VLX600 is a novel Iron Chelator Targeting Both Proliferating and Quiescent Cancer Cells. VLX600 is able to potentiate the effect of radiation in tumor spheroids in a synergistic manner. VLX600 is a lipophilic cation-based triazinoindolyl-hydrazone compound and mitochondrial oxidative phosphorylation (OxPhos) inhibitor, with potential antineoplastic activity.
Physicochemical Properties
| Molecular Formula | C17H16CLN7 |
| Molecular Weight | 353.81 |
| Exact Mass | 319.154 |
| Elemental Analysis | C, 57.71; H, 4.56; Cl, 10.02; N, 27.71 |
| CAS # | 1622945-04-3 |
| PubChem CID | 136276630 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 517.7±60.0 °C at 760 mmHg |
| Flash Point | 266.9±32.9 °C |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.757 |
| LogP | 2.18 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 24 |
| Complexity | 567 |
| Defined Atom Stereocenter Count | 0 |
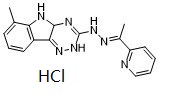
| SMILES | CC1=C2C(=CC=C1)C3=NNC(=NC3N2)N/N=C(/C)\C4=CC=CC=N4 |
| InChi Key | GCXMWESEENEVIC-SRZZPIQSSA-N |
| InChi Code | InChI=1S/C17H17N7/c1-10-6-5-7-12-14(10)19-16-15(12)22-24-17(20-16)23-21-11(2)13-8-3-4-9-18-13/h3-9,16,19H,1-2H3,(H2,20,23,24)/b21-11+ |
| Chemical Name | 1-(2-Pyridinyl)ethanone 2-(4a,5-dihydro-6-methyl-2H-1,2,4-triazino[5,6-b]indol-3-yl)hydrazone |
| Synonyms | VLX600 HCl; VLX-600 HCl; VLX 600 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product is not stable in solution, please use freshly prepared working solution for optimal results. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | OXPHOS/oxidative phosphorylation |
| ln Vitro | An autophagic response is induced by VLX600 (6 μM; 72 hours)[2]. HCT116 spheroids are cytotoxic to VLX600. A HIF-1α-dependent glycolytic response is induced by VLX600. In HCT116 cells, VLX600 limits oxygen consumption. Through a mechanism that is independent of HIF-1α, VLX600 prevents the phosphorylation of the mTOR downstream effectors 4EBP1 and p70-S6K. Reduced ATP levels are selectively caused by VLX600 in cancer cells, but not in normal cells[2]. |
| ln Vivo | In human tumor xenografts, VLX600 (16 mg/kg; iv; every third day for 16 days) exhibits anti-tumor activity[2]. |
| Cell Assay |
Cell Proliferation Assay[2] Cell Types: HCT116, HT29, SW620, HT8, DLD and RKO cells Tested Concentrations: 0.1, 1, 10, 100μM Incubation Duration: 72 hrs (hours) Experimental Results: Inhibited the proliferation of these cells. Western Blot Analysis[2] Cell Types: HCT116 cells Tested Concentrations: 6 μM Incubation Duration: 72 hrs (hours) Experimental Results: LC3-II was induced. |
| Animal Protocol |
Animal/Disease Models: NMRI nu/nu (nude) mice (HCT116 and HT29 colon cancer xenografts)[2] Doses: 16 mg/kg Route of Administration: Intravenously; every third day for 16 days Experimental Results: Anti-tumor activity was observed in both HCT116 and HT29 colon cancer xenografts. |
| References |
[1]. A novel tumor spheroid model identifies selective enhancement of radiation by an inhibitor of oxidative phosphorylation. Oncotarget. 2019 Sep 3;10(51):5372-5382. [2]. Induction of mitochondrial dysfunction as a strategy for targeting tumour cells in metabolically compromised microenvironments. Nat Commun. 2014;5:3295. |
| Additional Infomation | OxPhos Inhibitor VLX600 is a lipophilic cation-based triazinoindolyl-hydrazone compound and mitochondrial oxidative phosphorylation (OxPhos) inhibitor, with potential antineoplastic activity. Upon infusion, in normal cells and proliferating tumor cells where glucose is readily available, inhibition of OxPhos by VLX600 induces a hypoxia-inducible factor 1-alpha (HIF-1alpha)-dependent shift to, and an increase in glycolysis. Glycolysis alone does not produce enough energy to support the growth of tumor cells in this environment, and the induction of autophagy occurs. In the metabolically compromised tumor microenvironment, the availability of oxygen and glucose is limited due to poor vascularization and perfusion of tumor micro-areas. Tumor cells growing in this environment are thus unable to compensate for decreased mitochondrial function by increasing glycolysis. This leads to nutrient depletion, decreased energy production, induction of autophagy, tumor cell death and an inhibition of cell proliferation in quiescent tumor cells. Mitochondrial OxPhos, which is hyperactivated in cancer cells, plays a key role in the promotion of cancer cell proliferation. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8264 mL | 14.1319 mL | 28.2638 mL | |
| 5 mM | 0.5653 mL | 2.8264 mL | 5.6528 mL | |
| 10 mM | 0.2826 mL | 1.4132 mL | 2.8264 mL |