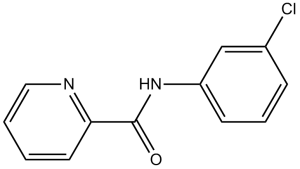
VU 0364770 (VU-0364770, VU0364770) is a potent positive allosteric modulator (PAM) of mGlu4 (metabotropic glutamate receptor 4) with anti-parkinsonian-like activity. It inhibits mGlu with an EC50 of 1.1 μM for human mGlu4. It exhibits little activity at 68 other receptors such as other mGlu subtypes. Positive allosteric modulators (PAMs) of metabotropic glutamate receptor 4 (mGlu₄), including N-phenyl-7-(hydroxyimino) cyclopropa[b]chromen-1a-carboxamide, can produce anti-parkinsonian-like effects in preclinical models of PD. VU0364770 produces efficacy alone and in combination with L-DOPA or an adenosine 2A antagonist in preclinical rodent models of Parkinson's disease.
Physicochemical Properties
| Molecular Formula | C12H9CLN2O | |
| Molecular Weight | 232.67 | |
| Exact Mass | 232.04 | |
| CAS # | 61350-00-3 | |
| Related CAS # | VU0364770 hydrochloride;1414842-70-8 | |
| PubChem CID | 836002 | |
| Appearance | Light yellow to yellow solid powder | |
| LogP | 3.371 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 2 | |
| Rotatable Bond Count | 2 | |
| Heavy Atom Count | 16 | |
| Complexity | 247 | |
| Defined Atom Stereocenter Count | 0 | |
| InChi Key | SUYUTNCKIOLMAJ-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C12H9ClN2O/c13-9-4-3-5-10(8-9)15-12(16)11-6-1-2-7-14-11/h1-8H,(H,15,16) | |
| Chemical Name | N-(3-chlorophenyl)pyridine-2-carboxamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Metabotropic glutamate receptor 4 (mGlu4) (Ki = 7.8 nM; EC50 = 23 nM for potentiation of glutamate-induced calcium mobilization) [1] |
| ln Vitro |
In recombinant systems, VU0364770 acts as a selective positive allosteric modulator of mGlu4. Strong PAM of several signaling pathways, VU0364770, increases the rat and human mGlu4 receptors' sensitivity to the endogenous agonist glutamate. VU0364770 raises the maximal response to glutamate from 100 to 227±17% and causes a concentration-dependent potentiation of the response to an EC20 concentration of glutamate with an EC50 of 1.1±0.2 μM. Full IC50 determinations are carried out for VU0364770 at the MAO-A and MAO-B isoforms due to concerns that this chemical scaffold may possess activity at MAO; these studies yield Kis of 8.5 and 0.72 μM for human MAO-A and human MAO-B, respectively. VU0364770 displays weak PAM activity (4.3-fold left shift of the glutamate CRC) at mGlu6 and antagonist activity (3.3-fold right shift of the glutamate CRC) at mGlu5, when tested at a 10 μM concentration at each mGlu receptor (compare to the 16.5-fold left shift of the glutamate concentration-response for mGlu4 at 10 μM). VU0364770 shows antagonist activity at mGlu5 with a potency of 17.9±5.5 μM and PAM activity at mGlu6 with a potency of 6.8±1.7 μM when further evaluated in a full concentration-response curve format (compare with the potency of VU0364770 on the rat mGlu4 receptor of 290±80}M)[1]. VU 0364770 is a selective positive allosteric modulator (PAM) of mGlu4. In Chinese hamster ovary (CHO) cells expressing human mGlu4, it potentiated glutamate-induced calcium mobilization in a concentration-dependent manner, with an EC50 of 23 nM and maximal potentiation of ~4.2-fold [1] It showed no significant activity against other mGlu receptor subtypes (mGlu1-3, 5-8) at concentrations up to 10 μM, and no binding affinity for ionotropic glutamate receptors, adenosine receptors, or dopamine receptors [1] In rat midbrain slices containing substantia nigra pars compacta (SNc), VU 0364770 (1-10 μM) enhanced mGlu4-mediated inhibition of GABAergic neurotransmission, as indicated by reduced frequency of miniature inhibitory postsynaptic currents (mIPSCs) [1] |
| ln Vivo |
In animal models, VU0364770 shows appropriate pharmacokinetic characteristics for systemic administration. VU0364770 has a 2.92 L/kg volume of distribution and quickly leaves the systemic circulation (165 ml/min/kg) upon intravenous injection. The highly protein-bound ligand VU0364770 exhibits free fractions in rat and human plasma of 1.8% and 2.7%, respectively. Following systemic administration of a 10 mg/kg dosage, VU0364770 also exhibits a better pharmacokinetic profile in comparison to previously reported mGlu4 PAMs, with greater central penetration and a total brain-to-plasma ratio of more than 1. Haloperidol-induced cataleptic seizures are reversed in a dose-dependent manner by VU0364770. following subcutaneous administration (F6,69=8.04; p<0.001)[1]. In 6-hydroxydopamine (6-OHDA)-lesioned rats (a Parkinson’s disease model), oral administration of VU 0364770 (10-30 mg/kg) dose-dependently reduced apomorphine-induced rotational behavior, with an ED50 of ~18 mg/kg. It also improved locomotor function in the open field test (increased total distance traveled and rearing counts) [1] Combination treatment of VU 0364770 (5 mg/kg, po) with a subtherapeutic dose of L-DOPA (10 mg/kg, ip) in 6-OHDA-lesioned rats produced synergistic anti-parkinsonian effects, reducing rotational behavior by ~65% (vs. ~20% with L-DOPA alone) [1] Co-administration of VU 0364770 (5 mg/kg, po) with an adenosine 2A (A2A) antagonist (1 mg/kg, ip) in 6-OHDA-lesioned rats enhanced locomotor activity more effectively than either drug alone, increasing open field distance by ~80% vs. vehicle [1] In MPTP-treated mice (another Parkinson’s disease model), VU 0364770 (15 mg/kg, po) improved motor coordination in the rotarod test (increased latency to fall) and reduced bradykinesia in the pole test (decreased time to descend) [1] |
| Enzyme Assay |
Radioligand binding assay for mGlu4: Prepare membrane homogenates from CHO cells expressing human mGlu4. Incubate homogenates with a fixed concentration of [3H]-L-AP4 (a selective mGlu4 agonist) and various concentrations of VU 0364770 at 25°C for 90 minutes. Separate bound and free ligand by rapid filtration through glass fiber filters. Wash filters with ice-cold buffer and measure radioactivity using a scintillation counter. Calculate Ki value from competition binding curves [1] Glutamate-induced calcium mobilization assay: Seed CHO-hmGlu4 cells in 96-well plates and culture until confluent. Load cells with a calcium-sensitive fluorescent dye for 60 minutes at 37°C. Preincubate cells with VU 0364770 (0.1-1000 nM) for 30 minutes, then stimulate with a submaximal concentration of glutamate (1 μM). Record fluorescent intensity changes in real time using a microplate reader. Calculate EC50 as the concentration that potentiates 50% of the maximal glutamate-induced calcium response [1] |
| Cell Assay |
Midbrain slice GABAergic neurotransmission assay: Prepare 250-μm-thick rat midbrain slices containing SNc and incubate in artificial cerebrospinal fluid (ACSF) at 32°C for 1 hour. Add VU 0364770 (1-10 μM) to the ACSF for 30 minutes. Use whole-cell patch-clamp recording to measure mIPSCs in SNc dopamine neurons. Analyze mIPSC frequency and amplitude to assess inhibition of GABAergic input [1] |
| Animal Protocol |
Suspended in an aqueous solution of 10% Tween 80; 1 to 2 ml/kg i.p./s.c. or 10 ml/kg p.o.; s.c. or p.o. administration Haloperidol-Induced Catalepsy 6-OHDA-lesioned rat Parkinson’s model: Adult male rats are anesthetized and stereotaxically injected with 6-OHDA into the right medial forebrain bundle. Four weeks after lesioning, rats with >200 apomorphine-induced rotations/30 minutes are selected for experiments. VU 0364770 is suspended in 0.5% methylcellulose and administered orally at doses of 10, 20, or 30 mg/kg 60 minutes before apomorphine injection (0.5 mg/kg, sc). Rotational behavior is recorded for 30 minutes [1] Combination treatment with L-DOPA: 6-OHDA-lesioned rats receive oral VU 0364770 (5 mg/kg) or vehicle 60 minutes before intraperitoneal injection of L-DOPA (10 mg/kg) plus benserazide (2.5 mg/kg, to inhibit peripheral L-DOPA metabolism). Rotational behavior is recorded for 120 minutes post-L-DOPA injection [1] MPTP-treated mouse Parkinson’s model: Adult male mice receive intraperitoneal injections of MPTP (20 mg/kg) once daily for 4 consecutive days. Seven days after the last MPTP injection, VU 0364770 (15 mg/kg, po) is administered 60 minutes before the rotarod test (accelerating from 4 to 40 rpm over 5 minutes) and pole test (measuring time to descend a vertical pole). Motor performance is recorded and compared to vehicle-treated MPTP mice [1] |
| ADME/Pharmacokinetics |
Oral absorption: VU 0364770 has good oral bioavailability of ~68% in rats [1] Distribution: It distributes widely into tissues, with a volume of distribution (Vdss) of ~2.1 L/kg in rats. Brain penetration is efficient, with a brain/plasma concentration ratio of ~0.8 in rats 1 hour after oral dosing [1] Metabolism: It is metabolized in the liver primarily via cytochrome P450 3A4 and 2C19, producing inactive glucuronide conjugates [1] Excretion: The elimination half-life (t1/2) is ~4.3 hours in rats. Approximately 58% of the dose is excreted in feces and 32% in urine, with <7% excreted as unchanged drug [1] Plasma protein binding: VU 0364770 has a plasma protein binding rate of ~91% in rats [1] |
| Toxicity/Toxicokinetics |
Acute toxicity study in rats showed no mortality or overt toxicity at oral doses up to 200 mg/kg [1] Subchronic toxicity assessment (14 days) in rats at oral doses of 10, 30, 100 mg/kg/day revealed no significant changes in body weight, food intake, hematological parameters, or liver/kidney function [1] |
| References |
[1]. The metabotropic glutamate receptor 4-positive allosteric modulator VU0364770 produces efficacy alone and in combination with L-DOPA or an adenosine 2A antagonist in preclinical rodent models of Parkinson's disease. J Pharmacol Exp Ther. |
| Additional Infomation |
VU 0364770 is a selective, orally bioavailable positive allosteric modulator (PAM) of metabotropic glutamate receptor 4 (mGlu4) [1] Its mechanism of action involves binding to the allosteric site of mGlu4, enhancing the receptor’s response to endogenous glutamate, which inhibits GABAergic neurotransmission in the SNc and restores dopamine system balance in Parkinson’s disease [1] It exhibits anti-parkinsonian efficacy alone and synergistic effects when combined with L-DOPA or A2A antagonists, supporting potential use in Parkinson’s disease treatment (monotherapy or adjunctive therapy) [1] Its high selectivity for mGlu4 minimizes off-target effects, and favorable ADME properties (good oral bioavailability, brain penetration) support clinical development potential [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (10.74 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (10.74 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.2979 mL | 21.4897 mL | 42.9793 mL | |
| 5 mM | 0.8596 mL | 4.2979 mL | 8.5959 mL | |
| 10 mM | 0.4298 mL | 2.1490 mL | 4.2979 mL |