Physicochemical Properties
| Molecular Formula | C27H38N4O5S |
| Molecular Weight | 530.6794257164 |
| Exact Mass | 530.256 |
| CAS # | 1448189-98-7 |
| PubChem CID | 117727555 |
| Appearance | White to yellow solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 755.7±60.0 °C at 760 mmHg |
| Flash Point | 410.9±32.9 °C |
| Vapour Pressure | 0.0±2.7 mmHg at 25°C |
| Index of Refraction | 1.572 |
| LogP | 2.63 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 37 |
| Complexity | 819 |
| Defined Atom Stereocenter Count | 3 |
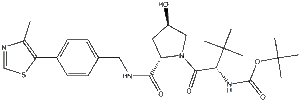
| SMILES | CC1=C(SC=N1)C2=CC=C(C=C2)CNC(=O)[C@@H]3C[C@H](CN3C(=O)[C@H](C(C)(C)C)NC(=O)OC(C)(C)C)O |
| InChi Key | PKNFPFFOAWITLF-RZUBCFFCSA-N |
| InChi Code | InChI=1S/C27H38N4O5S/c1-16-21(37-15-29-16)18-10-8-17(9-11-18)13-28-23(33)20-12-19(32)14-31(20)24(34)22(26(2,3)4)30-25(35)36-27(5,6)7/h8-11,15,19-20,22,32H,12-14H2,1-7H3,(H,28,33)(H,30,35)/t19-,20+,22-/m1/s1 |
| Chemical Name | tert-butyl ((S)-1-((2S,4R)-4-hydroxy-2-((4-(4-methylthiazol-5-yl)benzyl)carbamoyl)pyrrolidin-1-yl)-3,3-dimethyl-1-oxobutan-2-yl)carbamate |
| Synonyms | VHL Ligand 3 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
The PROTAC compound MZ1 (and related analogues MZ2, MZ3) are heterobifunctional molecules designed to bind two targets simultaneously: 1) The first or second bromodomains (BD1, BD2) of BET proteins (BRD2, BRD3, BRD4) via the JQ1 moiety. Isothermal titration calorimetry (ITC) measured binding affinities (Kd) of MZ1 for these bromodomains ranging from 115 nM to 382 nM. 2) The von Hippel-Lindau protein–ElonginB–ElonginC complex (VBC) via the VHL ligand moiety. ITC measured Kd of MZ1 for VBC was 150 nM. |
| ln Vitro |
One of two widely expressed and physiologically significant Cullin RING E3 ubiquitin ligase complexes that recognizes substrates is the VHL protein. Inducing ubiquitination and subsequent proteasomal destruction of a target protein, bifunctional PROTACs enlist VHL, one of the most well-known E3 ligases. Treatment of HeLa cells with 1 μM MZ1 for 24 hours induced complete removal (degradation) of BRD4 protein, while removal of BRD2 and BRD3 was incomplete, demonstrating a preferential degradation effect on BRD4 over its paralogs. MZ1 induced concentration-dependent degradation of BET proteins in HeLa cells, with more than 90% removal of all BET proteins observed at concentrations down to 1 μM. Preferential degradation of BRD4 was more prominent at lower concentrations (e.g., 0.1–0.5 μM). Time-course experiments in HeLa cells showed that MZ1 (1 μM or 100 nM) induced progressive and rapid removal of BET proteins over time, with BRD4 consistently exhibiting the strongest and fastest reduction. In U2OS cells transfected with GFP-BRD4, treatment with 5 μM MZ1 led to complete depletion of nuclear GFP-BRD4 fluorescence within 3 hours, as monitored by live-cell imaging. The degradation activity of MZ1 was dependent on productive recruitment of VHL, as the stereoisomer control cisMZ1 (which does not bind VHL) was completely inactive. The degradation induced by MZ1 was abrogated by co-treatment with the proteasome inhibitor MG132, confirming proteasome-dependence. Treatment with MZ1 (up to 10 μM) did not stabilize HIF-1α protein levels in HeLa cells, unlike the hypoxia-mimetic control CoCl2, indicating that it does not interfere with the natural VHL-HIF-1α interaction at effective concentrations. MZ1 treatment (1 μM and 100 nM) over 36 hours did not induce degradation of reported JQ1 off-targets DDB1 and RAD23B. The degradation effect of MZ1 was reversible. After a 4-hour pulse treatment with 1 μM MZ1 and subsequent compound washout, intracellular BRD4 levels began to recover detectably around 20 hours post-washout. Gene expression analysis in HeLa cells treated with 100 nM MZ1 for 24 hours showed downregulation of MYC and upregulation of P21 and AREG, similar to the pan-BET inhibitor JQ1. However, MZ1 had more subtle and less significant effects on FAS, TYRO3, and FGFR1 compared to JQ1, correlating with its selective BRD4 degradation profile. |
| Enzyme Assay |
Isothermal Titration Calorimetry (ITC) for Binding Affinity Measurement: The binding affinity of MZ1 for its protein targets was quantified using ITC. For BET bromodomains, titrations were performed at 30 °C. A solution of MZ1 (150 μM) was titrated into a solution of the individual bromodomain protein (first or second bromodomain of BRD2, BRD3, or BRD4) at a concentration of 15 μM. The heat change upon each injection was measured, and data were fitted to a binding model to derive the dissociation constant (Kd), enthalpy change (ΔH), and stoichiometry (N). For binding to the VBC complex, titrations were performed at 25 °C. A solution of MZ1 (150 μM) was titrated into a solution of the VBC complex (15 μM). In a reverse titration, VBC (150 μM) was titrated into MZ3 (15 μM). Control titrations with the parent VHL ligands (VHL-1, VHL-2) and the inactive control cisMZ1 were also conducted under similar conditions to benchmark the binding. |
| Cell Assay |
PROTAC Treatment and Western Blot Analysis: HeLa or U2OS cells were cultured in standard media. For degradation assays, cells were treated with the indicated concentrations of MZ1, MZ2, MZ3, cisMZ1, JQ1, or vehicle control (DMSO) for specified durations (e.g., 4, 12, 24, 36 hours). For mechanistic studies, cells were pre-treated or co-treated with 10 μM proteasome inhibitor MG132 for 1 hour prior to and during PROTAC treatment. After treatment, cells were harvested and lysed. Proteins were separated by SDS-PAGE, transferred to a membrane, and probed with specific primary antibodies against BRD2, BRD3, BRD4, HIF-1α, VHL, DDB1, RAD23B, or loading controls (e.g., β-actin). Protein levels were visualized using appropriate secondary antibodies and imaging systems. siRNA Transfection: To compare with genetic knockdown, HeLa cells were transfected with siRNA targeting individual BET proteins (BRD2, BRD3, BRD4) or a negative control siRNA using a standard transfection reagent. 24 hours after transfection, cells were treated with PROTACs, JQ1, or vehicle for an additional 24 hours before harvest for western blot analysis or gene expression analysis. Live-Cell Imaging for GFP-BRD4 Degradation: U2OS cells were transfected with a plasmid encoding GFP-tagged BRD4. 24 hours post-transfection, cells were treated with 5 μM MZ1 or cisMZ1. Fluorescence within cell nuclei was monitored and imaged over a time course of 4 hours using a live-cell imaging system. Gene Expression Analysis (RT-qPCR): HeLa cells were treated with compounds (e.g., 100 nM MZ1, JQ1, or control) for 12 or 24 hours. Total RNA was extracted, reverse transcribed into cDNA. Quantitative PCR was performed using gene-specific primers for MYC, P21, AREG, FAS, FGFR1, TYRO3, and the housekeeping gene GAPDH. Relative gene expression levels were calculated using the comparative Ct method (2^(-ΔΔCt)), normalized to the vehicle control. |
| References |
[1]. Selective Small Molecule Induced Degradation of the BET Bromodomain Protein BRD4. ACS Chem Biol. 2015;10(8):1770‐1777. |
| Additional Infomation |
MZ1 is a representative PROTAC molecule described in this study. PROTACs are heterobifunctional compounds designed to recruit an E3 ubiquitin ligase (here, VHL) to a target protein (here, BET bromodomains), leading to target ubiquitination and proteasomal degradation. The study demonstrates that linking the pan-BET inhibitor JQ1 to a high-affinity, drug-like VHL ligand (VHL-1) can result in a compound (MZ1) that induces selective degradation of BRD4 over BRD2 and BRD3, despite JQ1 itself having similar affinity for all BET bromodomains. This selectivity was unexpected and potentially arises from differences in ternary complex formation or ubiquitination efficiency. MZ1 acts as a chemical probe to dissect the biological functions of individual BET proteins, showing that selective BRD4 degradation elicits a transcriptional response distinct from pan-BET inhibition by JQ1. The VHL ligand portion used in MZ1 is derived from optimized, non-peptide small molecules (VHL-1, VHL-2) that bind to the VHL-HIF-1α interface, representing an advancement over earlier peptide-based PROTACs. The inactive control cisMZ1, which has an inverted stereochemistry at the key hydroxyproline residue critical for VHL binding, is used to confirm the VHL-dependency of the observed degradation. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~188.44 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.71 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.71 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8844 mL | 9.4219 mL | 18.8437 mL | |
| 5 mM | 0.3769 mL | 1.8844 mL | 3.7687 mL | |
| 10 mM | 0.1884 mL | 0.9422 mL | 1.8844 mL |