Physicochemical Properties
| Molecular Formula | C26H33FN4O5S |
| Molecular Weight | 532.6274 |
| Exact Mass | 532.215 |
| CAS # | 2306193-99-5 |
| Related CAS # | 2306193-99-5; |
| PubChem CID | 137628614 |
| Appearance | White to yellow solid powder |
| LogP | 3.2 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 37 |
| Complexity | 882 |
| Defined Atom Stereocenter Count | 3 |
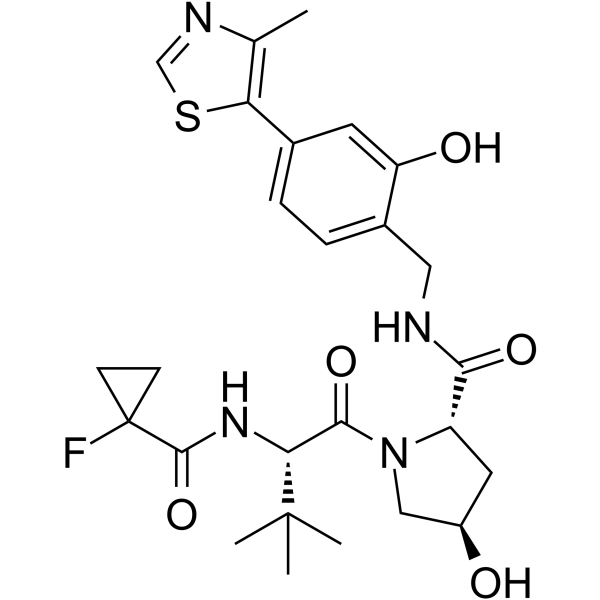
| SMILES | S1C([H])=NC(C([H])([H])[H])=C1C1C([H])=C([H])C(=C(C=1[H])O[H])C([H])([H])N([H])C([C@]1([H])C([H])([H])[C@]([H])(C([H])([H])N1C([C@]([H])(C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H])N([H])C(C1(C([H])([H])C1([H])[H])F)=O)=O)O[H])=O |
| InChi Key | OKBLHQUBMCCFKE-LVCYWYKZSA-N |
| InChi Code | InChI=1S/C26H33FN4O5S/c1-14-20(37-13-29-14)15-5-6-16(19(33)9-15)11-28-22(34)18-10-17(32)12-31(18)23(35)21(25(2,3)4)30-24(36)26(27)7-8-26/h5-6,9,13,17-18,21,32-33H,7-8,10-12H2,1-4H3,(H,28,34)(H,30,36)/t17-,18+,21-/m1/s1 |
| Chemical Name | (2S,4R)-1-[(2S)-2-[(1-fluorocyclopropanecarbonyl)amino]-3,3-dimethylbutanoyl]-4-hydroxy-N-[[2-hydroxy-4-(4-methyl-1,3-thiazol-5-yl)phenyl]methyl]pyrrolidine-2-carboxamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
VH032-cyclopropane-F targets bromodomain-containing protein 9 (BRD9) (EC50 for BRD9 degradation = 1.2 μM in SU-DHL-4 cells) [1] VH032-cyclopropane-F targets cereblon (CRBN) (E3 ubiquitin ligase) [1] |
| ln Vitro |
- BRD9 degradation activity: VH032-cyclopropane-F is a PROTAC that induces ubiquitin-dependent degradation of BRD9 in BAF complex-dependent cancer cell lines. In SU-DHL-4 (DLBCL) and OCI-Ly10 (DLBCL) cells, it degraded BRD9 with EC50 values of 1.2 μM and 1.8 μM respectively, achieving maximum degradation (>90%) at 10 μM. It showed high selectivity for BRD9 over other bromodomain proteins (BRD4, BRD7, BRD8; no significant degradation at 20 μM) [1] - Antiproliferative activity: The compound inhibited proliferation of BRD9-dependent cancer cell lines, with IC50 values of 0.8 μM (SU-DHL-4), 1.5 μM (OCI-Ly10), and 2.3 μM (RL) cells. It had no significant effect on BRD9-independent cell lines (RPMI-8226, IC50 > 20 μM) [1] - Apoptosis induction: At 5 μM, VH032-cyclopropane-F induced apoptosis in SU-DHL-4 cells with an apoptotic rate of 32%, compared to 4% in control cells. It upregulated cleaved caspase-3 and cleaved PARP levels, as detected by western blot [1] - Mechanism validation: BRD9 degradation by VH032-cyclopropane-F was reversed by CRBN inhibitor lenalidomide (10 μM), confirming CRBN-dependent degradation. It also downregulated c-Myc expression (inhibition rate 65% at 5 μM) via BRD9 depletion [1] |
| ln Vivo | - Antitumor efficacy: In SU-DHL-4 DLBCL xenograft-bearing nude mice, intraperitoneal administration of VH032-cyclopropane-F (20 mg/kg, twice weekly for 3 weeks) resulted in a tumor growth inhibition rate of 68% and reduced tumor weight by 62% compared to vehicle control. Tumor tissues from treated mice showed >80% BRD9 depletion and reduced c-Myc protein levels [1] |
| Enzyme Assay |
- BRD9 degradation kinetics assay: SU-DHL-4 cells were treated with VH032-cyclopropane-F (5 μM) for 0, 2, 4, 8, 12, and 24 hours. Cells were lysed, and BRD9 protein levels were detected by western blot. Degradation half-life (t1/2) was calculated as 3.5 hours, and maximum degradation was achieved at 8 hours [1] - CRBN dependence assay: SU-DHL-4 cells were pre-treated with lenalidomide (10 μM) for 1 hour, then treated with VH032-cyclopropane-F (5 μM) for 8 hours. Western blot analysis showed that lenalidomide blocked BRD9 degradation, confirming CRBN as the E3 ligase partner [1] - Bromodomain selectivity assay: Multiple cancer cell lines expressing different bromodomain proteins (BRD9, BRD4, BRD7, BRD8) were treated with VH032-cyclopropane-F (20 μM) for 8 hours. Western blot detected only BRD9 degradation, with no significant changes in other bromodomain proteins [1] |
| Cell Assay |
- Cell viability assay: BRD9-dependent (SU-DHL-4, OCI-Ly10, RL) and BRD9-independent (RPMI-8226) cell lines were seeded into 96-well plates at 5×10³ cells/well, treated with VH032-cyclopropane-F (0.1-20 μM) for 72 hours. Cell viability was measured by tetrazolium salt-based assay, and IC50 values were calculated [1] - Western blot assay: SU-DHL-4 cells were treated with VH032-cyclopropane-F (0.5-10 μM) for 8 hours. Cell lysates were probed with antibodies against BRD9, c-Myc, cleaved caspase-3, cleaved PARP, and GAPDH (loading control). Band intensities were quantified by densitometry [1] - Apoptosis assay: SU-DHL-4 cells were treated with VH032-cyclopropane-F (5 μM) for 24 hours, stained with Annexin V-FITC and PI, and apoptotic cells were quantified by flow cytometry [1] - Clonogenic assay: SU-DHL-4 cells were treated with VH032-cyclopropane-F (0.2-1 μM) for 24 hours, seeded into 6-well plates at 500 cells/well, and incubated for 14 days. Colonies were fixed, stained, and counted, with inhibition rate calculated relative to control [1] |
| Animal Protocol |
- DLBCL xenograft model: Female nude mice (6-8 weeks old) were subcutaneously injected with SU-DHL-4 cells (5×10⁶ cells/mouse). When tumors reached ~100 mm³, mice were randomly divided into two groups (n=6 per group): vehicle control and VH032-cyclopropane-F treatment group [1] - Drug formulation and administration: VH032-cyclopropane-F was dissolved in a mixture of DMSO, PEG400, and sterile water (volume ratio 1:4:5) to prepare the administration solution. Mice were administered intraperitoneally at 20 mg/kg, twice weekly for 3 weeks. The control group received an equal volume of the vehicle mixture [1] - Tumor monitoring and tissue analysis: Tumor volume was measured every 3 days (volume = length × width² / 2), and body weight was recorded weekly. At the end of treatment, mice were sacrificed, tumors were excised, weighed, and homogenized for western blot analysis of BRD9 and c-Myc protein levels [1] |
| ADME/Pharmacokinetics |
- Plasma protein binding: VH032-cyclopropane-F had a plasma protein binding rate of 87.5 ± 2.3% in human plasma, determined by equilibrium dialysis [1] - In vitro metabolic stability: The compound showed moderate metabolic stability in human liver microsomes, with a half-life (t1/2) of 2.9 hours and a metabolic clearance rate of 0.58 mL/min/mg protein [1] |
| Toxicity/Toxicokinetics |
- In vitro toxicity: VH032-cyclopropane-F showed low cytotoxicity to normal human peripheral blood mononuclear cells (PBMCs) with IC50 > 20 μM [1] - In vivo toxicity: In xenograft-bearing mice treated with VH032-cyclopropane-F (20 mg/kg, 3 weeks), no significant body weight loss (≤ 5% of initial weight) or histopathological abnormalities were observed in major organs (heart, liver, spleen, lungs, kidneys) [1] |
| References |
[1]. BAF complex vulnerabilities in cancer demonstrated via structure-based PROTAC design. Nat Chem Biol. 2019 Jul;15(7):672-680. [2]. FAK-targeting PROTAC demonstrates enhanced antitumor activity against KRAS mutant non-small cell lung cancer. Exp Cell Res. 2021 Nov 15;408(2):112868. |
| Additional Infomation |
- Chemical classification: VH032-cyclopropane-F is a proteolysis-targeting chimera (PROTAC) composed of a BRD9-binding moiety (VH032 derivative), a cyclopropane linker, and a CRBN-recruiting moiety [1] - Mechanism of action: The compound forms a ternary complex with BRD9 (target protein) and CRBN (E3 ubiquitin ligase), inducing ubiquitination and proteasomal degradation of BRD9, thereby inhibiting BRD9-dependent oncogenic signaling (e.g., c-Myc pathway) in cancer cells [1] - Target background: BRD9 is a core subunit of the BAF (SWI/SNF) chromatin remodeling complex, which is dysregulated in various hematological malignancies (e.g., DLBCL). BRD9 depletion leads to growth arrest and apoptosis of BAF-dependent cancer cells [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~187.75 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.69 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.69 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (4.69 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8775 mL | 9.3874 mL | 18.7748 mL | |
| 5 mM | 0.3755 mL | 1.8775 mL | 3.7550 mL | |
| 10 mM | 0.1877 mL | 0.9387 mL | 1.8775 mL |