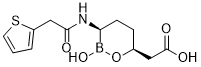
Vaborbactam (formerly PX-7009; MP-7009; RPX7009; trade name: Vabomere) is novel, potent and broad spectrum non-β-lactam (cyclic boronic acid pharmacophore) β-lactamase inhibitor discovered by Rempex Pharmaceuticals and approved in 2017 for the treatment of complicated urinary tract infections and pyelonephritis. It is not effective as an antibiotic by itself, but it restores the potency of existing antibiotics by inhibiting the beta-lactamase enzymes that would otherwise degrade them. When combined with an appropriate antibiotic it can be used for the treatment of gram-negative bacterial infections. In the United States, the combination drug meropenem/vaborbactam (Vabomere) was approved by the FDA (Food and Drug Administration) on 8/29/2017 for treating complicated urinary tract infections and pyelonephritis.
Physicochemical Properties
| Molecular Formula | C12H16BNO5S | |
| Molecular Weight | 297.13 | |
| Exact Mass | 297.084 | |
| Elemental Analysis | C, 48.51; H, 5.43; B, 3.64; N, 4.71; O, 26.92; S, 10.79 | |
| CAS # | 1360457-46-0 | |
| Related CAS # |
|
|
| PubChem CID | 56649692 | |
| Appearance | Light brown to yellow solid powder | |
| Density | 1.4±0.1 g/cm3 | |
| Index of Refraction | 1.572 | |
| Hydrogen Bond Donor Count | 3 | |
| Hydrogen Bond Acceptor Count | 6 | |
| Rotatable Bond Count | 5 | |
| Heavy Atom Count | 20 | |
| Complexity | 370 | |
| Defined Atom Stereocenter Count | 2 | |
| SMILES | S1C([H])=C([H])C([H])=C1C([H])([H])C(N([H])[C@]1([H])B(O[H])O[C@]([H])(C([H])([H])C(=O)O[H])C([H])([H])C1([H])[H])=O |
|
| InChi Key | IOOWNWLVCOUUEX-WPRPVWTQSA-N | |
| InChi Code | InChI=1S/C12H16BNO5S/c15-11(7-9-2-1-5-20-9)14-10-4-3-8(6-12(16)17)19-13(10)18/h1-2,5,8,10,18H,3-4,6-7H2,(H,14,15)(H,16,17)/t8-,10-/m0/s1 | |
| Chemical Name | 2-((3R,6S)-2-hydroxy-3-(2-(thiophen-2-yl)acetamido)-1,2-oxaborinan-6-yl)acetic acid | |
| Synonyms | RPX-7009; MP-7009; RPX7009; MP7009; REBO07; REBO 07; MP7; MP 7; RPX 7009; MP 7009; REBO-07; MP-7; Trade name: Vabomere | |
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Vaborbactam exhibits a wide range of β-lactamase inhibition, with a strong affinity for KPC, CTX-M, SHV, and CMY enzymes[1]. When the concentration of the inhibitor is fixed at ≥8 μg/mL, maximum potentiation is achieved (≥96.5% of isolates are inhibited at ≤2 μg/mL of SM 7338-vaborbactam). Vaborbactam restores SM 7338 activity for 72.7 to 98.1% of CPE isolates at ≤2 μg/mL. At a fixed concentration of 8 μg/mL, SM 7338-vaborbactam inhibits 93.7% of CPE isolates that show elevated SM 7338 MICs at ≤1 μg/mL (MIC50, ≤0.06 μg/mL for all organisms)[2]. Vaborbactam functions as a competitive inhibitor and prevents the b-lactamase from hydrolyzing it by creating a reversible dative bond with the blactamase[3]. | |
| ln Vivo |
|
|
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion The peak plasma concentrations (Cmax) and AUC of vaborbactam increase in a dose-proportional manner. In healthy adult subjects, the Cmax following administration of multiple 2 g dose as a 3-hour infusion was 55.6 mg/L and AUC was 588 mg•h/L. In patients with the same dosing regimen, the Cmax was 71.3 mg/L and AUC was 835 mg•h/L at steady state. The exposure of vaborbactam in terms of Cmax and AUC are not expected to change with repeated dosing, and there was no evidence of accumulation of vaborbactam in plasma in a repeated dosing study. Vaborbactam predominantly undergoes renal excretion, where about 75 to 95% of the dose is excreted unchanged in the urine over a 24 to 48 hour period. The steady-state volume of distribution of vaborbactam in patients was 18.6 L. The mean renal clearance for vaborbactam was 8.9 L/h. The mean non-renal clearance for vaborbactam was 2.0 L/h indicating nearly complete elimination of vaborbactam by the renal route. The clearance of vaborbactam in healthy subjects following administration of multiple doses of 2 g as a 3-hour infusion was 10.9 L/h. The clearance of vaborbactam in patients following administration of 2 g by 3 hour infusion was 7.95 L/h. Metabolism / Metabolites Vaborbactam does not undergo metabolism. Biological Half-Life The half life of vaborbactam in healthy subjects following multiple 2 g dose administration as a 3-hour infusion was 1.68 hours. The half life of vaborbactam following administration of 2 g by 3 hour infusion was 2.25 hours. |
|
| Toxicity/Toxicokinetics |
Protein Binding The average serum protein binding of vaborbactam is approximately 33%. |
|
| References |
[1]. Discovery of a Cyclic Boronic Acid β-Lactamase Inhibitor (RPX7009) with Utility vs Class A Serine Carbapenemases. J Med Chem. 2015 May 14;58(9):3682-92. [2]. Effect of the β-Lactamase Inhibitor Vaborbactam Combined with SM 7338 against Serine Carbapenemase-Producing Enterobacteriaceae. Antimicrob Agents Chemother. 2016 Aug 22;60(9):5454-8. [3]. Novel Beta-Lactamase Inhibitors: Unlocking Their Potential in Therapy. |
|
| Additional Infomation |
Vaborbactam is a β-lactamase inhibitor based on a cyclic boronic acid pharmacophore. It has been used in trials investigating the treatment of bacterial infections in subjects with varying degrees of renal insufficiency. In August 2017, a combination antibacterial therapy under the market name Vabomere was approved by the FDA for the treatment of adult patients with complicated urinary tract infections (cUTI). Vabomere consists of vaborbactam and [Meropenem] for intravenous administration. Vaborbactam is added to the therapy to reduce the extent meropenem degradation by inhibiting the serine beta-lactamases expressed by the microorganism of target. The treatment aims to resolve infection-related symptoms of cUTI and achieve negative urine culture, when the infections are proven or strongly suspected to be caused by susceptible bacteria. Vaborbactam is a beta Lactamase Inhibitor. The mechanism of action of vaborbactam is as a beta Lactamase Inhibitor. Drug Indication Indicated in combination with meropenem for the treatment of patients 18 years of age and older with complicated urinary tract infections (cUTI) including pyelonephritis caused by the following susceptible microorganisms: _Escherichia coli_, _Klebsiella pneumoniae_, and _Enterobacter cloacae_ species complex. FDA Label Treatment of Gram-negative bacterial infections Mechanism of Action Vaborbactam is a cyclic boronic acid pharmacophore β-lactamase inhibitor that elicits potent inhibition of _Klebsiella pneumoniae_ carbapenemase (KPC) enzymes and other Ambler class A and C enzymes such as serine β-lactamases that confer resistance to commonly-used antibiotics such as Carbapenems. Vaborbactam is a potent inhibitor of class A carbapenemases, such as KPC, as well as an inhibitor of other class A (CTX-M, SHV, TEM) and class C (P99, MIR, FOX) beta-lactamases. Vaborbactam interacts with β-lactamases of Ambler classes A and C via precovalent and covalent binding. It exerts no inhibitory actions on class D or class B carbapenemases. The production of contemporary β-lactamase by bacterial isolates potentiate the degradation of β-lactam antibiotic agents, rendering them clinically ineffective and posing challenges for patients receiving the standard antibiotic therapy. In combination with meropenem, varborbactam acts as a non-suicidal beta-lactamase inhibitor that protects meropenem from degradation mediated by serine beta-lactamases such as _Klebsiella pneumoniae_ carbapenemase (KPC). Pharmacodynamics Vaborbactam shows no antibacterial activity alone; it serves to restore the antibacterial activity of other antibacterial agents such as meropenem by attenuating their degradation by inhibiting certain serine beta-lactamases of microorganisms. Vaborbactam does not decrease the activity of meropenem against meropenem-susceptible organisms. Vaborbactam in combination with meropenem, which is a penem antibacterial drug, potentiates the bactericidal actions of meropenem against carbapenem-resistant KPC-containing _Escherichia coli_, _Klebsiella pneumoniae_, and _Enterobacter cloacae_ in a concentration-dependent manner. It restored the antimicrobial activity of meropenem in animal models of infection caused by some meropenem non-susceptible KPC-producing Enterobacteriaceae. |
Solubility Data
| Solubility (In Vitro) | H2O : ~5.26 mg/mL (~17.70 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 25 mg/mL (84.14 mM) in 108 mM sodium carbonate (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. Solubility in Formulation 2: ≥ 2.62 mg/mL (8.82 mM) (saturation unknown) in 5% DMSO + 40% PEG300 + 5% Tween80 + 50% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 3: ≥ 2.62 mg/mL (8.82 mM) (saturation unknown) in 5% DMSO + 95% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 4: 27.5 mg/mL (92.55 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.3655 mL | 16.8277 mL | 33.6553 mL | |
| 5 mM | 0.6731 mL | 3.3655 mL | 6.7311 mL | |
| 10 mM | 0.3366 mL | 1.6828 mL | 3.3655 mL |