-[(4-phenylphenyl)methyl]-5-(trifluoromethoxy)indole-2,3-dione is a member of biphenyls. VU 0365114 is a lipophilic compound with a logP > 4.5, which contributes to its limited solubility and overall poor physicochemical properties. It was developed through the optimization of VU0238429, an earlier M5-preferring PAM. The structure-activity relationship (SAR) indicated that the 5-trifluoromethoxy (5-OCF3) group on the isatin core was critical for M5 PAM activity and selectivity. This compound served as a key chemical tool and benchmark for further optimization efforts aimed at improving physicochemical properties by introducing basic heterocycles to enable salt formation and reduce lipophilicity. [1] VU 0365114 is identified as an allosteric modulator of the M5 muscarinic acetylcholine receptor. [2] The study utilized VU 0365114 as a pharmacological tool to reveal the presence of functional M5 receptors on human pancreatic β-cells, demonstrating that human β-cells express M5 receptors in addition to M3 receptors. [2] The findings suggest that endogenous acetylcholine in human islets acts on β-cell M5 receptors, and modulating this receptor can influence insulin secretion. [2] VU0365114 (designated as compound 6a in the study) was developed through chemical lead optimization starting from a pan-Gq mAChR PAM (VU0119498) and an M5-preferring PAM (VU0238429). The optimization strategy involved introducing biaryl motifs. [3] It represents one of the first highly selective M5 PAM tool compounds, enabling the study of the M5 receptor's role in the central nervous system (CNS), which was previously hindered by a lack of selective pharmacological agents. [3] The compound is considered an important tool for studying M5 function in cellular assays, electrophysiology, and via intracerebroventricular (icv) injection. [3] |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 77.5 mg/mL (~195.04 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.58 mg/mL (6.49 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.58 mg/mL (6.49 mM) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5167 mL | 12.5834 mL | 25.1667 mL | |
| 5 mM | 0.5033 mL | 2.5167 mL | 5.0333 mL | |
| 10 mM | 0.2517 mL | 1.2583 mL | 2.5167 mL |