Physicochemical Properties
| Molecular Formula | C22H38O5 |
| Molecular Weight | 382.53412 |
| Exact Mass | 382.271 |
| Elemental Analysis | C, 69.08; H, 10.01; O, 20.91 |
| CAS # | 120373-36-6 |
| PubChem CID | 5311236 |
| Appearance | Colorless to light yellow liquids |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 562.9±40.0 °C at 760 mmHg |
| Flash Point | 308.3±23.8 °C |
| Vapour Pressure | 0.0±3.5 mmHg at 25°C |
| Index of Refraction | 1.509 |
| LogP | 3.19 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 15 |
| Heavy Atom Count | 27 |
| Complexity | 460 |
| Defined Atom Stereocenter Count | 4 |
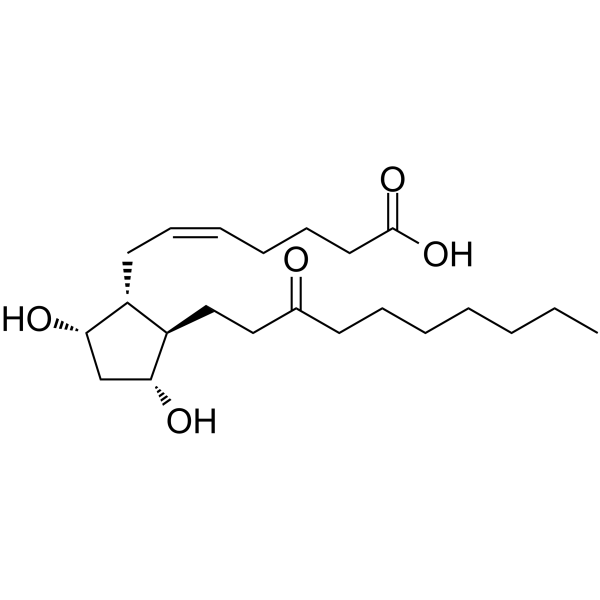
| SMILES | CCCCCCCC(CC[C@H]1C(O)CC(O)C1C/C=C/CCCC(=O)O)=O |
| InChi Key | TVHAZVBUYQMHBC-SNHXEXRGSA-N |
| InChi Code | InChI=1S/C22H38O5/c1-2-3-4-5-8-11-17(23)14-15-19-18(20(24)16-21(19)25)12-9-6-7-10-13-22(26)27/h6,9,18-21,24-25H,2-5,7-8,10-16H2,1H3,(H,26,27)/b9-6-/t18-,19-,20+,21-/m1/s1 |
| Chemical Name | (Z)-7-[(1R,2R,3R,5S)-3,5-dihydroxy-2-(3-oxodecyl)cyclopentyl]hept-5-enoic acid |
| Synonyms | Unoprostone; 120373-36-6; Unoprostone [MI]; Unoprostone [INN]; 13,14-dihydro-15-keto-20-ethyl PGF2alpha; Unoprostane; CHEBI:39455; 6X4F561V3W; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Anti-ocular hypertension; BK channels |
| ln Vitro | One hour prior to H2O2 treatment, pretreatment with unoprostone (0.01, 0.1, and 1 μM) inhibits H2O2-induced cell mortality in a concentration-dependent manner, showing maximal effects at 0.1 μM and 1 μM concentrations [1]. Pretreatment with unoprostone at doses ranging from 0.1 to 3 μM inhibited light-induced apoptosis in a concentration-dependent fashion; at concentrations of 1 and 3 μM, the impact was noteworthy. Unoprostone decreases morphological alterations and dramatically prevents low mitochondrial membrane potential and cell death brought on by light irradiation [1]. With a Ki of 3.86 μM, unoprostone exhibits binding affinity to the prostaglandin F2α receptor (FP) [2]. |
| ln Vivo |
In a dose-dependent way, unoprostone (0.06%, 0.12%; local instillation; 3 µl) lowers intraocular pressure (IOP) in mice [3]. The baseline IOP (mean +/- SEM) in WT and FPKO mice was 15.0 +/- 0.2 and 15.0 +/- 0.3 mm Hg, respectively, during the day, and 18.9 +/- 0.4 and 19.2 +/- 0.4 mm Hg, respectively, at night. In WT mice, latanoprost significantly lowered IOP both during the day and at night, at 2 to 6 hours and 1 to 6 hours after application, respectively. Maximal IOP reduction was observed at 3 hours after drug instillation both during the day (10.9 +/- 1.8%) and at night (23.2 +/- 1.1%). At 3 hours after instillation, latanoprost (10.9 +/- 1.8% and 23.2 +/- 1.1%, daytime and nighttime, respectively), travoprost (15.9 +/- 1.4% and 26.1 +/- 1.2%) and bimatoprost (8.8 +/- 2.0 and 19.8 +/- 1.5%) significantly lowered IOP in WT mice both during the day and at night; isopropyl unoprostone significantly lowered IOP at night (13.7 +/- 1.9%) but not during the day (5.3 +/- 3.2%). In FPKO mice, latanoprost, travoprost, bimatoprost, and unoprostone showed no significant IOP-lowering effect. Bunazosin significantly lowered IOP in both WT (22.1 +/- 1.6%) and FPKO mice (22.2 +/- 2.1%). Conclusions: A single application of latanoprost, travoprost, bimatoprost, or unoprostone had no effect on IOP in FPKO mice with presumed functional uveoscleral outflow pathways. The prostanoid FP receptor plays a crucial role in the mechanism of early IOP lowering of all commercially available prostaglandin analogues.[3] |
| Cell Assay |
Cell Viability Assay[1] Cell Types: H2O2-induced photoreceptor cells Tested Concentrations: 0.01, 0.1, 1 μM Incubation Duration: 1 hour before H2O2 treatment Experimental Results: Prevents H2O2-induced cell death in a concentration-dependent manner. |
| Animal Protocol |
Animal/Disease Models: Male mice (6 weeks old) [3] Doses: 0.06%, 0.12% Route of Administration: local infusion; 3 µl Experimental Results: Reduce IOP in mice in a dose-dependent manner. FPKO and wild-type (WT) mice were bred and acclimatized under a 12-hour light-dark cycle. IOP was measured under general anesthesia by a microneedle. Method: To evaluate the effects of each drug, a single drop (3 muL) of each drug solution was topically applied in a masked manner to a randomly selected eye. IOP reduction was evaluated by the difference in IOP between the treated eye and the untreated contralateral eye in the same mouse. First, the diurnal variation and baseline IOP in WT and FPKO mice were measured. Then, to determine the window feasible for demonstrating the most marked ocular hypotensive effect, 0.005% latanoprost was applied to WT mice during the day or at night. The time when the ocular hypotensive effect was larger was selected for further studies to evaluate the effects of latanoprost (0.005%), travoprost (0.004%), bimatoprost (0.03%), and unoprostone (0.12%). In addition, bunazosin (0.1%) was also applied to demonstrate functional uveoscleral outflow in FPKO mice. All experiments were conducted under a masked study design.[3] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion After application to the eye, unoprostone isopropyl is absorbed through the cornea and conjunctival epithelium where it is hydrolyzed by esterases to unoprostone free acid. mean peak unoprostone free acid plasma concentration was <1.5 ng/mL and dro pped below the lower limit of quantitation (<0.250 ng/mL) 1 hour following instillation, indicating low systemic absorption and rapid plasma excretion. Elimination of unoprostone free acid from human plasma is rapid. Plasma levels of unoprostone free acid dropped below the lower limit of quantitation ( < 0.25 ng/mL) 1 hour following ocular instillation. Urinary elimination is the predominant elimination route. Intended for local use only, very low systemic absorption. Intended for local use only, very low systemic absorption. Metabolism / Metabolites After ocular application, unoprostone isopropyl is hydrolyzed by esterases in the cornea to its biological active metabolite, unoprostone free acid. Unoprostone free acid is then metabolized to several inactive metabolites with lower molecular weight and increased polarity via ε- or β-oxidation. No secondary conjugation is found and no significant effect on hepatic microsomal enzyme activity has been observed. Biological Half-Life Half-life of Unoprostone is 14 minutes. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation No information is available on the use of unoprostone during breastfeeding. Because of its short half-life it is not likely to reach the bloodstream of the infant or cause any adverse effects in breastfed infants. Professional guidelines consider prostaglandin eye drops acceptable during breastfeeding. To substantially diminish the amount of drug that reaches the breastmilk after using eye drops, place pressure over the tear duct by the corner of the eye for 1 minute or more, then remove the excess solution with an absorbent tissue. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding Intended for local use only, very low systemic absorption. |
| References |
[1]. Unoprostone reduces oxidative stress- and light-induced retinal cell death, and phagocytoticdysfunction, by activating BK channels. Mol Vis. 2011;17:3556-65. Epub 2011 Dec 30. [2]. Real-time intracellular Ca2+ mobilization by travoprost acid, bimatoprost, unoprostone, and otheranalogs via endogenous mouse, rat, and cloned human FP prostaglandin receptors. J Pharmacol Exp Ther. 2003 Jan;304(1):238-45. [3]. The effects of prostaglandin analogues on IOP in prostanoid FP-receptor-deficient mice. Invest Ophthalmol Vis Sci. 2005 Nov;46(11):4159-63. |
| Additional Infomation |
Unoprostone is an oxo monocarboxylic acid, a prostaglandins Falpha and a ketone. It has a role as an antiglaucoma drug and an antihypertensive agent. Unoprostone isopropyl is a prostaglandin analogue. Ophthalmic Solution 0.15% is a synthetic docosanoid. Unoprostone isopropyl has the chemical name isopropyl (+)-(Z)-7-[(1R,2R,3R,5S)-3,5 dihydroxy-2-(3-oxodecyl)cyclopentyl]-5-heptenoate. The main indication of Unoprostane is treatment of glucoma. Unoprostone is a Prostaglandin Analog. Drug Indication For the lowering of intraocular pressure in patients with open-angle glaucoma or ocular hypertension who are intolerant of other intraocular pressure lowering medications or insufficiently responsive (failed to achieve target IOP determined after multiple measurements over time) to another intraocular pressure lowering medication. FDA Label Mechanism of Action Unoprostone is believed to reduce elevated intraocular pressure (IOP), by increasing the outflow of aqueous humor. The mechanism of action for the IOP-lowering effect of unoprostone is controversial. Early studies showed that unoprostone increases aqueous humor outflow through the uveoscleral pathway similar to the 20-carbon prostaglandin analogs, such as latanoprost.8 More recent evidence, however, shows that it may work, at least in part, through stimulation of Ca2+-activated BK and CIC-2 type channels, leading to increased trabecular meshwork outflow. Pharmacodynamics Unoprostone will begin to reduce IOP 30 minutes after ocular instillation. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6142 mL | 13.0709 mL | 26.1417 mL | |
| 5 mM | 0.5228 mL | 2.6142 mL | 5.2283 mL | |
| 10 mM | 0.2614 mL | 1.3071 mL | 2.6142 mL |