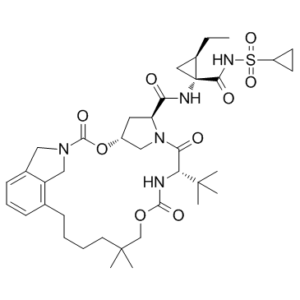
Vaniprevir (formerly known as MK-7009) is a non-covalent competitive inhibitor of the hepatitis C virus (HCV) NS3/4A protease. It is is a macrocyclic that is being developed by Merck & Co., and is currently in clinical testing. In Japan, it was approved in Japan for treating hepatitis C in 2014 under the brand name Vanihep. Vaniprevir is active against both the genotype 1 and genotype 2 NS3/4a protease enzymes and has good plasma exposure and excellent liver exposure in multiple species. Vaniprevir is a potent HCV protease inhibitor with a predictable resistance profile and favorable safety profile that is suitable for QD or BID administration.
Physicochemical Properties
| Molecular Formula | 757.94 |
| Molecular Weight | C38H55N5O9S |
| Exact Mass | 757.372 |
| CAS # | 923590-37-8 |
| PubChem CID | 24765256 |
| Appearance | White to off-white solid powder |
| Density | 1.33g/cm3 |
| LogP | 5.273 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 53 |
| Complexity | 1550 |
| Defined Atom Stereocenter Count | 5 |
| SMILES | CC[C@@H]1C[C@@]1(C(=O)NS(=O)(=O)C2CC2)NC(=O)[C@@H]3C[C@@H]4CN3C(=O)[C@@H](NC(=O)OCC(CCCCC5=C6CN(CC6=CC=C5)C(=O)O4)(C)C)C(C)(C)C |
| InChi Key | KUQWGLQLLVFLSM-ONAXAZCASA-N |
| InChi Code | InChI=1S/C38H55N5O9S/c1-7-25-18-38(25,33(46)41-53(49,50)27-14-15-27)40-31(44)29-17-26-20-43(29)32(45)30(36(2,3)4)39-34(47)51-22-37(5,6)16-9-8-11-23-12-10-13-24-19-42(21-28(23)24)35(48)52-26/h10,12-13,25-27,29-30H,7-9,11,14-22H2,1-6H3,(H,39,47)(H,40,44) |
| Chemical Name | Cyclopropanecarboxamide, N-(((6-(2-carboxy-2,3-dihydro-1H-isoindol-4-yl)-2,2-dimethylhexyl)oxy)carbonyl)-3-methyl-L-valyl-(4R)-4-hydroxy-L-prolyl-1-amino-N-(cyclopropylsulfonyl)-2-ethyl-, (1-2)-lactone, (1R,2R)- |
| Synonyms | MK7009; MK-7009; MK 7009 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | MK-7009, an inhibitor of genotype 1a and 1b proteases at subnanomolar concentrations with modestly shifted potency against genotype 2a and 2b proteases at low nanomolar concentrations. Potent activity was also observed in a cell-based HCV replicon assay in the presence of added human serum (50%). [1] |
| ln Vivo | In multiple species evaluated in preclinical studies, the MK-7009 concentrations in the liver were maintained at a significant multiple of the cell-based replicon 50% effective concentration over 12 to 24 h following the administration of moderate oral doses (5 to 10 mg per kg of body weight). MK-7009 also had excellent selectivity against both a range of human proteases and a broad panel of pharmacologically relevant ion channels, receptors, and enzymes. [1] |
| Cell Assay |
Broad counterscreening of the activity of MK-7009 against a panel of 169 ion channels, receptors, and enzymes was carried out at MDS Pharma Services, in which MK-7009 was evaluated for its activity at a concentration of 10 μM.[1] Inhibition of the cell-based HCV replicon assay by combinations of MK-7009 and either interferon alfa 2b or MK-0608, a nucleoside analog inhibitor of the HCV RNA-dependent RNA polymerase , was determined. MK-7009 and the second inhibitor were placed at various concentrations in a matrix in a 96-well plate format. [1] |
| Animal Protocol |
For studies in which MK-7009 was dosed intravenously to fasted rats, dogs, and rhesus monkeys, the compound was administered as a bolus (1.0, 0.1, and 0.1 ml/kg of body weight, respectively) in dimethyl sulfoxide. [1] Two HCV-uninfected chimpanzees (weights, approximately 57 and 52 kg) were dosed orally by the voluntary ingestion of MK-7009 in a chocolate milk vehicle (0.67 ml/kg; Nestle brand).[1] |
| References |
[1]. Liverton, Nigel J.; Carroll, Steven S.; Di Muzio, Jillian;.MK-7009, a potent and selective inhibitor of hepatitis C virus NS3/4A protease. Antimicrobial Agents and Chemotherapy (2009), Volume Date 2010, 54(1), 305-311. [2]. Kong J, Chen CY, Balsells-Padros J, Cao Y, Dunn RF, Dolman SJ, Janey J, Li H, Zacuto MJ.Synthesis of the HCV protease inhibitor Vaniprevir (MK-7009) using ring-closing metathesis strategy.J Org Chem. 2012 Apr 20;77(8):3820-8. Epub 2012 Apr 10. [3]. Manns MP, Gane E, Rodriguez-Torres M, Stoehr A, Yeh CT, Marcellin P, Wiedmann RT, Hwang PM, Caro L, Barnard RJ, Lee AW; for the MK-7009 Protocol 007 Study Group.Vaniprevir with pegylated interferon alpha-2a and ribavirin in treatment-na?ve patients with c. [4]. Song ZJ, Tellers DM, Journet M, Kuethe JT, Lieberman D, Humphrey G, Zhang F, Peng Z, Waters MS, Zewge D, Nolting A, Zhao D, Reamer RA, Dormer PG, Belyk KM, Davies IW, Devine PN, Tschaen DM.Synthesis of vaniprevir (MK-7009): lactamization to prepare a 20-m. [5]. McCauley JA, McIntyre CJ, Rudd MT, Nguyen KT, Romano JJ, Butcher JW, Gilbert KF, Bush KJ, Holloway MK, Swestock J, Wan BL, Carroll SS, DiMuzio JM, Graham DJ, Ludmerer SW, Mao SS, Stahlhut MW, Fandozzi CM, Trainor N, Olsen DB, Vacca JP, Liverton NJ.Discove. |
| Additional Infomation | Vaniprevir has been used in trials studying the treatment and diagnostic of Hepatitis C, Chronic Hepatitis C, Hepatitis C, Chronic, Chronic Hepatitis C Infection, and Chronic Genotype 1 Hepatitis C Virus Infection. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |