Physicochemical Properties
| Molecular Formula | C22H29FN6 |
| Molecular Weight | 396.504267454147 |
| Exact Mass | 396.243 |
| Elemental Analysis | C, 66.64; H, 7.37; F, 4.79; N, 21.20 |
| CAS # | 1350547-65-7 |
| Related CAS # | 1350547-65-7 |
| PubChem CID | 53355503 |
| Appearance | White to yellow solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 579.4±58.0 °C at 760 mmHg |
| Flash Point | 304.2±32.3 °C |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C |
| Index of Refraction | 1.663 |
| LogP | 4.16 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 29 |
| Complexity | 492 |
| Defined Atom Stereocenter Count | 0 |
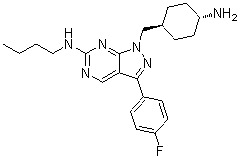
| SMILES | FC1C=CC(=CC=1)C1C2=CN=C(NCCCC)N=C2N(CC2CCC(CC2)N)N=1 |
| InChi Key | OGEBRHQLRGFBNV-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C22H29FN6/c1-2-3-12-25-22-26-13-19-20(16-6-8-17(23)9-7-16)28-29(21(19)27-22)14-15-4-10-18(24)11-5-15/h6-9,13,15,18H,2-5,10-12,14,24H2,1H3,(H,25,26,27) |
| Chemical Name | 1-(((1r,4r)-4-aminocyclohexyl)methyl)-N-butyl-3-(4-fluorophenyl)-1H-pyrazolo[3,4-d]pyrimidin-6-amine |
| Synonyms | UNC569; UNC-569; UNC 569 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Mer (IC50 = 2.9 nM); Axl (IC50 = 37 nM); Tyro3 (IC50 = 48 nM); Mer (Ki = 4.3 nM) UNC569 is a Mer receptor tyrosine kinase inhibitor (IC50 = 3.0 nM). It also inhibits the related TAM family kinases Axl (IC50 = 16 nM) and Tyro3 (IC50 = 150 nM).[1] |
| ln Vitro |
In all cell lines, UNC569 (24 hours) elevated short-term PARP[2] and Caspase 3 levels as well as cellular fluorescence. Potent suppression of activated Mer and downstream signaling, including PI3K, is processed by UNC569 (1 μM; 1.5 hr). In human B-ALL (acute lymphoblastic leukemia) 697 and Jurkat cell lines, UNC569 (1 hr) inhibits Mer phosphorylation levels with IC50 values of 141 nM and 193 nM [1]. UNC569 inhibited Mer kinase activity in a biochemical assay with an IC50 of 3.0 nM and a Ki of 2.0 nM.[1] In a selectivity panel against 72 kinases, tested at a concentration 10-fold higher than its Mer IC50, UNC569 inhibited only a few other kinases by greater than 50%: FLT3 (82%), MAPKAPK2 (92%), Ret (59%), and its mutant Ret Y791F (56%).[1] In human pre-B acute lymphoblastic leukemia (ALL) 697 cells, UNC569 inhibited Mer autophosphorylation with a cell-based IC50 of 141 nM.[1] |
| ln Vivo |
The pharmacokinetic characteristics of UNC569 (3 mg/kg) in vivo were assessed via in vitro (IV) and intravenous (PO) methods. Low systemic clearance (19.5 mL/min/kg), good wound bioavailability (57%) and high vial distribution (Vss of 5.83 L/kg) are characteristics of UNC569 [1]. For two weeks, bleached zebrafish were immersed in 4 μM UNC569. Comparing UNC569 to vehicle- and mock-treated fish, the results demonstrated a more than 50% reduction in tumor burden [2]. In a transgenic zebrafish model of MYC-driven T-ALL, continuous treatment with 4 µM UNC569 for 2 weeks via immersion induced an average 47.8% regression in tumor burden (as quantified by GFP fluorescence), which was statistically superior to control groups (DMSO or mock treatment). 10 out of 18 treated fish showed >50% disease regression, with no apparent toxicity observed. [2] |
| Enzyme Assay |
Mer kinase inhibition was measured using a microfluidic capillary electrophoresis (MCE) assay. The assay was performed at the ATP Km concentration. Phosphorylated and unphosphorylated substrate peptides were separated and analyzed using a lab-on-a-chip reader system. The percentage of inhibition was determined, and IC50 values were calculated.[1] |
| Cell Assay |
Apoptosis analysis [2] Cell Types: 697 and Jurkat cells Tested Concentrations: 0.4μM, 0.8μM, 1μM, 1.2μM, 1.4μM, 1.6μM, 1.8μM, 2μM Incubation Duration: 24 hrs (hours) Experimental Results: Induction of apoptosis. in all cell lines. Western Blot Analysis[2] Cell Types: 697 and Jurkat cells Tested Concentrations: 1 µM Incubation Duration: 1.5 hrs (hours) Experimental Results: Inhibition of Mer activation and downstream signaling through ERK1/2 and AKT. To measure inhibition of Mer autophosphorylation in cells, human pre-B leukemia 697 cells were treated with various concentrations of UNC569 for 1 hour. A phosphatase inhibitor was then added to stabilize phosphorylated Mer. Cells were lysed, and Mer protein was immunoprecipitated from the lysates. Phospho-Mer levels were detected by Western blot using a rabbit polyclonal antibody raised against a triphosphorylated Mer peptide. Band intensities were quantified by densitometry, and IC50 values were determined using nonlinear regression analysis.[1] |
| Animal Protocol |
The in vivo pharmacokinetic properties of UNC569 were assessed in mice. The compound was administered via both intravenous (IV) and oral (PO) routes. Specific details regarding the formulation, dosing frequency, and animal strain used are provided in the Supporting Information of the publication but are not detailed in the main text.[1] |
| ADME/Pharmacokinetics |
In mice, UNC569 exhibited low systemic clearance (19.5 mL/min/kg), a high volume of distribution (Vss = 5.83 L/kg), and good oral bioavailability (57%).[1] |
| References |
[1]. Discovery of Novel Small Molecule Mer Kinase Inhibitors for the Treatment of Pediatric Acute Lymphoblastic Leukemia. ACS Med Chem Lett. 2012 Feb 9;3(2):129-134. [2]. UNC569, a novel small-molecule mer inhibitor with efficacy against acute lymphoblastic leukemia in vitro and in vivo. Mol Cancer Ther. 2013 Nov;12(11):2367-77. |
| Additional Infomation |
UNC569 is a pyrazolopyrimidine derivative and a lead compound discovered through structure-based design for inhibiting Mer kinase.[1] The co-crystal structure of UNC569 bound to the Mer kinase domain was solved at 2.69 Å resolution (PDB: 3TCP). The structure revealed an unexpected binding mode where the inhibitor is confined to the adenine pocket and does not extend into the hydrophobic back pocket, partly due to a unique "gate" formed by residues including Ile650 in Mer.[1] UNC569 is described as having a promising selectivity profile and excellent drug metabolism and pharmacokinetic (DMPK) properties, making it a viable lead candidate for further optimization toward clinical applications.[1] The study rationale is based on Mer being ectopically expressed in pediatric ALL and contributing to pro-survival signaling and chemoresistance, making it a therapeutic target.[1] |
Solubility Data
| Solubility (In Vitro) | DMSO: ~31.3 mg/mL (~78.8 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.31 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5221 mL | 12.6103 mL | 25.2207 mL | |
| 5 mM | 0.5044 mL | 2.5221 mL | 5.0441 mL | |
| 10 mM | 0.2522 mL | 1.2610 mL | 2.5221 mL |