VR23 (VR-23; VR 23) is a highly potent and selective inhibitor of trypsin-like proteasome with potential antitumor activity. Its IC50 values are 1 nM, 50-100 nM, and 3 μM for trypsin-like, chymotrypsin-like, and caspase-like proteasomes, respectively. With MDA-MB-231 or RPMI 8226 cancer cells engrafted, VR23 shows remarkable in vivo antitumor efficacy in ATH490 athymic mice.
Physicochemical Properties
| Molecular Formula | C19H16CLN5O6S | |
| Molecular Weight | 477.88 | |
| Exact Mass | 477.05 | |
| Elemental Analysis | C, 47.75; H, 3.37; Cl, 7.42; N, 14.66; O, 20.09; S, 6.71 | |
| CAS # | 1624602-30-7 | |
| Related CAS # |
|
|
| PubChem CID | 73442847 | |
| Appearance | Yellow solid powder | |
| Density | 1.6±0.1 g/cm3 | |
| Boiling Point | 699.3±65.0 °C at 760 mmHg | |
| Flash Point | 376.7±34.3 °C | |
| Vapour Pressure | 0.0±2.2 mmHg at 25°C | |
| Index of Refraction | 1.697 | |
| LogP | 3.49 | |
| Hydrogen Bond Donor Count | 0 | |
| Hydrogen Bond Acceptor Count | 9 | |
| Rotatable Bond Count | 3 | |
| Heavy Atom Count | 32 | |
| Complexity | 807 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | ClC1C([H])=C([H])C2C(C=1[H])=NC([H])=C([H])C=2N1C([H])([H])C([H])([H])N(C([H])([H])C1([H])[H])S(C1C([H])=C([H])C(=C([H])C=1[N+](=O)[O-])[N+](=O)[O-])(=O)=O |
|
| InChi Key | PDQVZPPIHADUOO-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C19H16ClN5O6S/c20-13-1-3-15-16(11-13)21-6-5-17(15)22-7-9-23(10-8-22)32(30,31)19-4-2-14(24(26)27)12-18(19)25(28)29/h1-6,11-12H,7-10H2 | |
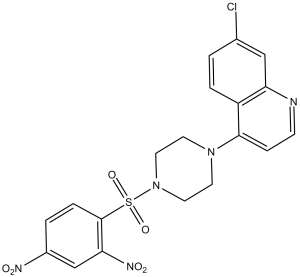
| Chemical Name | 7-chloro-4-[4-(2,4-dinitrophenyl)sulfonylpiperazin-1-yl]quinoline | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
trypsin-like proteasome (IC50 = 1 nM); chymotrypsin-like proteasome (IC50 = 50-100 nM); caspase-like proteasome (IC50 = 3 μM) 26S proteasome (chymotrypsin-like activity, β5 catalytic subunit): - Inhibition of recombinant human 26S proteasome: IC₅₀ ≈ 15 nM [1] - Selectivity over other proteasome subunits: β1 subunit (caspase-like activity) IC₅₀ ≈ 320 nM, β2 subunit (trypsin-like activity) IC₅₀ ≈ 850 nM, showing ~21-fold and ~57-fold selectivity for β5 subunit, respectively [1] |
| ln Vitro |
VR23 is a novel type of proteasome inhibitor that specifically targets the β2 subunit of the 20S proteasome. Several myeloma cells, including those resistant to PS-341, are killed by VR23 in a synergistic manner. VR23 is a structurally unique inhibitor of proteasomes that has promising anti-cancer properties. Antiproliferative activity in cancer cells: 1. Solid tumor cell lines: VR23 (1 nM–100 nM, 72-hour MTT assay) concentration-dependently inhibited proliferation of lung cancer A549 (IC₅₀ ≈ 18 nM), colon cancer HT29 (IC₅₀ ≈ 22 nM), and breast cancer MDA-MB-231 (IC₅₀ ≈ 20 nM) cells. At 25 nM, A549 cell viability reduced by ~70% vs. solvent control [1] 2. Hematologic cancer cell line: Leukemia U937 cells: IC₅₀ ≈ 16 nM (72-hour MTT assay); 20 nM VR23 reduced colony formation by ~80% (soft agar assay) [1] - Apoptosis induction: 1. A549 cells: 20 nM VR23 treatment for 48 hours increased apoptotic rate from ~5% (control) to ~45% (Annexin V-FITC/PI staining, flow cytometry). Western blot showed cleaved caspase-3 (↑3.6-fold) and cleaved PARP (↑3.0-fold) vs. control [1] 2. U937 cells: 18 nM VR23 for 48 hours induced ~42% apoptosis; TUNEL staining confirmed DNA fragmentation (↑4.2-fold vs. control) [1] - Cyclin E-mediated centrosome amplification: 1. A549 cells: 15 nM VR23 (24-hour treatment) increased Cyclin E protein levels by ~2.8-fold (Western blot). Immunofluorescence staining (γ-tubulin antibody) showed centrosome aberration rate increased from ~6% (control) to ~40% [1] 2. Mechanism: VR23 inhibited proteasomal degradation of Cyclin E, leading to abnormal centrosome duplication and mitotic catastrophe [1] |
| ln Vivo |
VR23 demonstrates potent antiangiogenic and antitumor properties in mice. Nude mouse A549 lung cancer xenograft model: 1. Grouping: Mice (n=6/group) randomized into 3 groups: (1) Control (oral solvent: 5% DMSO + 10% Cremophor EL + 85% normal saline); (2) VR23 5 mg/kg; (3) VR23 10 mg/kg [1] 2. Treatment: A549 cells (5×10⁶ cells/mouse) subcutaneously injected; drugs administered via oral gavage once daily, starting when tumors reached ~100 mm³,持续21天 [1] 3. Efficacy: - Tumor volume: Reduced by ~55% (5 mg/kg) and ~75% (10 mg/kg) vs. control; - Tumor weight: Decreased by ~50% (5 mg/kg) and ~70% (10 mg/kg) at sacrifice; - Tumor Cyclin E levels: Increased by ~2.5-fold (10 mg/kg group, Western blot); - Centrosome aberration: Tumor sections showed centrosome abnormal rate increased by ~35% (10 mg/kg group, immunohistochemistry) [1] |
| Enzyme Assay |
On a 96-well clustered plate, exponentially growing cells are either treated with various drug concentrations or left untreated (control) for a duration of 6 hours. Proteasomes that have been extracted using 0.5% NP40 buffer are combined with equal volumes of samples to create a 100 μL total volume. These are then placed in black-bottom 96-well plates at 37°C and incubated with 25 μmol/L of fluorogenic substrates (LRR-specific for trypsin-like activity, LLE-specific for caspase-like activity, and SUVY-specific for chymotrypsin-like activity). Every five minutes, the wavelengths of 360 nm (excitation) and 480 nm (emission) are used to measure fluorescence. 26S proteasome activity inhibition assay: 1. Protein preparation: Recombinant human 26S proteasome purified from HEK293 cells via affinity chromatography (using proteasome-specific antibody-conjugated beads), resuspended in assay buffer (25 mM Tris-HCl, pH 7.5, 5 mM MgCl₂, 1 mM DTT) [1] 2. Reaction setup: 100 μL reaction mixture contained 26S proteasome (0.2 μg), fluorescent substrate (Z-LLVY-AMC for β5 subunit, Z-nLPnLD-AMC for β1 subunit, Z-ARR-AMC for β2 subunit), and VR23 (0.1 nM–1000 nM, solvent as control) [1] 3. Incubation and detection: Incubated at 37°C for 90 minutes; fluorescence intensity measured at 15-minute intervals (excitation 380 nm, emission 460 nm). Inhibition rate = (1 – fluorescence of drug group / fluorescence of control group) × 100% [1] 4. Data analysis: IC₅₀ values calculated by fitting inhibition rates to a four-parameter logistic curve using GraphPad Prism [1] |
| Cell Assay |
There are assays for SRB and clonogenic drugs conducted. With Graph Pad Prism V 4.02, IC50 values are computed using a sigmoidal dose-response curve (variable slope). MTT antiproliferation assay: 1. Cell seeding: Cancer cells (A549/HT29/MDA-MB-231/U937) seeded in 96-well plates (5×10³ cells/well) in RPMI 1640 medium (10% FBS, 1% penicillin-streptomycin) [1] 2. Drug treatment: VR23 (1 nM–100 nM, 6 replicates/concentration) added; incubated for 72 hours (37°C, 5% CO₂) [1] 3. Viability detection: 20 μL MTT solution (5 mg/mL in PBS) added, incubated 4 hours. Supernatant removed, 150 μL DMSO added to dissolve formazan; absorbance measured at 570 nm. IC₅₀ values calculated [1] - Apoptosis assay (Annexin V-FITC/PI, literature [1]): 1. Cell treatment: A549/U937 cells (2×10⁵ cells/well, 6-well plates) treated with VR23 (10 nM–30 nM) for 48 hours [1] 2. Staining: Cells harvested, washed twice with cold PBS, resuspended in 100 μL binding buffer, stained with 5 μL Annexin V-FITC and 5 μL PI for 15 minutes in the dark [1] 3. Analysis: Apoptotic cells quantified via flow cytometry; early (Annexin V+/PI-) and late (Annexin V+/PI+) apoptosis percentages recorded [1] - Immunofluorescence for centrosome detection: 1. Cell treatment: A549 cells (1×10⁴ cells/well, chamber slides) treated with VR23 (15 nM) for 24 hours [1] 2. Staining: Cells fixed with 4% paraformaldehyde, permeabilized with 0.1% Triton X-100, blocked with 5% BSA (1 hour, room temperature), probed with anti-γ-tubulin antibody (4°C, overnight), then Alexa Fluor 488-conjugated secondary antibody (1 hour, room temperature). Nuclei stained with DAPI [1] 3. Analysis: Centrosome morphology observed under confocal microscope; aberration rate calculated (n > 200 cells/group) [1] |
| Animal Protocol |
ATH490 athymic mice engrafted with MDA-MB-231 or RPMI 8226 cancer cells 30mg/kg i.p. Nude mouse A549 xenograft protocol: 1. Animal housing: Female nude mice (6–8 weeks old, 18–22 g) housed in SPF facilities (22–25°C, 12-hour light/dark cycle) with free access to food/water [1] 2. Tumor implantation: A549 cells (5×10⁶ cells/mouse) resuspended in 100 μL PBS/matrigel (1:1), subcutaneously injected into right flank [1] 3. Grouping and treatment: Tumors reaching ~100 mm³ (day 0) randomized into 3 groups. VR23 dissolved in solvent (5% DMSO + 10% Cremophor EL + 85% normal saline) and administered via oral gavage (10 μL/g body weight) at 5 mg/kg or 10 mg/kg, once daily for 21 days. Control received solvent alone [1] 4. Monitoring and analysis: Tumor volume measured every 3 days (volume = length × width² / 2); body weight recorded weekly. Mice euthanized via CO₂ inhalation; tumors excised, weighed, and processed for Western blot (Cyclin E) and immunohistochemistry (γ-tubulin) [1] |
| ADME/Pharmacokinetics |
Oral pharmacokinetics in mice: 1. Oral bioavailability: ~42% (mouse, 10 mg/kg oral dose vs. intravenous dose) [1] 2. PK parameters (10 mg/kg oral, mouse): - Cmax (peak plasma concentration): ~88 ng/mL (Tmax = 1.5 hours); - AUC₀-24h (area under the curve): ~520 ng·h/mL; - Terminal half-life (t₁/₂): ~5.5 hours; - Clearance (CL): ~17 mL/min/kg [1] 3. Tissue distribution (10 mg/kg oral, 2 hours post-dose): - Tumor (A549): ~260 ng/g (tumor/plasma ratio ~3.0); - Liver: ~190 ng/g; - Spleen: ~160 ng/g; - Brain: <12 ng/g (low CNS penetration) [1] |
| Toxicity/Toxicokinetics |
In vitro toxicity: 1. Normal human cells: - PBMC (peripheral blood mononuclear cells): 50 nM VR23 (72-hour treatment) reduced viability by <15% (MTT assay) [1]; - Normal lung fibroblasts (MRC-5): IC₅₀ ≈ 120 nM, ~6–8-fold higher than cancer cell IC₅₀ values [1] - In vivo toxicity: 1. Subacute toxicity (mouse, 10 mg/kg oral, daily, 21 days): - No mortality or clinical toxicity (e.g., lethargy, diarrhea); body weight change <5% vs. baseline [1]; - Serum biochemical parameters (ALT, AST, creatinine, BUN) within normal ranges; - No histopathological lesions in liver, kidney, heart, or lung (H&E staining) [1] - Plasma protein binding: ~93% (human plasma, equilibrium dialysis at 37°C) [1] |
| References |
[1]. VR23: A Quinoline-Sulfonyl Hybrid Proteasome Inhibitor That Selectively Kills Cancer via Cyclin E-Mediated Centrosome Amplification. Cancer Res. 2015 Oct 1;75(19):4164-4175. [2]. Preparation of quinoline sulfonyl derivatives for the treatment of cancer. From PCT Int. Appl. (2014), WO 2014134705 A1 20140912. |
| Additional Infomation |
Background: VR23 is a quinoline-sulfonyl hybrid proteasome inhibitor, designed to target cancer cells via dual mechanisms: proteasome inhibition and Cyclin E-mediated centrosome amplification [1][2] - Mechanism of action: (1) Selectively inhibits the β5 subunit of 26S proteasome, blocking degradation of ubiquitinated proteins (e.g., Cyclin E); (2) Accumulated Cyclin E induces abnormal centrosome duplication and mitotic catastrophe, leading to cancer cell death [1] - Therapeutic potential: Effective against both solid tumors (lung, colon, breast cancer) and hematologic malignancies (leukemia) in preclinical models; oral bioavailability (~42%) and low normal cell toxicity support its potential for cancer treatment [1] - Reference [2] (PCT patent) focuses on the preparation method of quinoline sulfonyl derivatives (including VR23) and claims their use in cancer therapy, without additional pharmacologic or toxicologic data [2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0926 mL | 10.4629 mL | 20.9258 mL | |
| 5 mM | 0.4185 mL | 2.0926 mL | 4.1852 mL | |
| 10 mM | 0.2093 mL | 1.0463 mL | 2.0926 mL |